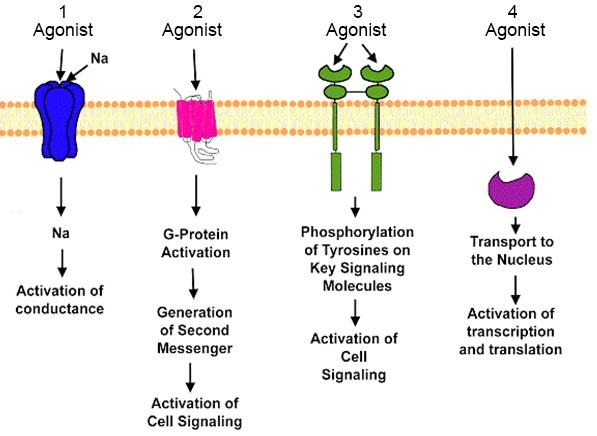
Adverse Drug & Reactions
Adverse Drug Reactions (ADRs)
Before we memorize definitions, understand the real-world impact of Adverse Drug Reactions. In the least developing countries, people die and are buried without anyone ever knowing whether they died from the actual disease they were fighting, or from the medication they used to treat it. Medications are powerful chemical tools. When used incorrectly, or when the body reacts unexpectedly, they can be lethal. Your goal as a medical professional is to foresee, identify, and manage these reactions.
1. Terms
In medical pharmacology, words have very strict meanings. You must be able to distinguish between an ADR, an ADE, and a Side Effect.
What is an Adverse Drug Reaction (ADR)?
An Adverse Drug Reaction is formally defined as any unintended or noxious (harmful) effect which meets the following strict criteria:
- It is suspected to be due to a drug.
- It occurs at doses normally used in man (this is crucial—if someone takes 50 pills on purpose, the resulting liver failure is an overdose, not a standard ADR according to older definitions, though the FDA includes it now).
- It is severe enough that it may require treatment, a decrease in the dose, or total withdrawal of the drug.
- It dictates caution in the future use of the same drug for that patient.
Adverse Drug Event (ADE) vs. ADR
An Adverse Drug Event (ADE) is a broader umbrella term. It is any untoward (unlucky/bad) occurrence that may present during medical treatment.
The Difference: An ADE does not necessarily have a causal relationship with the treatment.
You give a patient a blood pressure pill. An hour later, they trip on a rug, fall, and break their arm. The broken arm happened during medical treatment (making it an Adverse Drug Event), but the pill didn't cause the rug to be there. However, if the blood pressure pill caused severe dizziness, causing them to fall, that is an Adverse Drug Reaction (ADR) because there is a causal link.
What is a Side Effect?
A Side Effect is an extended pharmacological action of a drug. It is entirely predictable based on how the drug works in the body.
Atropine is given as an anticholinergic drug to dry up secretions in the lungs before surgery or to treat a slow heart rate. Because we know it blocks acetylcholine (the "rest and digest" chemical), we completely expect it to cause dryness of the mouth. The dry mouth is a side effect—a direct extension of its normal pharmacological action.
2. Regulatory Perspectives: WHO vs. FDA
Different health organizations define ADRs slightly differently, which affects how statistics are reported globally.
The WHO Definition
The World Health Organization (WHO) describes an ADR as: "The noxious and unintended drug effect which occurs at doses employed in man for prophylaxis (prevention), diagnosis, or therapy."
- The Limitation: This definition only encompasses part of the problem. It strictly says "at doses normally employed." The use of this highly restrictive definition hinders the reporting of ADRs because it ignores human error, addiction, and accidental poisonings.
The FDA Definition
The US Food and Drug Administration (FDA) uses a much broader, more realistic definition. The FDA defines an ADR as: "An undesirable effect, reasonably associated with the use of the drug, that may occur as a part of the pharmacological action of a drug OR may be unpredictable in its occurrence."
- Reporting Purposes: To capture the full scope of drug harm, the FDA strictly includes incidents of overdose (whether accidental, suicidal, or criminal) and incidents due to drug dependence or withdrawal after the cessation of drug administration.
3. Grading the Severity of Adverse Drug Reactions
When an ADR happens, it is categorized into one of four grades based on how aggressively the medical team must respond:
- Minor: No therapy, no specific antidote, and no prolongation of hospitalization is required. (Example: A mild, temporary headache after taking a medication that resolves on its own).
- Moderate: Requires a change in drug therapy, specific medical treatment, or prolongs the patient's hospital stay. (Example: A drug causes a severe rash that requires prescribing antihistamines and keeping the patient overnight for observation).
- Severe: Potentially life-threatening, causes permanent damage, or requires intensive medical treatment (ICU). (Example: A drug causes severe anaphylactic shock restricting breathing, requiring intubation).
- Lethal: The drug directly or indirectly contributes to the death of the patient.
4. Classification Systems: Rawlins and Thompson
There are several ways to classify ADRs. The simplest, most foundational method was proposed by Rawlins and Thompson. They divided all drug reactions into two major classes: Type A and Type B (also known as Type 1 and Type 2).
| Feature | Type A (Type 1) - Augmented | Type B (Type 2) - Bizarre |
|---|---|---|
| Synonyms | Predictable, toxic, quantitative, dose-related. | Unpredictable, allergic, idiosyncratic, qualitative, dose-independent. |
| Mechanism | Predictable and clearly understood based on the drug's normal mechanism of action. | Usually poorly understood. It has nothing to do with the drug's intended action. |
| Site of Action | 1. Same site of primary drug action. 2. Another site for primary and secondary actions. |
Unrelated to the normal site of action. |
| Incidence (Frequency) | High (70% to 75% of all ADRs). Very common. | Low (around 30%). Comparatively rare. |
| Morbidity (Sickness) | Generally High (many people feel mild to moderate sickness). | Severe illness is common when it strikes. |
| Mortality (Death rate) | Low. Rarely kills the patient. | High. Often causes serious illness and death. |
| Reproducibility | Reproducible (If you give the high dose again, it will happen again). | Not reproducible reliably in laboratory settings. Often not observed during conventional pharmacological and toxicological screening programs. |
| Treatment strategy | Adjust (Decrease) the dose. | Stop treatment immediately. Withdraw the drug completely. |
Deep Dive: Causes of Type A (Type 1) Reactions
Type A reactions happen because there is simply too much active drug in the body, or the body is too sensitive to it. The causes are broken down into three areas:
- Pharmaceutical Causes: Increased availability at the site of absorption. (e.g., A manufacturing error makes a pill release its contents too quickly).
- Pharmacokinetic (PK) Causes: Increased level at the site of action due to abnormalities in A, D, M, E (Absorption, Distribution, Metabolism, Excretion). Scenario: A patient with kidney failure cannot excrete a drug, so it builds up to toxic levels in the blood.
- Pharmacodynamic (PD) Causes:
- Enhanced organ or tissue responsiveness due to an enhanced number or sensitivity of receptors.
- Homeostatic imbalance.
- A concurrent disease state altering normal body function.
Examples of Type A: Bradycardia (slow heart rate) with β-adrenoceptor blockers (beta-blockers are meant to slow the heart; too much causes severe slowing). Hemorrhage with anticoagulants (blood thinners meant to stop clots; too much causes bleeding). Hypoglycemia with sulphonylureas (diabetes drugs meant to lower sugar; too much drops it dangerously low).
Deep Dive: Causes of Type B (Type 2) Reactions
Type B reactions are the dangerous, unpredictable "wild cards" of pharmacology. They account for many sudden drug withdrawals from the public market.
- Pharmaceutical Causes: Decomposition of the active constituent. (Example: Outdated, expired tetracycline breaks down into a toxic compound that causes a dangerous kidney condition called Fanconi-like syndrome). Effects of additives: Solubilizers, stabilizers, colorizers, and excipients can induce anaphylactoid reactions. (Example: Cremophor EL, a surfactant added to enhance the solubility of IV diazepam, has induced severe reactions in some patients).
- Pharmacokinetic Causes: The body liberates or creates an abnormal, highly toxic metabolite during the breakdown process.
- Pharmacodynamic Causes:
- Genetic (Idiosyncratic quirks in a person's DNA).
- Immunologic (True allergic reactions).
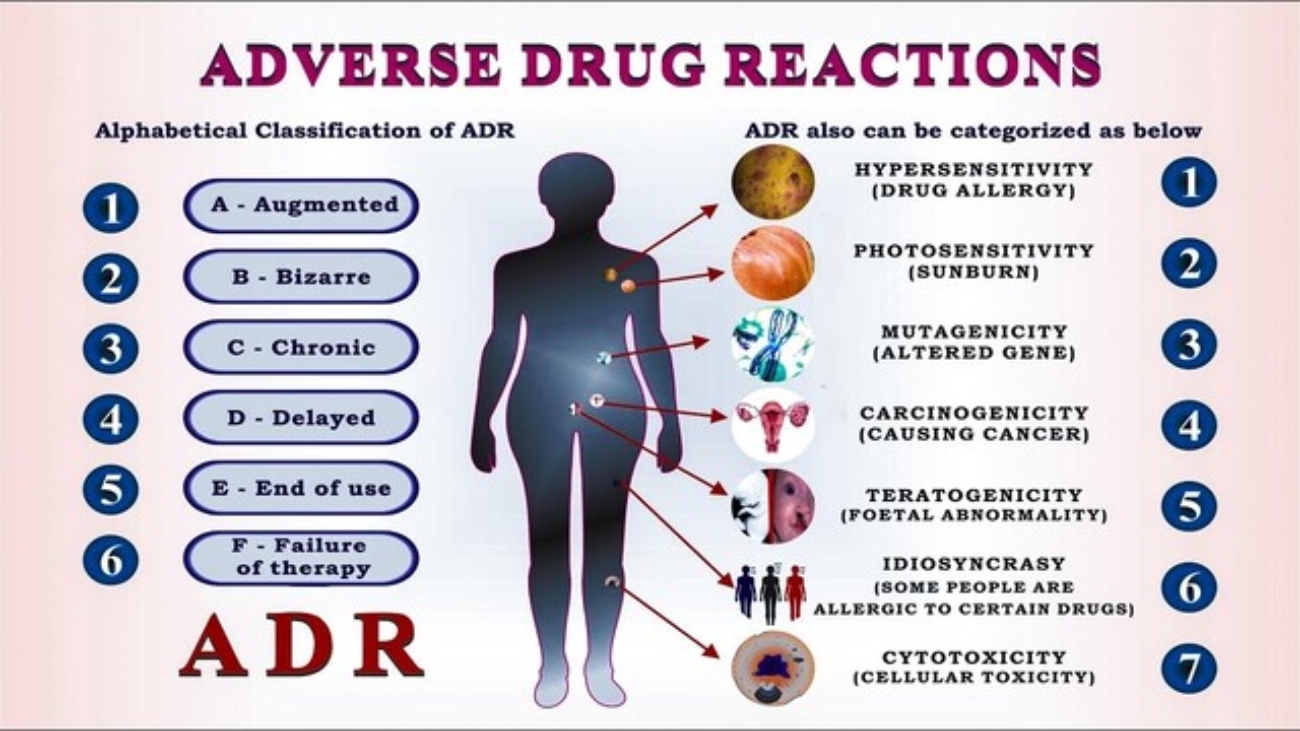
- Neoplastic (The drug causes cancer).
- Teratologic (The drug causes birth defects).
Examples of Type B: Anaphylaxis due to penicillin (a massive immune system overreaction). Stevens-Johnson syndrome (a severe, blistering skin reaction).
5. The Expanded Classification: Wills and Brown (A, B, C, D, E, F)
To make the system more comprehensive, Wills and Brown modified the Thompson classification, expanding it into six alphabetical categories (A through F).
Augmented
Concept: These are augmented (exaggerated) from the normal pharmacological properties of the drug. They are highly predicted, dose-related, preventable, and mostly reversible.
Frequency: They are the most common, accounting for 75% of all ADRs.
Examples: Anti-hypertensives (α1-antagonists) causing severe hypotension (low blood pressure). Anti-diabetics (Insulin) causing hypoglycemia.
Caution/Management: Decrease the dose. If that fails, withdraw and use an alternative drug.
Bizarre or Unpredictable
Concept: Unpredictable, uncommon, not related to the dose, and not related to the normal mechanism of drug action. They have higher mortality and morbidity.
Develop on the basis of:
- Immunological reaction (Allergy): e.g., Penicillin hypersensitivity.
- Genetic predisposition (Idiosyncratic reactions): A patient lacks a specific enzyme due to their genetics, causing a weird reaction to a drug.
- Pseudo-allergy: e.g., Ampicillin Rash (looks like an allergy but isn't mediated by IgE in the same way).
Examples: Anaphylaxis by penicillins. Stevens-Johnson syndrome.
Caution/Management: Stop the drug immediately. Avoid it entirely in the future. Instruct the patient to inform all future physicians about this allergy.
Chronic (Continuous) Use
Concept: Uncommon and unpredictable. These reactions are strictly related to the long-term accumulation of the dose or prolonged exposure over months or years.
Examples:
- Analgesic (NSAID) Nephropathy: Long-term, continuous daily use of painkillers (NSAIDs) causes interstitial nephritis (inflammation of the spaces between renal tubules) or renal necrosis. The kidneys slowly fail over years.
- Corticosteroids: Years of steroid use leads to the suppression of the Hypothalamic-Pituitary-Adrenal (HPA) axis. The body forgets how to make its own natural steroids.
Caution/Management: Reduce the dose or withdraw the drug. To prevent it, use alternate day therapy (intermittent therapy) or mega/pulse dose therapy (give a lot at once, then stop for a long time).
Delayed (Time Lag)
Concept: These become apparent only after some time has passed from the initial use of the drug. They are predictable, uncommon, and not dose-dependent.
Examples:
- Teratogenesis: Drugs causing birth defects. Classic Example: Thalidomide given to pregnant women for morning sickness caused Phocomelia (flipper-like fore limbs) in their babies months later.
- Mutagenesis / Carcinogenesis: Drugs that mutate DNA and cause cancer years later.
Crucial Clinical Note: Clear Cell Adenocarcinoma caused by DES (Diethylstilbestrol). Mothers were given DES in the 1950s to prevent miscarriage. Decades later, their teenage daughters developed a rare vaginal cancer (clear cell adenocarcinoma). This is the ultimate delayed reaction.
Caution/Management: Avoid use. Use only if absolutely indicated and life-saving.
End of Use (Dose Stopped Abruptly)
Concept: Uncommon but predictable. Occurs entirely because a drug is withdrawn too quickly. It causes drug withdrawal syndromes and rebound phenomenons.
Examples:
- Sudden withdrawal of long-term therapy with β-blockers. The heart has grown extra receptors to fight the blocker. If you remove the blocker suddenly, normal adrenaline hits all those extra receptors, inducing dangerous rebound tachycardia (fast heart rate) and severe hypertension.
- Sudden withdrawal of opiates (heroin, morphine) leading to severe physical withdrawal sickness.
Caution/Management: Never stop abruptly. Reintroduce slowly, then taper the drug gradually over weeks. Alternatively, use a concomitant drug with an antagonistic effect or a partial agonist to ease them off.
Failure of Therapy
Concept: Common. Simply put, the drug fails to do its job (ineffectiveness). It is dose-related and often caused by drug interactions or enzyme induction (the liver clears the drug too fast).
Examples: An inadequate dosage of an oral contraceptive, or taking a contraceptive alongside a liver enzyme inducer, leading to an unwanted pregnancy due to the failure of the oral contraceptive.
Caution/Management: Increase the dosage. Carefully consider the effects of concomitant (simultaneous) therapy that might be destroying the drug.
6. Teratogenicity: Drugs and Pregnancy (FDA Categories)
A teratogen is any agent that can disturb the development of an embryo or fetus. The FDA classifies drugs into five distinct risk categories (A, B, C, D, X) based on animal and human studies.
| Category | Animal Risk | Human Risk | Description & Examples |
|---|---|---|---|
| Category A | – (No Risk) | – (No Risk) | Studies have proven a complete absence of teratogenicity. Completely safe. Examples: Thyroid hormone, Folic acid (actually prevents defects). |
| Category B | +/- (Some risk or no studies) | -/0 (No risk or no studies) | Animal studies may show slight risk, but human studies show no risk. Generally considered safe. Example: AZT (Antiretroviral for HIV). |
| Category C | +/0 (Risk shown or no studies) | 0 (No human studies available) | Animal studies show an adverse effect, but there are no adequate human studies. Use only if benefit justifies the risk. Example: Aspirin. |
| Category D | + (Proven Risk) | + (Proven Risk) | Positive evidence of human fetal risk exists. However, the benefits may outweigh the risk in life-threatening situations for the mother. Examples: ACE inhibitors, Anticonvulsants (seizure meds). |
| Category X | + (Proven Risk) | + (Proven Risk) | Absolute Contraindication. The risks heavily outweigh any possible benefit. Never give to a pregnant woman. Examples: Oral contraceptives, statins (cholesterol drugs), high doses of Vitamin A, misoprostol, clomiphene. |
7. Other Forms and Terminology of ADRs
- Drug Induced Diseases: The drug creates a new pathology.
- Aspirin can cause PUDs (Peptic Ulcer Diseases) by eating away stomach lining protection.
- Tuberculosis (TB) drugs (like Isoniazid) are highly toxic to the liver and can cause drug-induced hepatitis.
- Idiosyncratic Drug Reaction: An abnormal, unexpected reaction caused by a patient's specific genetic predisposition.
- Example: Chloramphenicol (an antibiotic) can cause a rare, deadly condition called aplastic anemia (where the bone marrow stops making blood cells) in genetically susceptible individuals.
- Drug Intolerance: A lower threshold to the normal pharmacological action of a drug. Example: Chloroquine intolerance.
- Drug Allergies (Hypersensitivity Types 1, 2, 3, 4): Immune system attacks. Example: Type 1 Anaphylactic reactions to penicillins (life-threatening).
- Management: Immediate Adrenaline (Epinephrine) and corticosteroids.
- Prevention: Give a tiny "test dose" to check for allergy before a full dose.
- Phototoxicity & Photoallergies: Drugs that make the skin violently react to sunlight.
- Phototoxicity (acts like a severe sunburn): Caused by fluoroquinolones, tetracyclines.
- Photoallergies (immune reaction to sun+drug): Caused by sulfa drugs & fluoroquinolones.
- Drug Dependence: Psychological or physical reliance on a drug. Examples: Morphine, codeine, heroin.
8. Factors Affecting Drug Response and Variability
Why does 10mg of a drug work perfectly for Person A, but cause a severe ADR in Person B? Variability is driven by multiple patient-specific factors:
1. Body Weight
The average dose of a drug is usually calculated in terms of mg/kg of body weight. However, this basic calculation can be flawed:
- Edema: If a patient has edema (swelling), their weight increases solely due to the accumulation of Extracellular Fluid (ECF), not active tissue. Dosing based on this false weight will result in an overdose.
- Malnutrition: A severely malnourished person has a reduced capacity to metabolize drugs (fewer liver enzymes and proteins). Doses must be heavily reduced.
2. Age
Pharmacokinetics drastically change at the extremes of age.
- Newborns & Infants: Liver and renal functions are less developed. The Glomerular Filtration Rate (GFR) in the kidneys is very low. Crucially, the Blood-Brain Barrier (BBB) is much more permeable in infants, allowing drugs to easily cross into the brain and cause dangerous accumulation.
- Elderly: Both hepatic (liver) and renal (kidney) functions naturally decline with age, slowing down drug clearance and increasing the risk of toxicity.
3. Route of Drug Administration
The route governs the speed and intensity of the drug response.
- Intravenous (IV) doses are usually much smaller than oral doses because 100% of the drug enters the blood immediately. The onset of action is incredibly quick.
- A drug may have entirely different uses depending on the route.
- Given orally: It produces purgation (acts as a strong laxative).
- Applied locally on inflamed areas: It decreases swelling.
- Given intravenously (IV): It produces profound CNS depression and hypotension (lowers blood pressure, used in eclampsia).
4. Sex & Hormonal Status
- Females generally have a smaller body size and higher fat percentage, requiring doses on the lower side of the range.
- Physiological changes during pregnancy heavily alter drug disposition (more blood volume, faster kidney filtration). Also, drugs given during pregnancy may affect the fetus. Consideration must be given to menstruation and lactation (drugs passing into breast milk).
- Some drugs, like methyldopa and beta-blockers, interfere with sexual function (causing impotence) in males, but do not have this effect in females.
5. Genetic Factors & Tachyphylaxis
The amount of microsomal enzymes in the liver is genetically controlled. Because of this, the required dose of a drug can vary 4 to 6 folds among different people!
- Genetic Defect Example 1: G6PD deficiency. People with this genetic trait will experience massive hemolysis (red blood cell destruction) if given drugs like Primaquine (antimalarial) or Sulfonamides.
- Genetic Defect Example 2: Slow Acetylators. Some people genetically metabolize the TB drug Isoniazid very slowly, leading to toxic buildup and nerve damage.
Tachyphylaxis (Acute Tolerance)
Tachyphylaxis is defined as a rapid reduction in responsiveness to a drug due to repeated administration at frequent intervals. The drug stops working almost immediately.
How it works: It is usually seen with indirectly acting drugs like ephedrine, tyramine, and amphetamine. These drugs don't stimulate receptors themselves; instead, they act by forcing the body to release its stored catecholamines (like adrenaline). If you give the drug repeatedly, the synthesis of new catecholamines cannot match the rapid release. The body's stores deplete rapidly (like squeezing a sponge dry). Once the stores are empty, the drug has no effect. Another mechanism is the slow dissociation of the drug from receptors, blocking them from resetting.
6. Pathological Conditions (Disease Status)
Diseases heavily influence drug disposition. Hepatic (liver), renal (kidney), and cardiovascular (heart) diseases have a profound influence on drug clearance and actions. Drugs must be carefully monitored or avoided if these organs are failing.
7. Metabolic Disturbances & Time of Administration
- Metabolic: Changes in water, electrolytes, temperature, and acid-base balance modify drug effects.
- Example: Aspirin reduces body temperature only in the presence of a fever; it has absolutely zero effect on body temperature when it is normal.
- Example: Iron is absorbed much better by the body during states of iron deficiency compared to when levels are normal.
- Time of Administration: When you take a pill matters.
- Before meals: To prevent mixing with food, or to prevent the formation of insoluble complexes (e.g., Tetracycline binds to calcium in food and becomes useless).
- Immediate effect: Drugs for motion sickness must be taken before travel.
- Prevent side effects: Insulin and sulfonylureas must be given before meals to prevent dangerous hypoglycemia that would occur if given on an empty stomach with no food incoming.
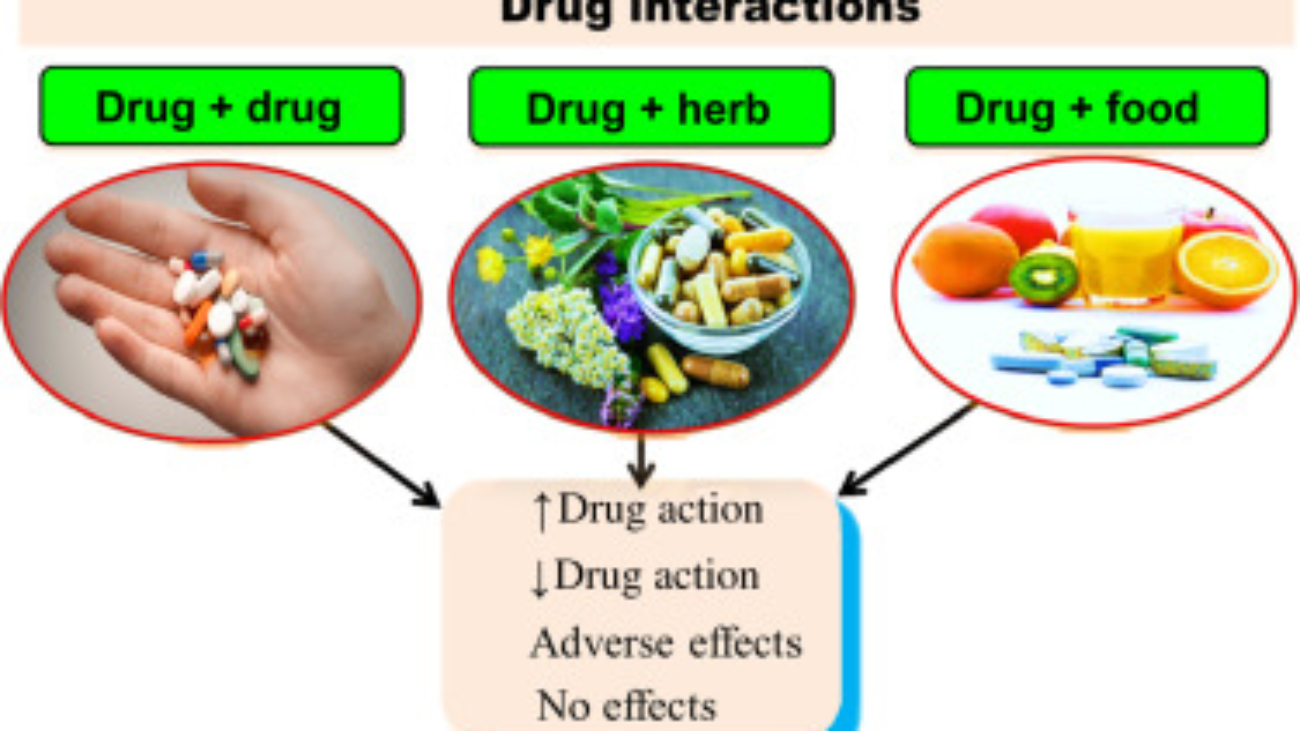
9. Drug Interactions
A drug interaction occurs when one drug modifies the response of another. This does not always mean concurrent use is forbidden; many are used beneficially or managed with dose adjustments. Interactions are split into two categories: Pharmacodynamic and Pharmacokinetic.
A. Pharmacodynamic Interactions
This is when the effect of one drug is changed by the presence of another drug acting at the same biochemical or molecular site (e.g., fighting for the same drug receptor or second messenger system). They might act on the same target organ, or different targets that share a common physiological process.
The results can be:
- Additive: 1 + 1 = 2. The effects simply add together.
- Synergistic: 1 + 1 = 10. The combined effect is massively greater than the sum of their individual effects.
- Potentiation: Drug A has no effect on a process, but makes Drug B much stronger.
- Antagonistic: 1 + 1 = 0. One drug cancels out or blocks the effect of the other.
B. Pharmacokinetic Interactions
This is when one drug alters the actual concentration (amount) of another drug in the system. It affects the A, D, M, E parameters: Bioavailability, Volume of Distribution, Peak level, Clearance, and Half-life.
Such changes lead to massive shifts in plasma concentrations, increasing the risk of side effects or diminishing efficacy. These are much more complicated and difficult to predict because the interacting drugs often have completely unrelated intended actions (e.g., a heartburn pill stopping an antibiotic from absorbing).
10. Massive List of Specific Interaction Examples
I. Drug-Drug Interactions
When a drug interferes with another drug.
- Aspirin + Warfarin → Synergism. Both thin the blood via different pathways. Result: Excessive, dangerous bleeding.
- Antibiotic + Blood thinner → Antagonism. Result: Less effect of the thinner. (Note to students: This is exactly what the lecture states. Some broad-spectrum antibiotics can alter gut flora and Vitamin K production, causing fluctuations in blood thinner efficacy).
- Decongestants + Antihypertensives → Potentiation. Decongestants narrow blood vessels. Result: Dangerously high blood pressure, defeating the blood pressure medicine.
- Codeine + Paracetamol → Addition. Both relieve pain through different mechanisms. Result: Increased, highly effective analgesic effect.
- Clavulanic acid + Amoxicillin → Synergism. Clavulanic acid blocks the bacterial enzyme that destroys amoxicillin. Result: Massively increased antibiotic effect (sold together as Augmentin).
- NSAID + Cox 2 inhibitors → Synergism. Both block clotting factors and irritate the stomach. Result: Increased bleeding and ulcer risk.
- SSRI's (Antidepressants) + Vitamin K (or Anticoagulants) → Synergism. SSRIs interfere with platelet aggregation. Result: Increased bleeding risk.
- Antiemetics (Anti-nausea) + Tranquilizers → Unknown/Dangerous effect. Both depress the central nervous system. Result: Breathing problems and severe sedation.
- H2 blockers + PPI's → Alteration. Both reduce stomach acid. Result: Massive increase in the pH (alkalinity) of the stomach, which can stop other drugs from dissolving.
- Phenobarbital + Warfarin → Antagonism. Phenobarbital aggressively induces liver enzymes, which chew up and destroy the Warfarin too fast. Result: Less effect of the blood thinner, risking clots.
- Erythromycin + Warfarin → Synergism. Erythromycin blocks liver enzymes, preventing Warfarin from being cleared. Warfarin builds up. Result: Increased, severe bleeding.
II. Drug-Food Interactions
When the food you eat stops a medicine from working the way it should.
- Bisphosphonates + Any drug/food → Reduced effectiveness. These bone medications must be taken on a strictly empty stomach with pure water, or they will not absorb at all.
- Benzodiazepines + Grapefruit juice → Inhibits liver enzymes. Grapefruit blocks the enzyme CYP3A4, causing the sedative to build up to toxic, coma-inducing levels.
- Digoxin + Oatmeal (High fiber) → Decreased absorption of the heart drug.
- Aspirin + Milk → Upset stomach.
- Acetaminophen (Paracetamol) + Alcohol → Liver damage. Both rely on the same liver pathways; combined, they produce a highly toxic metabolite.
- MAO Inhibitors (old antidepressants) + Food containing Tyramine (aged cheese, wine) → Severe headache / Hypertensive Crisis. MAOIs stop the breakdown of tyramine, leading to a massive, lethal spike in blood pressure.
- Tetracyclines + Calcium food (Dairy/Milk) → Reduced absorption. The calcium binds physically to the drug in the gut, forming an insoluble complex that is pooped out.
- Warfarin + Vitamin K foods (Spinach, Kale) → Reduced effect. Warfarin works by blocking Vitamin K. Eating too much Vitamin K reverses the drug, causing blood clots.
- Celecoxib + Milk → Upset stomach.
- Naproxen + Fatty food → Upset stomach / altered absorption.
- Oxycodone + Alcohol → Coma, asthma (respiratory depression). Combining two powerful CNS depressants is lethal.
- Caffeine + Food → Rapid heart beat.
III. Drug-Disease Interactions
When a drug perfectly treats one disease but accidentally worsens a secondary existing medical condition.
- Nasal decongestants + Hypertension → Increased blood pressure. The decongestant clears the nose by squeezing blood vessels; it squeezes vessels everywhere else too, spiking blood pressure.
- NSAID’S + Asthmatic patients → Airway obstruction. NSAIDs block COX enzymes, forcing all arachidonic acid down the LOX pathway. This produces leukotrienes, which cause severe bronchoconstriction (asthma attacks).
- Minoxidil + Heart failure → Fluid retention. Minoxidil causes vasodilation, prompting the kidneys to aggressively retain sodium and water, drowning a weak heart.
- Calcium channel blocker + Heart failure → Negative inotropic activity. These drugs weaken the force of heart muscle contractions. A failing heart cannot afford to be weakened further.
- Nicotine + High blood pressure → Increased heart rate and BP. Nicotine is a powerful stimulant and vasoconstrictor.
- Beta blockers + Heart failure / Asthma → Worsen asthma. Non-selective beta blockers slow the heart (treating heart issues) but accidentally block Beta-2 receptors in the lungs, causing deadly airway spasms in asthmatics.
- Metformin + Heart failure → Increased lactate level. Metformin can cause a rare buildup of lactic acid (lactic acidosis). Heart failure causes poor oxygen delivery to tissues, skyrocketing the risk of this fatal complication.
Adverse Drug Reactions Quiz
Pharmacology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Adverse Drug Reactions Quiz
Pharmacology
Preparing questions...
Exam Completed!
See your performance breakdown below.