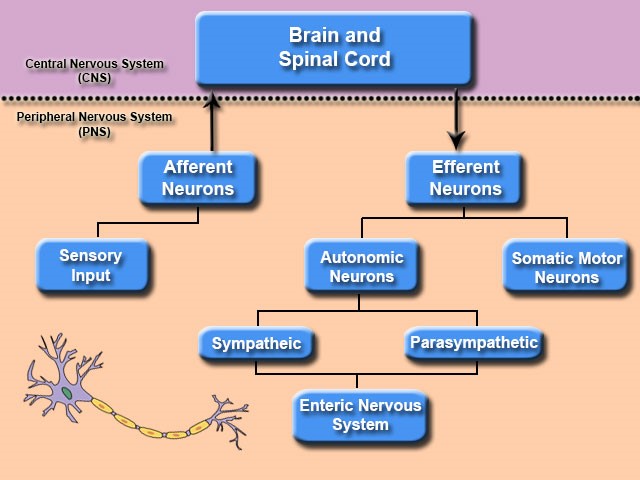
CNS Embryology & Brain Hemispheres
Embryology of the Central Nervous System (CNS)
The development of the nervous system begins very early in embryonic life and is a highly complex and tightly regulated process.
1. Neural Plate Formation (Week 3)
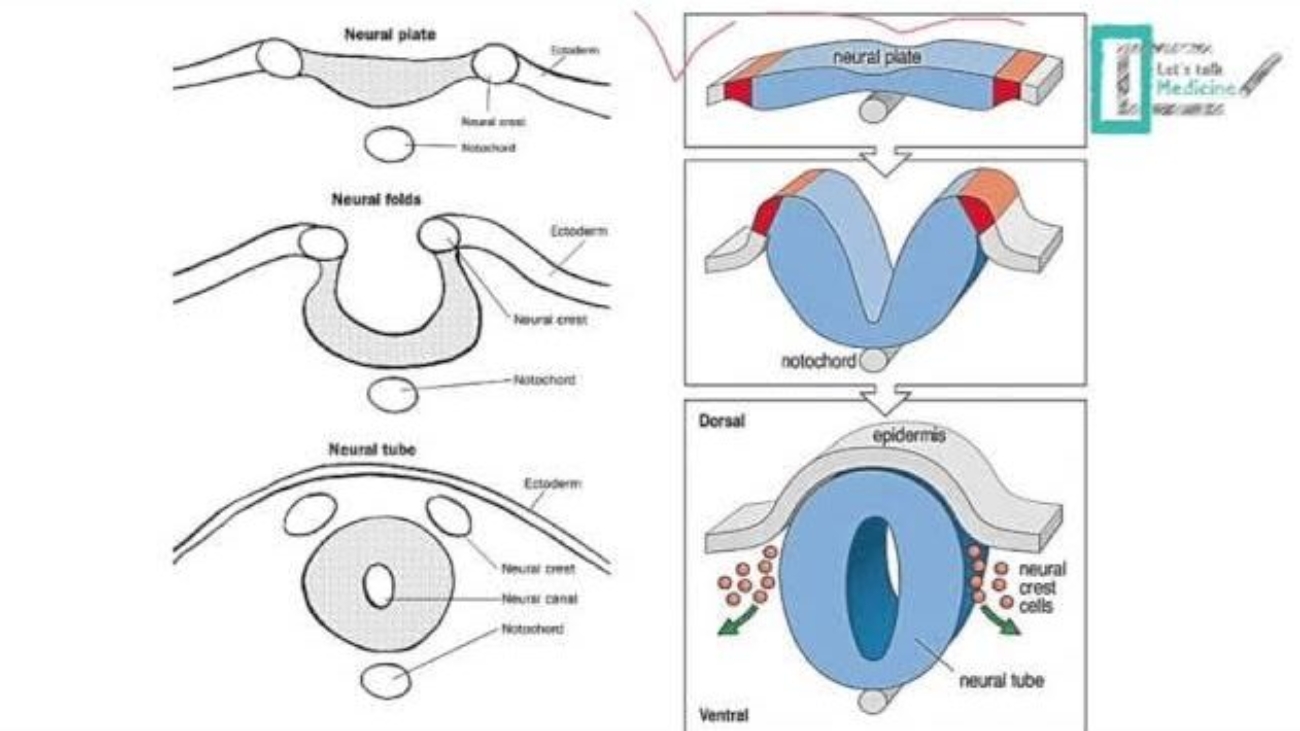
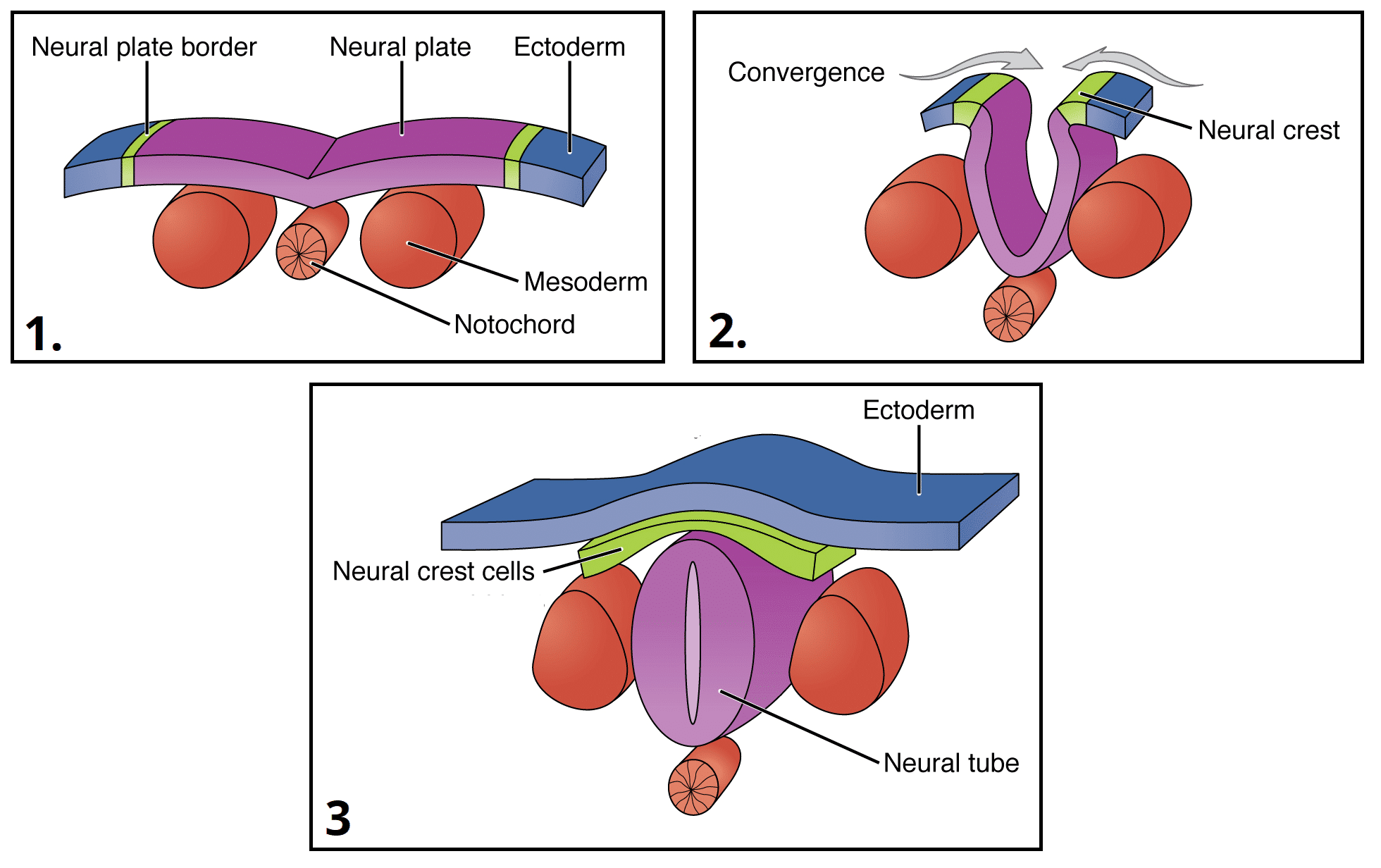
- Origin: the CNS appears as a slipper-shaped plate of ectoderm called the neural plate.
- Induction: This process is induced by the underlying notochord (a transient rod-like structure formed from mesoderm) and paraxial mesoderm. The notochord secretes signaling molecules (e.g., Sonic Hedgehog, SHH) that induce the overlying ectoderm to thicken and differentiate into the neural plate.
- Location: It forms in the mid-dorsal region, anterior to the primitive node, running cranially from the Hensen's node (primitive node).
2. Neural Fold and Neural Tube Formation (Week 3-4)
- Neural Folds: The lateral edges of the neural plate elevate to form neural folds, with a depressed neural groove forming in the midline.
- Fusion: The neural folds eventually meet in the midline and fuse. This fusion typically begins in the cervical region (around the 4th somite level) and proceeds bidirectionally:
- Cranially: Towards the head.
- Caudally: Towards the tail.
- Neural Tube: The fusion of the neural folds transforms the neural plate into the neural tube. This hollow tube will ultimately give rise to the entire CNS (brain and spinal cord).
- Neural Crest Cells: As the neural folds fuse and the neural tube closes, a population of cells at the crests of the neural folds detaches. These are the neural crest cells, a remarkably pluripotent group of cells that migrate extensively throughout the embryo and give rise to a vast array of structures, including:
- Parts of the PNS (sensory ganglia, autonomic ganglia).
- Melanocytes (pigment cells).
- Adrenal medulla.
- Craniofacial bones and cartilage.
- Schwann cells.
3. Neuropore Closure (Week 4)
- Communication with Amniotic Cavity: Once fusion is initiated, the open ends of the neurotube form the cranial (anterior) neuropore and the caudal (posterior) neuropore. These neuropores temporarily communicate with the amniotic cavity, allowing for exchange of fluid.
- Closure Timing: This is a critical stage.
- Closure of the Cranial Neuropore: Occurs at approximately the 18-20 somite stage (around day 25). This closure is essential for normal brain development.
- Closure of the Caudal Neuropore: Occurs approximately 2 days later (around day 27). This closure is essential for normal spinal cord development.
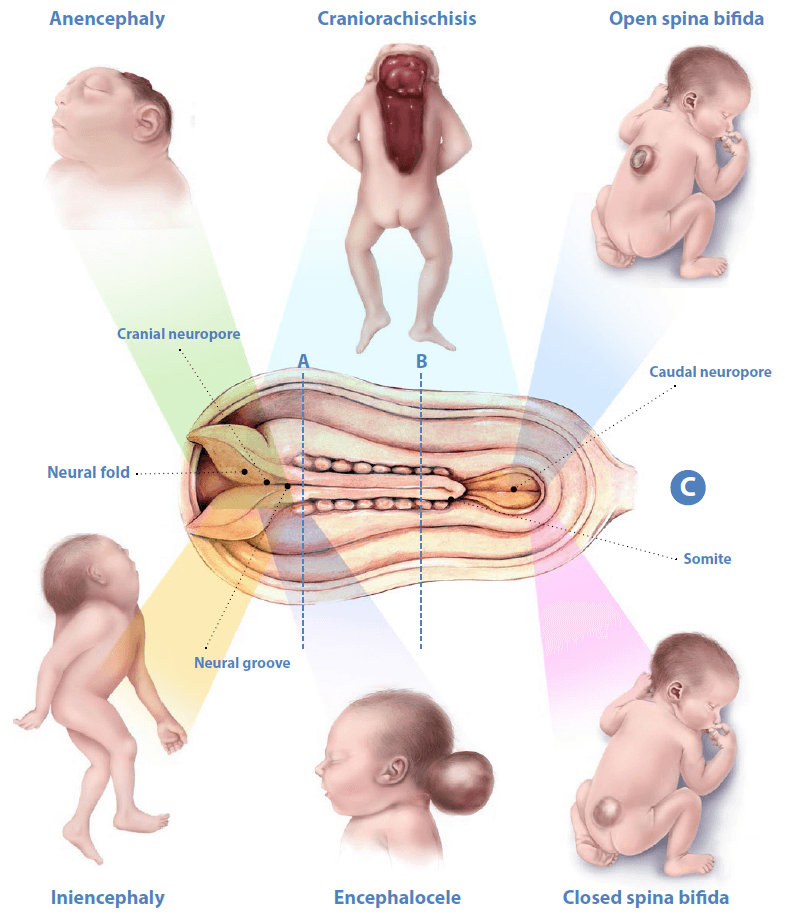
- Anencephaly: Failure of the cranial neuropore to close, leading to absence of a major portion of the brain, skull, and scalp. Incompatible with life.
- Spina Bifida: Failure of the caudal neuropore to close, resulting in incomplete closure of the vertebral column and exposure of the spinal cord. Severity varies (spina bifida occulta, meningocele, myelomeningocele).
- Folic Acid: Supplementation with folic acid (a B vitamin) before and during early pregnancy significantly reduces the incidence of NTDs.
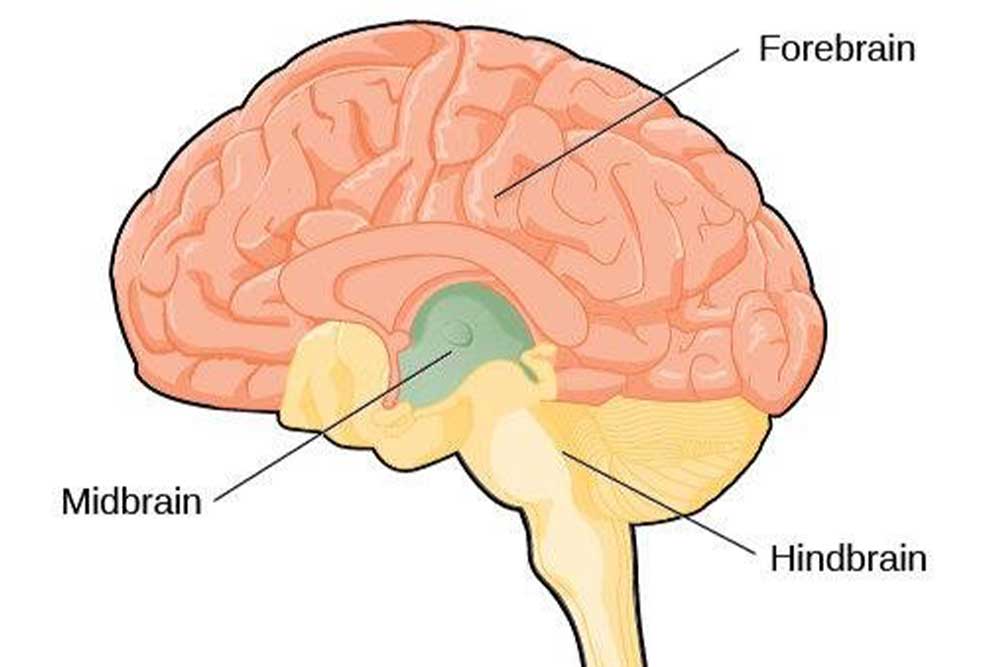
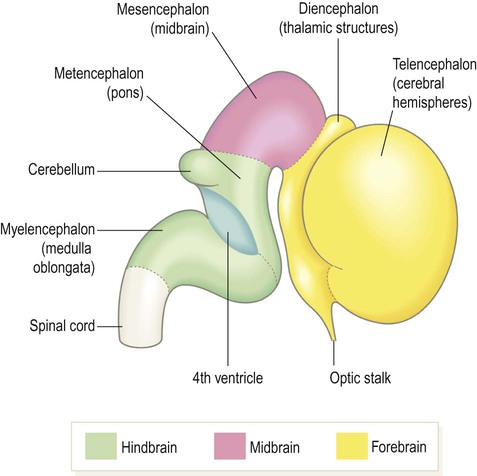
4. Primary Brain Vesicles (Late Week 4)
Once the cranial neuropore closes, the cephalic (cranial) end of the neural tube undergoes rapid growth and forms three distinct dilations, the primary brain vesicles:
- Prosencephalon (Forebrain): The most rostral vesicle.
- Mesencephalon (Midbrain): The middle vesicle, a relatively short segment.
- Rhombencephalon (Hindbrain): The most caudal vesicle, continuous with the future spinal cord.
5. Secondary Brain Vesicles (Week 5)
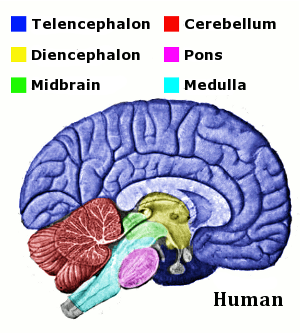
By the fifth week, the primary vesicles further subdivide, resulting in five secondary brain vesicles:
- Prosencephalon (Forebrain) divides into:
- Telencephalon: The most rostral part. It consists of a midline portion and two large lateral outgrowths that will become the primitive cerebral hemispheres.
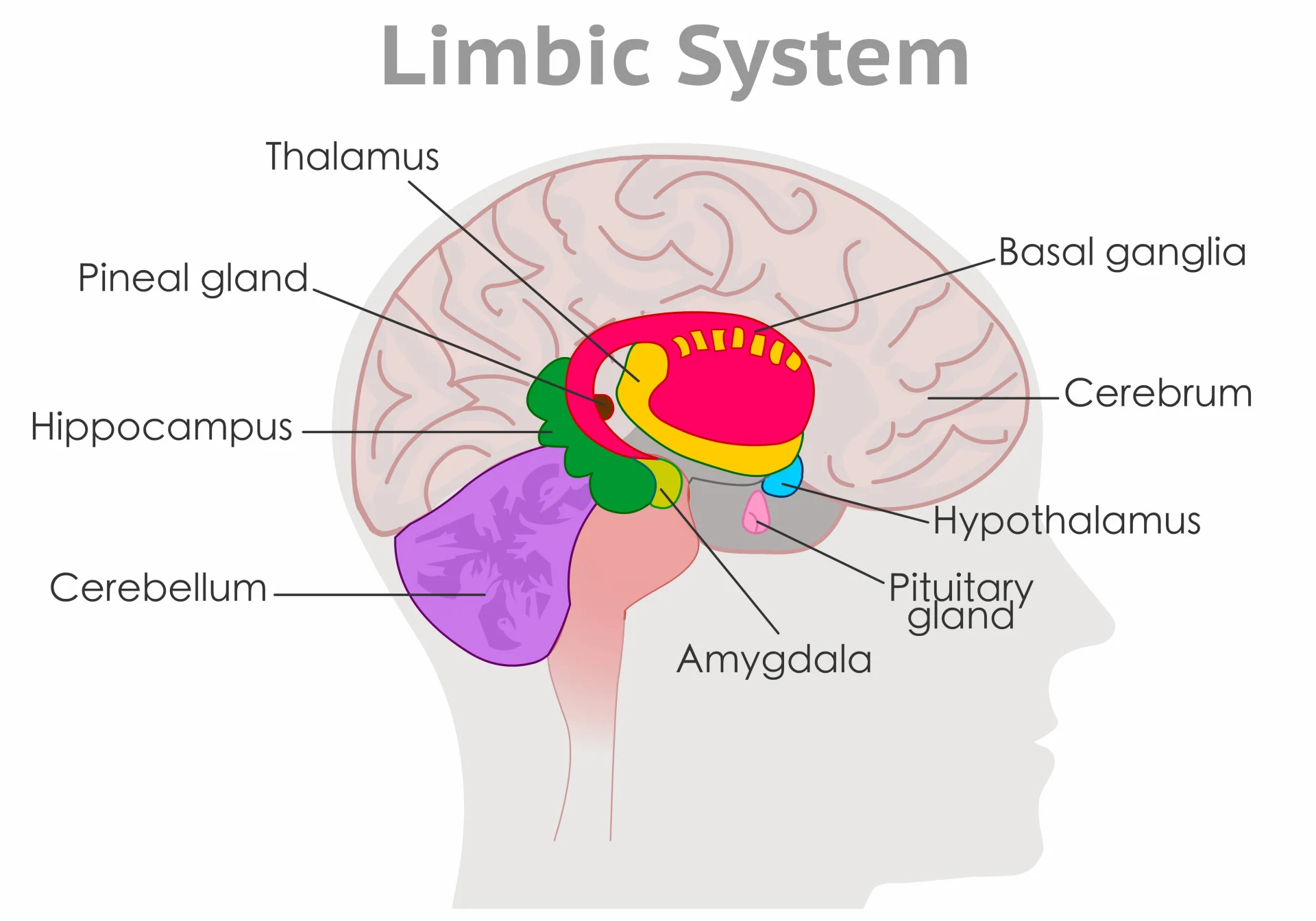
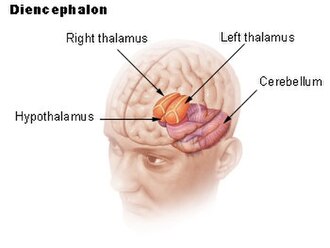
- Diencephalon: Forms the central core of the forebrain, with outgrowths that include the optic vesicles (which will form the retina and optic nerve).
- Mesencephalon (Midbrain) remains undivided.
- Rhombencephalon (Hindbrain) divides into:
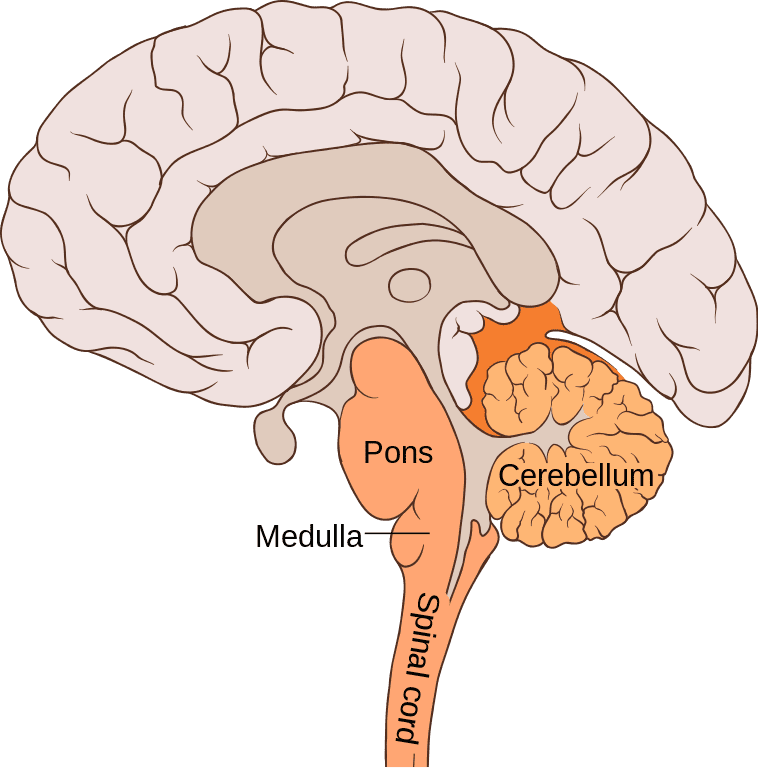
- Metencephalon: Will develop into the pons and cerebellum.
- Myelencephalon: Will develop into the medulla oblongata.
| Primary Vesicle | Secondary Vesicles | Adult Brain Structure |
|---|---|---|
| Prosencephalon | Telencephalon | Cerebral Hemispheres (cortex, white matter, basal ganglia) |
| Diencephalon | Thalamus, Hypothalamus, Epithalamus | |
| Mesencephalon | Mesencephalon | Midbrain |
| Rhombencephalon | Metencephalon | Pons, Cerebellum |
| Myelencephalon | Medulla Oblongata | |
| Caudal Neural Tube | Spinal Cord |
6. Brain Flexures
During this period of rapid growth and subdivision, the developing brain bends at specific points, forming flexures:
- Cephalic (Midbrain) Flexure: Occurs in the midbrain region, bending the forebrain ventrally.
- Cervical Flexure: Occurs at the junction of the rhombencephalon and spinal cord.
- Pontine Flexure: Occurs in the metencephalon, creating the characteristic shape of the pons and cerebellum.
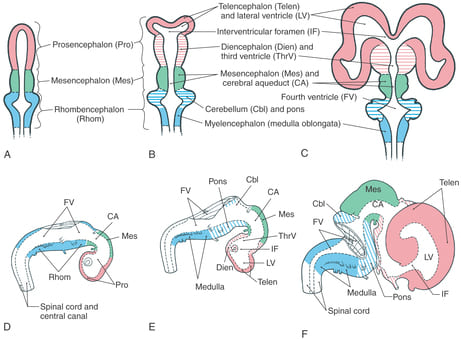
7. Development of the Ventricular System
- Lumen Continuity: You've correctly highlighted a critical point: The lumen (central canal) of the spinal cord is continuous with the cavities within the brain vesicles. This continuous lumen ultimately forms the entire ventricular system of the adult brain, which is filled with cerebrospinal fluid (CSF).
- Specific Luminal Derivatives:
- Lumen of the Telencephalon forms the Lateral Ventricles (one in each cerebral hemisphere).
- Lumen of the Diencephalon forms the Third Ventricle.
- Lumen of the Mesencephalon narrows to form the Cerebral Aqueduct (of Sylvius).
- Lumen of the Metencephalon and Myelencephalon combine to form the Fourth Ventricle.
- Lumen of the caudal neural tube remains as the Central Canal of the Spinal Cord.
- Connections:
- The Lateral Ventricles communicate with the Third Ventricle through the Interventricular Foramina of Monro.
- The Third Ventricle communicates with the Fourth Ventricle via the Cerebral Aqueduct.
- The Fourth Ventricle communicates with the subarachnoid space (surrounding the brain and spinal cord) via the Foramina of Luschka (lateral apertures) and the Foramen of Magendie (median aperture), and also with the central canal of the spinal cord.
Congenital Anomalies of the CNS
1. Spina Bifida
A neural tube defect (NTD) resulting from the incomplete closure of the neural tube and/or the vertebrae in the spinal column. The severity varies greatly.
- Types:
- Spina Bifida Occulta: The mildest form, a small gap in the vertebrae, usually no neurological deficits, often asymptomatic. A tuft of hair or a dimple on the lower back might be the only sign.
- Meningocele: The meninges (membranes surrounding the spinal cord) protrude through the vertebral opening, forming a fluid-filled sac. The spinal cord remains within the vertebral canal. May cause minor neurological problems.
- Myelomeningocele (Meningiomyelocoele): The most severe form, where the spinal cord and nerves protrude through the opening, forming a sac. This leads to significant neurological deficits below the level of the lesion, including paralysis, bladder/bowel dysfunction, hydrocephalus, and learning difficulties.
- Cause: Failure of the caudal neuropore to close completely during early embryonic development.
- Prevention: Folic acid supplementation before and during early pregnancy significantly reduces the risk.
2. Hydrocephalus
An abnormal accumulation of cerebrospinal fluid (CSF) within the brain's ventricles or subarachnoid space, leading to increased intracranial pressure and often enlargement of the head (especially in infants before skull sutures close).
- Causes:
- Obstruction: Blockage of CSF flow (e.g., aqueductal stenosis, tumors, adhesions). This is non-communicating (obstructive) hydrocephalus.
- Impaired Absorption: Problems with CSF reabsorption into the bloodstream (e.g., arachnoid granulations dysfunction, post-hemorrhage, post-infection). This is communicating hydrocephalus.
- Overproduction: Rare, e.g., choroid plexus papilloma.
- Symptoms (in infants): Rapid increase in head circumference, bulging fontanelle, "sunsetting" eyes, vomiting, irritability, seizures.
- Treatment: Surgical placement of a shunt (e.g., ventriculoperitoneal shunt) to divert excess CSF to another body cavity where it can be absorbed.
3. Microcephaly
An abnormally small head circumference for the child's age and sex, typically defined as more than two standard deviations below the mean.
- Diagnosis: As you stated, based on biometry (occipito-frontal diameter (OFD) and biparietal diameter (BPD) are reduced), often detected prenatally or at birth.
- Causes: A wide range, indicating that the brain either didn't develop properly or stopped growing. Examples include:
- Genetic abnormalities: Chromosomal disorders, single gene mutations.
- Prenatal infections: Zika virus, toxoplasmosis, cytomegalovirus, rubella.
- Exposure to toxins: Alcohol (Fetal Alcohol Syndrome), certain drugs.
- Severe malnutrition.
- Perinatal complications: Brain injury, lack of oxygen.
- Complications: Mental retardation/Intellectual disability, Associated anomalies (seizures, cerebral palsy). Prognosis varies.
4. Macrocephaly
An abnormally large head circumference, typically defined as more than two standard deviations above the mean.
- Causes:
- Benign Familial Macrocephaly: Often a harmless genetic trait.
- Hydrocephalus: Can cause macrocephaly, especially if it develops before the skull sutures fuse.
- Brain Tumors: Large tumors can increase head size.
- Subdural Hematomas: Accumulation of blood under the dura mater.
- Genetic Syndromes: Such as Sotos syndrome, Fragile X syndrome.
- Megalencephaly: An abnormally large brain.
5. Anencephaly
A severe neural tube defect characterized by the absence of a major portion of the brain, skull, and scalp. The cerebral hemispheres are absent or reduced to small masses.
- Cause: Failure of the cranial neuropore to close completely during early embryonic development (around day 25).
- Prognosis: Always fatal, usually within hours or days after birth.
Cerebral Hemispheres
- Growth and Shape: You correctly note their "C-shape" growth (especially relevant during embryological development as they grow back over the diencephalon and brainstem).
- Longitudinal Fissure: Divides the brain into two halves.
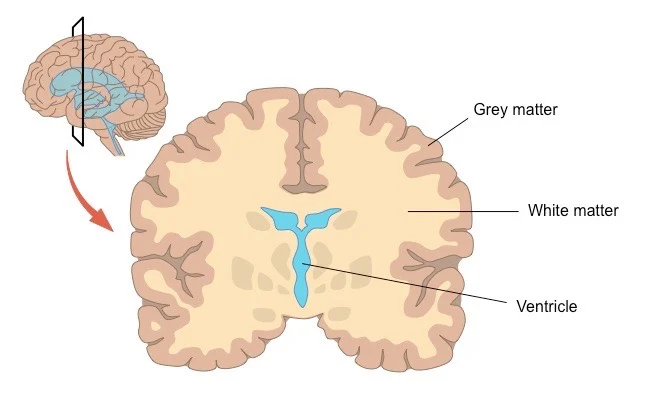
- Cerebral Cortex (Grey Matter):
- The outer layer of each hemisphere, composed primarily of neuron cell bodies, dendrites, unmyelinated axons, and glial cells. This is where most of the higher-level processing occurs.
- Its convoluted surface (gyri and sulci) significantly increases the surface area for this grey matter, allowing for a much larger number of neurons.
- Contralateral Control: "The left hemisphere controls the right half of the body, and vice-versa, because of a crossing of the nerve fibers in the medulla." This is known as decussation. The primary motor pathways (corticospinal tracts) cross over (decussate) in the pyramids of the medulla. Similarly, most sensory pathways also decussate.
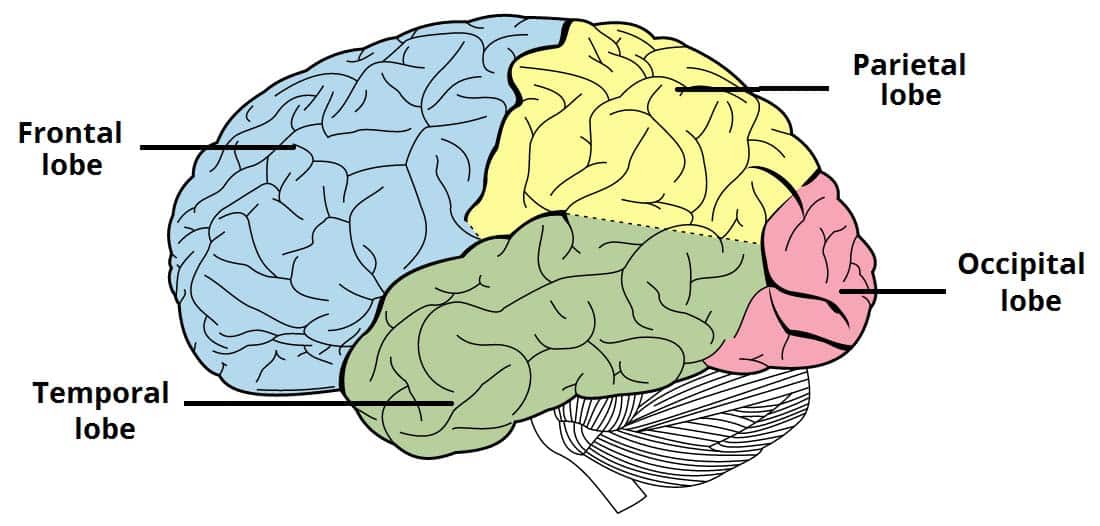
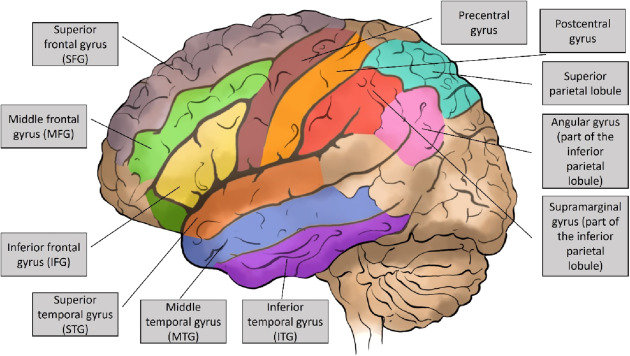
- Functional Divisions (Lobes): "The central sulcus and the lateral sulcus, divide each cerebral hemisphere into four sections, called lobes." This is a key anatomical landmarking.
- Central Sulcus (Fissure of Rolando): Divides the frontal lobe from the parietal lobe. It's especially important because it separates the primary motor cortex (anterior to it, in the precentral gyrus) from the primary somatosensory cortex (posterior to it, in the postcentral gyrus).
- Lateral Sulcus (Sylvian Fissure): Separates the frontal and parietal lobes from the temporal lobe below.
- Parieto-occipital Sulcus: Not as deep as the central or lateral, but helps demarcate the parietal lobe from the occipital lobe.
- Somatotopic Organization: "Starting from the top of the hemisphere, the upper regions of the motor and sensory areas control the lower parts of the body." This refers to the homunculus (little man) representation.
- In both the primary motor and somatosensory cortices, different body parts are mapped to specific regions of the gyrus in an inverted fashion. For example, the feet and legs are represented at the top of the gyrus (medial surface), and the head and face are represented near the lateral sulcus.

Cerebral Dominance (Lateralization)
The tendency for one cerebral hemisphere to be more involved in certain functions than the other. It's not that one hemisphere is "dominant" over the other for all functions, but rather that specific functions are lateralized.
Language and Manual Skills:
Left Hemisphere
- Language: For the vast majority of people (around 90-95% of right-handers and 70% of left-handers), the left hemisphere is dominant for language functions (speech production and comprehension).
- Broca's Area: Located in the frontal lobe, typically in the left hemisphere. Essential for speech production. Damage leads to Broca's aphasia (non-fluent aphasia), where speech is slow, effortful, and grammatically incorrect, but comprehension is relatively preserved.
- Wernicke's Area: Located in the temporal lobe, typically in the left hemisphere. Essential for language comprehension. Damage leads to Wernicke's aphasia (fluent aphasia), where speech is fluent but often meaningless ("word salad"), and comprehension is severely impaired.
- Characteristics: Logical, Analytical, Sequential Processing.
Right Hemisphere
- Non-Verbal Skills: Tends to be dominant for spatial perception, facial recognition, visual-spatial processing, musical ability, and emotional perception (interpreting tone of voice, facial expressions).
- Characteristics: Holistic, Intuitive, Parallel Processing.
- Appreciation of sound from left ear: More accurately, sounds from both ears project to both hemispheres, but there's a slight contralateral dominance or specialized processing for certain auditory aspects.
- Sensation of left body / Perception of left visual field: This refers to the contralateral representation.
Handedness and Language Dominance:
- Right-handed people: ~95% have left-hemisphere dominance for language.
- Left-handed people: This group is more diverse.
- ~70% have left-hemisphere dominance for language (like right-handers).
- ~15% have right-hemisphere dominance for language.
- ~15% have bilateral language representation.
Cortical Localization (Specific Gyri and Sulci)
These are key landmarks.
- AnGy - Angular Gyrus: Located in the parietal lobe, involved in language, number processing, spatial cognition, memory retrieval.
- Csul - Central Sulcus: Already discussed, divides frontal and parietal.
- LonFis - Longitudinal Fissure: Already discussed, separates hemispheres.
- MFGy - Middle Frontal Gyrus: Part of the frontal lobe, involved in working memory, cognitive control.
- OGy - Occipital Gyri: Part of the occipital lobe, visual processing.
- PoCGy - Postcentral Gyrus: Located in the parietal lobe, posterior to the central sulcus; contains the primary somatosensory cortex.
- PoSul - Parieto-occipital Sulcus: Divides parietal and occipital lobes.
- PrCGy - Precentral Gyrus: Located in the frontal lobe, anterior to the central sulcus; contains the primary motor cortex.
- PrCSul - Precentral Sulcus: Anterior to the precentral gyrus.
- SFGy - Superior Frontal Gyrus: Part of the frontal lobe, involved in self-awareness, working memory.
- SFSul - Superior Frontal Sulcus: Separates superior and middle frontal gyri.
- SMGy - Supramarginal Gyrus: Located in the parietal lobe, involved in language, empathy.
- SPLob - Superior Parietal Lobule: Part of the parietal lobe, involved in spatial orientation and working memory.
Hemispheric Specialization
It's crucial to remember that while certain functions are lateralized (predominantly handled by one hemisphere), the brain always works as an integrated whole, with constant communication between the two hemispheres via the corpus callosum. The concept of "left-brain" vs. "right-brain" personalities is an oversimplification; rather, it describes tendencies for processing styles.
Right Hemisphere Functions
The right hemisphere is often described as more involved in "non-linear" or "holistic" processing.
- Emotional Functions:
- Emotional Prosody: The ability to understand and express the emotional tone of voice. Damage can lead to aprosodia.
- Primary Emotionality: Processing and experiencing raw emotions.
- Empathy and Comprehension of Emotionality: Understanding and sharing the feelings of others. Interpreting facial expressions, body language.
- Affective Behavior: Influence on mood and emotional regulation. Right hemisphere damage can sometimes lead to an indifferent or euphoric affect.
- Wit and Humor: Understanding jokes, irony, and satire.
- Attentional Functions:
- Arousal and Vigilance: Maintaining a general state of alertness.
- Attentiveness (Spatial Attention): Crucially, the right parietal lobe is dominant for directing attention to both the right and left sides of space. Damage to the right parietal lobe can lead to spatial neglect (hemispatial neglect), where the individual ignores the left side of their body and environment.
- Cognitive Functions:
- Spatial Orientation & Relations: Navigating in space, understanding maps, judging distances, mental rotation of objects.
- Sequencing of symbols, objects, and events: Involved in non-verbal or visual sequencing.
- Timing and Time Perception: Contributes to the perception of duration and rhythm.
- Music Appreciation: Processing melodies, harmonies, and overall musical structure.
- Recognition of Objects and Faces: Recognizing familiar faces (prosopagnosia can result from damage, often to the fusiform gyrus).
- Geometric Communication: Understanding visual designs and spatial relationships.
- Non-verbal Communication: Interpreting gestures, facial expressions, body language.
- Praxias: Coordinated motor behaviors, particularly those involving spatial reasoning or complex sequences.
- Primary Visual Imagery & Symbolization: Picture-to-picture storage/representation; Symbolization; Picture-to-word storage/representation.
- Frontal Lobe Contributions (Right Side Specific): Fundamental Movement of Left Body; Left Voluntary Gaze; Motor Persistence; Order (Formal Type); Planning, Volition, Diligence, Executive Control, Social Conduct.
Summary of Right Brain
- Random
- Intuitive
- Holistic
- Synthesizing
- Subjective
- Looks at wholes
Summary of Left Brain
- Logical
- Analytical
- Sequential
- Linear
- Objective
- Focuses on details
Left Hemisphere Functions
- Language Representation:
- Dominance in ~97% (right-handers) and ~70% (left-handers).
- Brain Plasticity: Neuroplasticity allows the brain, especially in childhood, to reassign functions to spared areas. The earlier the injury, the better the chances for the undamaged hemisphere to compensate for language functions. This capacity diminishes with age.
- Cognitive Functions: Uses logic, Detail oriented, Facts rule, Words and language, Present and past, Math and science, Can comprehend (linguistic comprehension).
General Frontal Lobe Functions
These functions apply to both hemispheres but can have lateralized biases.
- Higher Functions: Abstract thought, personality, emotion (especially social and executive aspects).
- Motor Function: Primary motor cortex.
- Problem Solving: Executive function.
- Spontaneity and Initiative: Initiating actions and thoughts.
- Memory: Working memory, prospective memory.
- Language: Especially the left frontal lobe (Broca's area).
- Judgment and Impulse Control: Regulating behavior.
- Social and Sexual Behavior: Modulating appropriate responses.
- Vulnerability to Injury: The anterior location makes them highly susceptible to trauma.
Clinical Anatomy and Considerations
- Dementia: "Diffuse hemispheric disease - a progressive and hopeless condition." Characterized by a decline in cognitive function (memory, language, problem-solving) severe enough to interfere with daily life. Causes: Alzheimer's, vascular dementia, Lewy body dementia, etc.
- Bilateral Representation of Hearing and Smell:
- Hearing: Auditory pathways are largely bilateral. Unilateral brain injury typically does not cause complete deafness in either ear.
- Smell (Olfaction): Olfactory tracts project directly to the primary olfactory cortex and amygdala/hippocampus. Projections are largely ipsilateral initially, but subsequent processing involves both hemispheres. Unilateral damage rarely causes total anosmia.
- Treatment Modalities (Neurosurgery):
- Hemispherectomy: Surgical removal or disconnection of an entire cerebral hemisphere.
- Indication: Severe, intractable epilepsy (e.g., Rasmussen's encephalitis) in very young children.
- Goal: To stop debilitating seizures.
- Temporal Lobectomy: Surgical removal of a portion of the temporal lobe.
- Indication: Drug-resistant temporal lobe epilepsy.
- Goal: To remove the seizure focus.
- Hemispherectomy: Surgical removal or disconnection of an entire cerebral hemisphere.
- Traumatic Brain Injury (TBI):
- Cerebral Contusion (Bruising): Bruising of the brain tissue.
- Pia Stripped: Often implies that the pia mater is damaged or detached from the underlying brain tissue.
- Cerebral Lacerations: Tearing of the brain tissue. Causes: Severe injuries like gunshot wounds or depressed cranial fractures.
- Cerebral Contusion (Bruising): Bruising of the brain tissue.
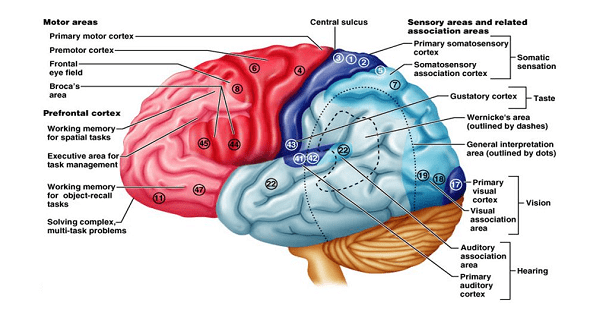
Functional Localization of Cerebral Cortex
A. Sensory Areas
These areas receive and interpret sensory information from the body and external environment.
- Primary Sensory Area (Primary Somatosensory Cortex - S1):
- Location: Primarily located in the postcentral gyrus of the parietal lobe (Brodmann Areas 3, 1, 2).
- Function: Receives direct input from the thalamus (ventral posterior nucleus) carrying general somatic sensations. It's where the initial conscious perception of these sensations occurs.
- Somatotopic Organization: Exhibited by the Sensory Homunculus.
- Secondary Sensory Areas:
- Location: Surround the primary sensory areas (e.g., area S2).
- Function: Involved in more complex processing of sensory information, integration of different sensory modalities, and possibly sensory memory.
B. Motor Areas
These areas are involved in planning, initiating, and executing voluntary movements.
- Primary Motor Area (Primary Motor Cortex - M1):
- Location: Located in the precentral gyrus of the frontal lobe (Brodmann Area 4).
- Function: Directly controls the execution of voluntary movements. It contains large pyramidal neurons (Betz cells).
- Somatotopic Organization: Exhibited by the Motor Homunculus.
- Secondary Motor Areas (Premotor Cortex):
- Location: Anterior to the primary motor cortex (Brodmann Area 6). Includes the premotor area proper and the supplementary motor area (SMA).
- Function:
- Premotor Area: Involved in planning and orienting movements, especially those guided by external sensory cues.
- Supplementary Motor Area (SMA): Involved in planning and organizing complex sequences of movements, especially internally generated movements or learned sequences. Crucial for bimanual coordination.
C. Speech Areas
- Broca's Area: Location: Inferior frontal gyrus (left hemisphere). Function: Speech production. Damage: Broca's aphasia.
- Wernicke's Area: Location: Posterior part of the superior temporal gyrus (left hemisphere). Function: Language comprehension. Damage: Wernicke's aphasia.
D. Association Areas
These areas integrate information from various sensory and motor areas and are responsible for higher-level cognitive functions like memory, reasoning, decision-making, and personality.
Homunculi (Little Men)
The concept of the homunculus illustrates the somatotopic organization of the primary motor and somatosensory cortices.
- Sensory Homunculus: Location: Postcentral gyrus. The size of the cortical area is proportional to the density of sensory receptors. Lips, face, and hands have large representations. Orientation: Inverted (feet at top, head lateral).
- Motor Homunculus: Location: Precentral gyrus. The size of the cortical area is proportional to the fineness and complexity of movements. Hands, fingers, and facial muscles have large representations. Orientation: Inverted.
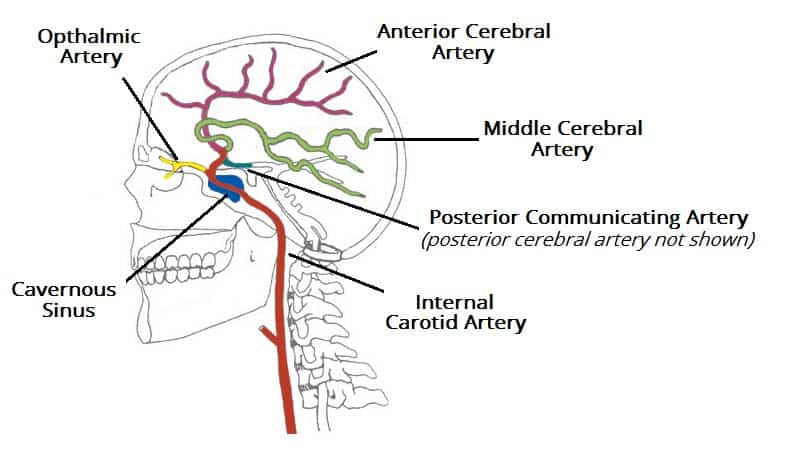
Blood Supply of the Brain (Cerebral Vasculature)
The brain receives a rich and redundant blood supply from two main arterial systems: the internal carotid arteries and the vertebral arteries.
A. Internal Carotid Artery System
- Ophthalmic Artery: Supplies the eye and surrounding structures.
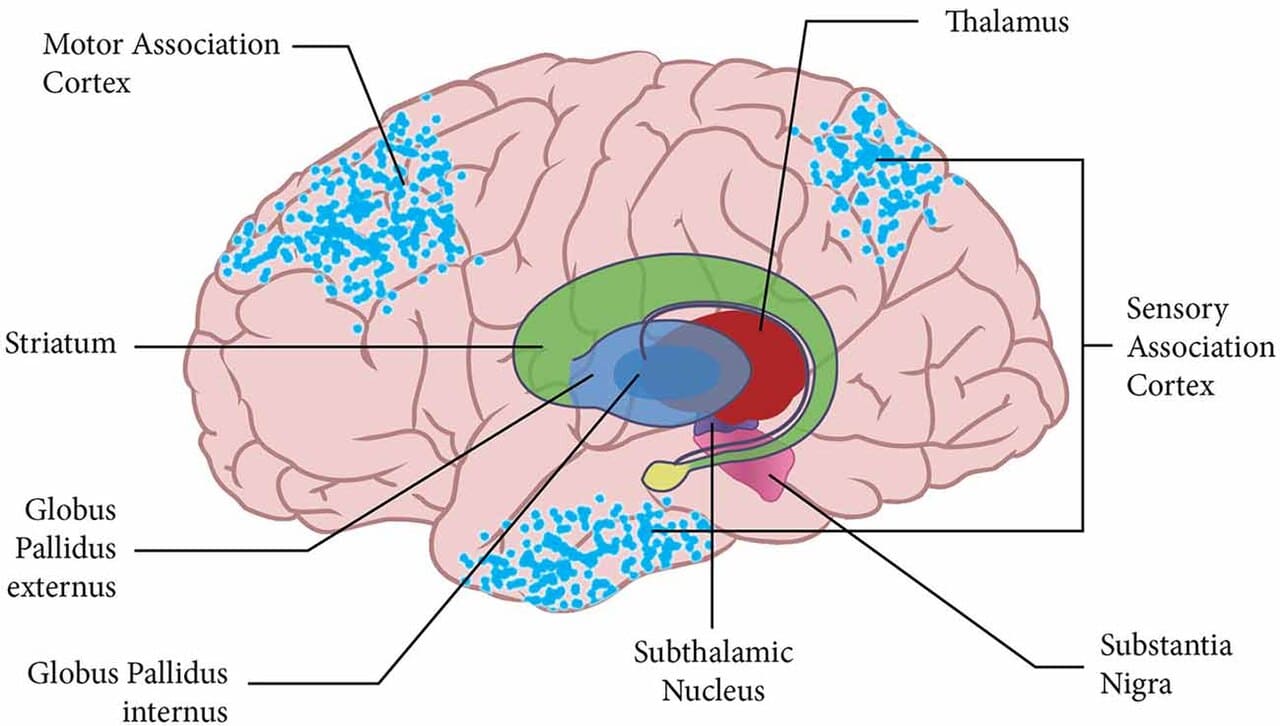
- Anterior Choroidal Artery: Supplies choroid plexus, hippocampus, basal ganglia, internal capsule.
- Middle Cerebral Artery (MCA):
- Distribution: Supplies the lateral surface of the cerebral hemispheres (frontal, parietal, temporal lobes). Includes primary motor/sensory cortices for upper limb/face, Broca's, Wernicke's.
- Clinical Significance: Most common artery in stroke. Leads to contralateral hemiparesis (face/arm > leg), sensory loss, aphasia (dominant hemisphere).
- Anterior Cerebral Artery (ACA):
- Distribution: Supplies medial surface of frontal/parietal lobes. Includes primary motor/sensory cortices for lower limb.
- Clinical Significance: Stroke leads to contralateral leg weakness and sensory loss.
- Anterior Communicating Artery: Connects the two ACAs.
B. Vertebrobasilar System
- Vertebral Artery Branches: Posterior Inferior Cerebellar Artery (PICA), Anterior Spinal Artery, Posterior Spinal Arteries.
- Basilar Artery Branches: Anterior Inferior Cerebellar Artery (AICA), Pontine Arteries, Superior Cerebellar Artery (SCA), Posterior Cerebral Artery (PCA).
- Posterior Cerebral Artery (PCA):
- Distribution: Supplies occipital lobe (primary visual cortex), inferior temporal lobe, thalamus, midbrain.
- Clinical Significance: Stroke can lead to contralateral homonymous hemianopia and memory deficits.
- Posterior Cerebral Artery (PCA):
- Posterior Communicating Artery: Connects PCA to internal carotid system.
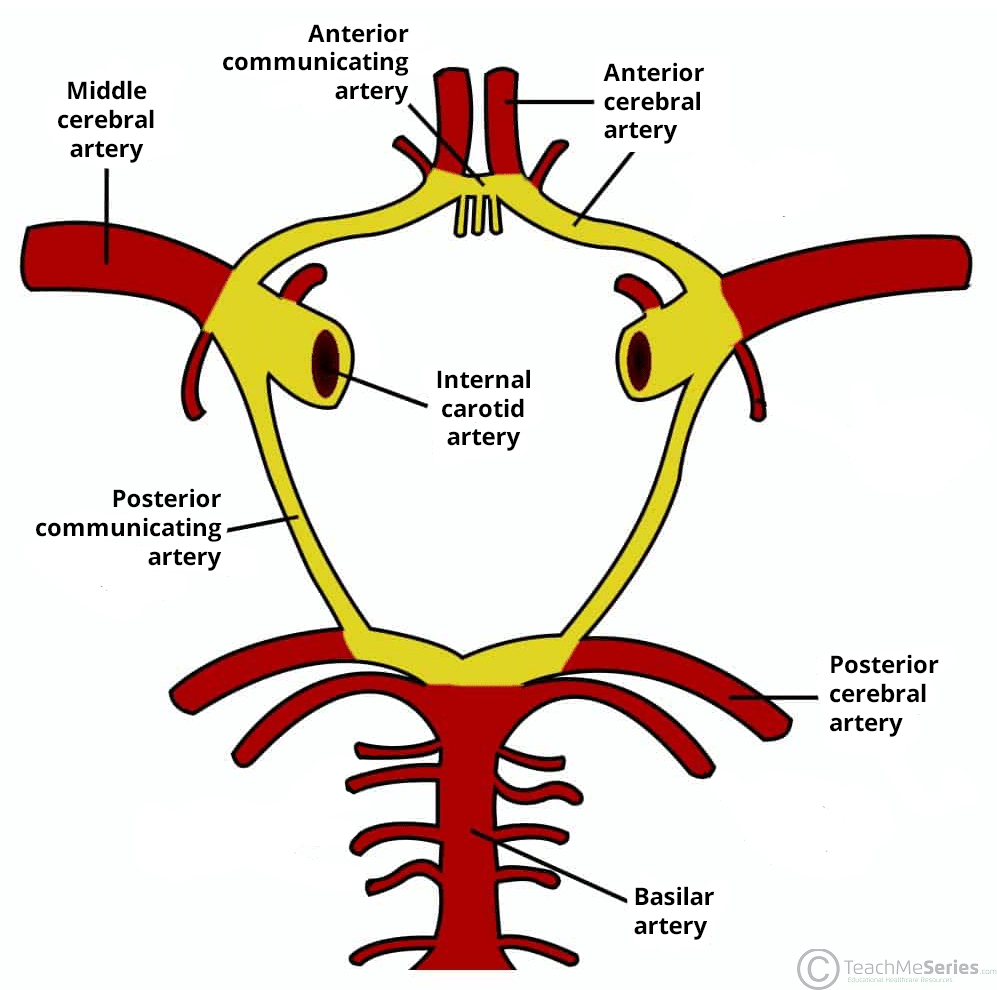
C. Circle of Willis
- Formation: Arterial anastomosis at the base of the brain (Anterior communicating, ACA, Internal Carotid, Posterior communicating, PCA).
- Function: Provides a critical collateral circulation.
D. Arteries of the Scalp and Face
- External Carotid Artery Branches: Superior Thyroid, Lingual, Facial, Maxillary, Superficial Temporal, Posterior Auricular, Occipital Arteries.
- Carotid Sinus: Baroreceptor located at the bifurcation of the common carotid artery.
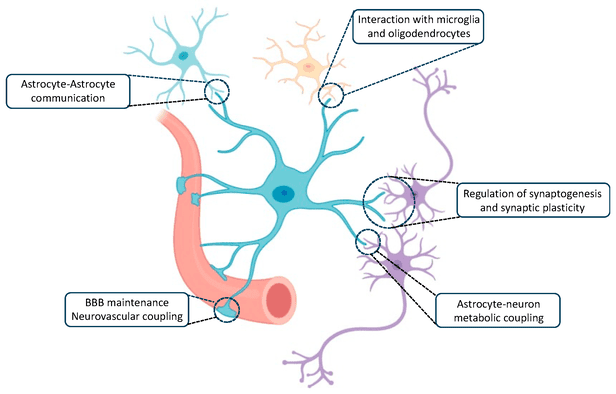
Astrocytes (A Type of Glial Cell)
Astrocytes are the most numerous glial cells in the CNS and play a critical, multifaceted role in brain function and health.
Create Supportive Framework
Provide physical support/scaffolding for neurons, occupy spaces, help define neuronal territories.
Create "Blood-Brain Barrier" (BBB)
Extend end feet encircling capillaries. Induce tight junctions between endothelial cells. Regulate passage of substances from blood to brain.
Monitor & Regulate Interstitial Fluid
Neurotransmitter Uptake (glutamate), Ion Homeostasis (K+), Metabolic Support (lactate, glycogen).
Secrete Chemicals
Neurotrophic factors/signaling molecules guiding neuronal migration and synaptogenesis.
Scar Tissue Formation (Gliosis)
Undergo reactive astrogliosis after injury. Form glial scar. Helps wall off injury but can inhibit axonal regeneration.
CNS Embryology Quiz
Systems Anatomy
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
CNS Embryology Quiz
Systems Anatomy
Preparing questions...
Exam Completed!
See your performance breakdown below.