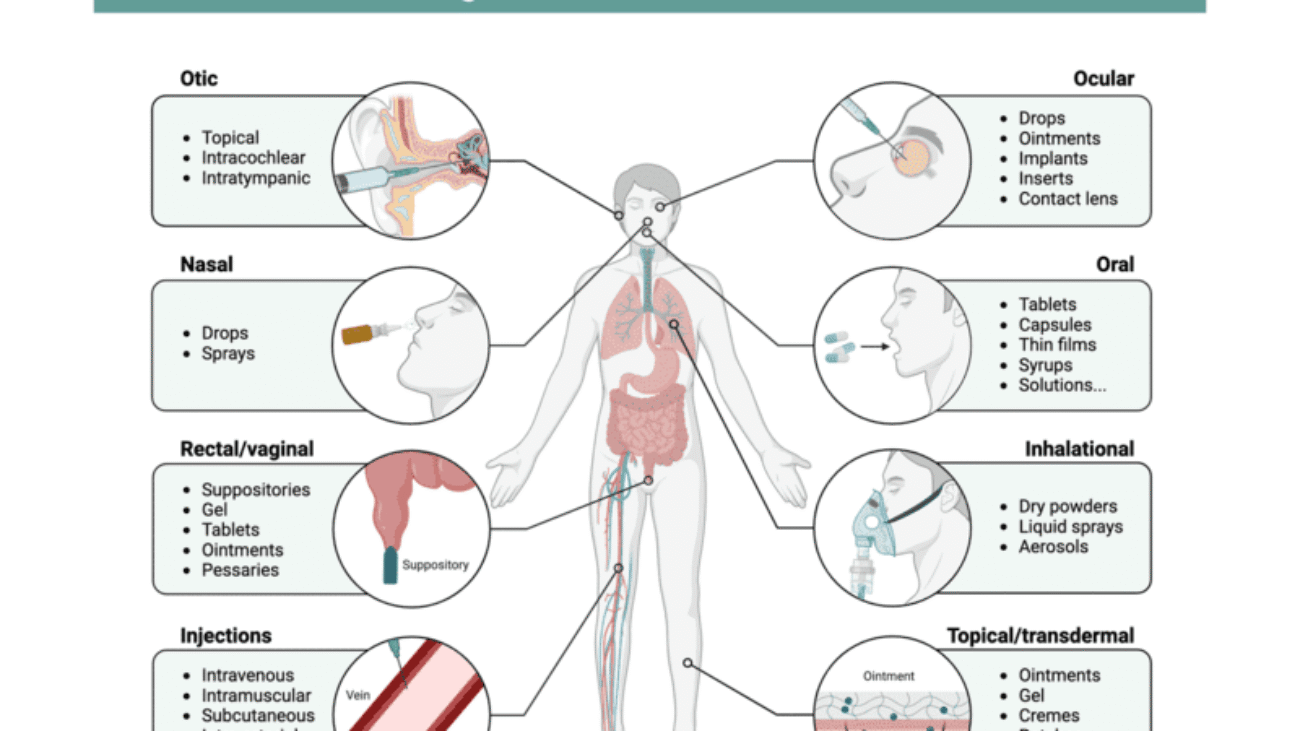
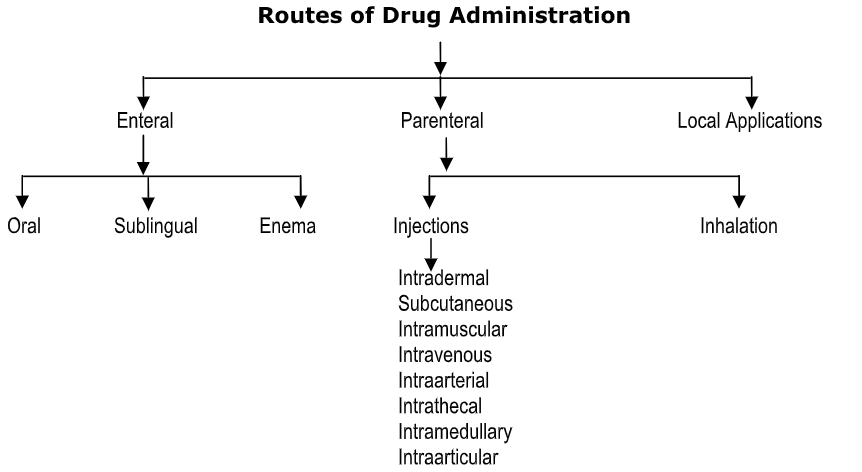
Routes of Administration
Fundamental Definitions and Concepts
What is a Drug?
In the strictest scientific sense, a drug is defined as any chemical agent or substance which affects, alters, or modifies any biological process within a living organism. It is important to realize that the body does not distinguish between a "therapeutic medication," an "environmental toxin," or a "recreational substance"—to the body's cells, they are all simply foreign chemicals (xenobiotics) that bind to biological targets and induce a change.
What is Pharmacology?
Pharmacology is the comprehensive scientific study of exactly how these drugs affect biological systems. It investigates the entire lifecycle of a drug interaction: from how the drug is manufactured and sourced, to how it travels through the bloodstream, how it binds to microscopic cellular receptors, and ultimately, how the body destroys and removes it.
The Five Major Branches of Pharmacology
To fully understand drug action, pharmacology is systematically divided into distinct domains:
1
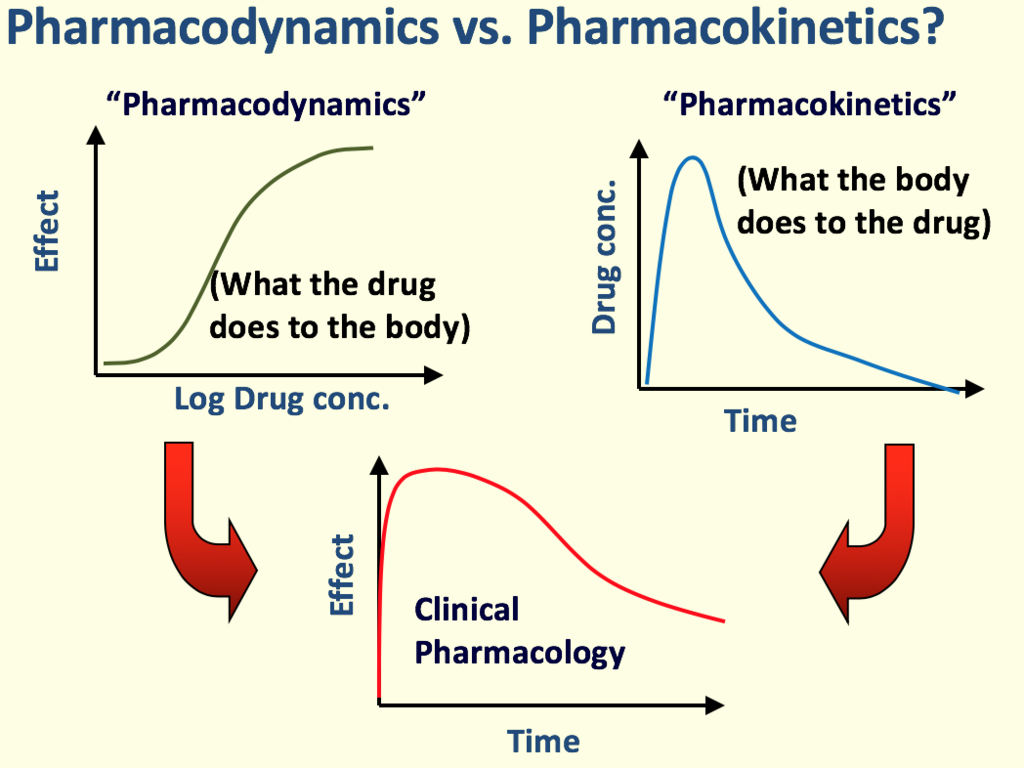
Pharmacokinetics
"What the body does to the drug." This encompasses the four pillars of drug biodisposition: Absorption (getting in), Distribution (moving around), Metabolism/Biotransformation (being broken down by enzymes), and Excretion (leaving the body).
2
Pharmacodynamics
"What the drug does to the body." This looks at the microscopic level: drug receptors (the protein locks that drugs fit into), the physiological effects of the drug, cellular responses, and potential toxicity or adverse effects.
3
Pharmacotherapeutics
The clinical study of the strictly medical use of drugs to prevent, diagnose, or treat diseases.
4
Pharmacognosy
The highly specialized study of identifying, extracting, and isolating crude materials from natural sources to be used as drugs.
5
Toxicology
The study of the poisonous, adverse, and toxic effects of chemicals on living systems.

Sources of Drugs and Forms of Medication
Sources of Drugs (Pharmacognosy)
Historically, all drugs came from nature. Today, we source drugs from five primary categories:
- Plants: The oldest source of medicine. Examples include Morphine (from the opium poppy), Digoxin (from the foxglove plant for heart failure), and Quinine (from the cinchona tree bark for malaria).
- Animals: Historically, many hormones were extracted from slaughtered livestock. Examples include Insulin (previously extracted from pig and cow pancreases) and Heparin (a blood thinner extracted from pig intestines).
- Minerals: Earthly elements used directly for health. Examples include Iron (for anemia), Lithium (for bipolar disorder), and Magnesium (as an antacid or laxative).
- Synthetic: Today, the vast majority of drugs are entirely synthesized in chemistry laboratories. This allows for massive scaling, precise purity, and the structural modification of natural drugs to reduce side effects.
- Microbes: Many life-saving drugs are produced by harnessing bacteria and fungi. The most famous example is Penicillin (produced by the Penicillium fungus).
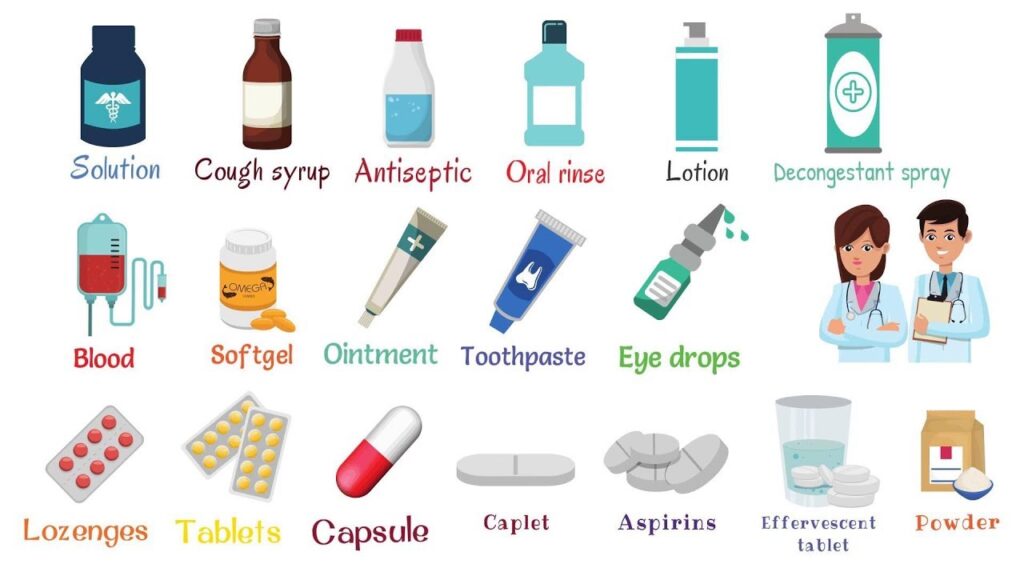
Forms of Medication
Medications are practically never pure, raw chemicals. They are carefully formulated into specific "preparations" or "dosage forms." The form of the medication strictly dictates its route of administration. The composition of the medicine is intricately designed by pharmaceutical scientists to enhance its absorption, dictate its metabolism rate, and ensure patient compliance.
Common forms include:
- Tablet: A solid dosage form made by highly compressing powdered drug and inactive binders into a hard pill.
- Capsule: A drug enclosed within a gelatin shell. They dissolve quickly in the stomach, releasing the powder or liquid inside.
- Elixir: A clear, sweetened, hydro-alcoholic liquid intended for oral use, perfect for drugs that do not dissolve easily in pure water.
- Enteric-coated: A specially designed tablet with an acid-resistant shell. It passes through the highly acidic stomach unharmed and only dissolves when it reaches the alkaline environment of the small intestine. This protects sensitive drugs from destruction and protects the stomach from irritating drugs.
- Suppository: A solid, bullet-shaped mass that is inserted into a body cavity (rectum or vagina) which is explicitly designed to melt at exact human body temperature (37°C) to release the drug.
- Suspension: A liquid preparation containing undissolved solid drug particles. Because the drug settles at the bottom, these must be shaken vigorously before administration.
- Transdermal patch: An adhesive patch placed on the skin that delivers a specific, slow, and continuous dose of medication through the skin and directly into the bloodstream.

Routes of Drug Administration
A route of administration is the specific anatomical path by which a drug, fluid, poison, or other substance is brought into contact with the body.
Routes of administration are broadly classified into three main channels based on whether they act locally or systematically, and whether they involve the digestive tract:
- Enteral: Through the gastrointestinal tract (Oral, Sublingual, Buccal, Rectal).
- Parenteral: Bypassing the gastrointestinal tract, usually via injection (IV, IM, SC, etc.).
- Topical: Applied to a specific surface for a localized effect (Skin, eyes, ears, lungs).
Enteral Routes of Administration
The term Enteral comes from the Greek word enteron, meaning intestine. It refers to anything involving the alimentary tract, from the mouth down to the rectum.
A. Oral Route or Per Os (P.O.)
The oral route involves swallowing a drug. It is the most common, oldest, and generally most universally accepted route of administration. It utilizes the body's natural machinery used for digesting food, absorbing nutrients, and eliminating wastes.
Advantages of the Oral Route:
- It is safe: Because absorption is relatively slow, there is a window of opportunity to induce vomiting or pump the stomach in case of an accidental overdose.
- It is convenient: Patients can take it themselves anywhere.
- It is cheap: Tablets and capsules do not require sterile manufacturing conditions like injectable fluids do.
- No skilled personnel required: The patient does not need a nurse or doctor to administer the dose.
Disadvantages of the Oral Route:
- Unpalatable drugs: Bitter or foul-tasting drugs can cause severe irritation to the intestinal tract, resulting in nausea, vomiting, and diarrhea.
- Destruction by enzymes and acid: Some drugs are completely annihilated by stomach acid (HCl) or digestive enzymes before they can be absorbed. For example, Insulin is a protein; if swallowed, the stomach digests it just like a piece of meat, destroying its therapeutic value.
- Not suitable for emergencies: It takes time for a pill to reach the stomach, dissolve, and be absorbed into the blood. When quick, life-saving action is desired, this route is too slow.
- Not suitable for unconscious patients: An unconscious or actively vomiting patient cannot safely swallow a pill due to the high risk of choking or aspirating the drug into the lungs.
- Requires patient cooperation: Uncooperative patients (e.g., small children, psychiatric patients, or animals) may refuse to swallow or secretly spit the pill out.
- Slow, unpredictable, and irregular absorption: The presence of food (which delays gastric emptying), the varied stages of digestion, and the fluctuating acidity/alkalinity of digestive juices create massive variability in how much drug actually gets absorbed.
Crucial Concept
The First-Pass Effect
The oral route is highly not recommended for drugs undergoing an extensive First-Pass Effect.
What is it? When a drug is absorbed through the stomach and small intestine, it does NOT go straight to the heart to be pumped to the rest of the body. Instead, the blood from the gut is funneled directly into the Hepatic Portal Vein, which leads straight into the liver.
The liver acts as a chemical checkpoint. It is packed with drug-metabolizing enzymes. Many drugs are heavily metabolized (destroyed or altered) by the liver to a great extent before they ever reach the systemic circulation to be distributed to their site of action. If a drug has a 90% first-pass effect, swallowing 100mg means only 10mg will actually reach the rest of the body.
B. Sublingual Route
Derived from Latin (sub = under, lingua = tongue), this route involves placing the drug strictly underneath the tongue.
The mucosa (inner lining) under the tongue is extremely thin and supported by a massive, rich network of small blood vessels (capillaries). Drugs placed here dissolve in saliva and diffuse directly across the thin membrane into these veins.
Sublingual Classic Example: Nitroglycerine
Nitroglycerin is a highly lipid-soluble drug used to treat severe angina (crushing chest pain caused by the heart muscle not getting enough oxygenated blood). If given orally, the liver destroys nearly 100% of it via the first-pass effect. When placed sublingually, it bypasses the liver entirely, jumping directly into the systemic venous circulation. It reaches the heart in seconds, dilating blood vessels and saving the patient's life instantly.
Advantages:
- Rapid absorption: Due to the rich blood supply and thin membrane.
- Low enzyme activity: Saliva does not have the harsh drug-destroying enzymes that the stomach does.
- NO first-pass effect: The veins under the tongue drain into the superior vena cava, bypassing the liver entirely.
- Quick termination: If the patient experiences a bad side effect, they can simply spit the remaining tablet out to immediately stop absorption.
Disadvantages:
- Discomfort: Holding a tablet under the tongue and avoiding swallowing saliva is uncomfortable.
- Possibility of swallowing: If accidentally swallowed, the drug will be subjected to the first-pass effect and rendered useless.
- Unpalatable & bitter drugs: It is highly unpleasant to hold a bad-tasting drug in the mouth.
- Irritation: Can cause ulcers or irritation of the delicate oral mucosa.
- Volume limitations: Only very small quantities of a drug can be administered this way.
C. Buccal Cavity Route
Similar to sublingual, but the dosage form is placed snugly between the gums and the inner lining of the cheek (the buccal pouch).
Advantages:
- Ease of administration and termination: Can be easily placed and easily removed.
- Avoidance of hepatic first-pass metabolism: Like the sublingual route, it drains directly into systemic veins.
- Salivary secretion: Ensures adequate dissolution of the drug.
- Bypasses stomach acid: Highly suitable for drugs prone to acidic degradation.
- Minimal diffusion hindrance: A lack of heavy mucus secretion from goblet cells in the cheek means the drug diffuses easily without a mucus barrier building up beneath it.
- Can be used in unconscious patients: Can be slipped into the cheek pouch of an unresponsive patient safely (if formulated correctly to avoid choking).
- Controlled release: Initial mucoadhesion (sticking to the cheek) time can be engineered into the tablet to provide a steady, slow release of the drug over hours.
Limitations:
- Not suitable for drugs requiring high, bulky doses.
- High possibility that the patient forgets the tablet is there and accidentally swallows it.
- Eating, drinking, and talking may be severely restricted while the tablet is in place.
- Restricted for drugs that are severe irritants, have a terribly bitter taste/odor, or are unstable at salivary pH.
- Limited surface area available for drug absorption compared to the massive surface area of the small intestine.
- Lower permeability: The buccal membrane is thicker and slightly less permeable than the incredibly thin sublingual membrane.
D. Rectal Administration
In this route, the drug is administered deep into the rectum. The drug may be given rectally for a localized effect (like treating hemorrhoids) or for a full systemic effect when the patient cannot take medications orally.
Different Forms of Rectal Administration:
- Suppositories: Small, solid, cone-shaped medicated masses. They are inserted into the rectum where they melt cleanly at body temperature. Example: Ergotamine suppositories for severe migraine headaches when the patient is too nauseous to swallow pills.
- Enemas: The procedure of introducing large volumes of liquid (solutions or suspensions) directly into the rectum and colon via the anus.
- Evacuant Enema: Used as a bowel stimulant to treat severe constipation (e.g., soft soap enema or MgSO4 enema). The volume may reach up to 2 liters. Note: They should be warmed to body temperature before administration to prevent thermal shock to the bowel.
- Retention Enema: Volume does not exceed 100 ml, and no warming is strictly needed. Designed to be held in the rectum to be absorbed.
- Local effect: e.g., A Barium enema used as a contrast substance to allow doctors to take highly detailed radiological imaging (X-rays) of the lower bowel.
- Systemic effect: The administration of substances into the bloodstream. Done when mouth delivery is impossible (e.g., antiemetics to stop vomiting, or nutrient enemas containing carbohydrates, vitamins, and minerals for starving patients who cannot eat).
Advantages of Rectal Administration:
- Incredibly useful for delivering drugs during active, severe vomiting or when the patient is totally unable to swallow (dysphagia or unconsciousness).
- Suitable for drugs that are highly irritant to the stomach lining, which would otherwise cause severe ulcers (e.g., Aminophylline, Indomethacin).
- Of particular, exceptional value in pediatric medicine, especially for small, uncooperative children who refuse to swallow bitter pills or syrups.
- Partial avoidance of First-Pass Effect: The venous drainage of the rectum is split. The lower and middle rectal veins drain straight into the systemic circulation (bypassing the liver), while only the superior rectal vein drains into the portal system. Thus, it experiences little to no first-pass effect compared to oral ingestion.
- Higher blood concentrations can often be rapidly achieved compared to oral dosing.
Disadvantages of Rectal Administration:
- Inconvenient and Embarrassing: Most patients (and caregivers) find this route culturally or personally objectionable and deeply embarrassing.
- Absorption is slow, erratic, and irregular: The rectum does not have the microvilli of the small intestine, making absorption highly unpredictable, especially if the rectum is full of fecal matter.
- Irritation: Repeated administration can easily cause severe inflammation, proctitis, or irritation of the delicate rectal mucosa.
Parenteral Routes of Administration
The term parenteral is literally translated from the Greek words: para (meaning outside or alongside) and enteron (meaning the intestine). Therefore, parenteral administration means any delivery method that bypasses the intestinal tract.
Practically, parenteral administration involves injection or infusion by means of a hollow needle or catheter inserted directly through the skin barrier into the body tissues or blood vessels.
Parenteral forms deserve extremely special clinical attention due to:
- Their structural and manufacturing complexity (they must be absolutely 100% sterile and free of pyrogens).
- Their widespread use in modern medicine.
- Their massive potential for profound therapeutic benefit (saving lives instantly) coupled with severe danger (if the wrong dose is injected, it cannot be easily removed).
General Advantages of Parenteral Administration:
- The drug is never destroyed by destructive gastric acid or digestive enzymes.
- A much higher, more accurate concentration of the drug in the blood is almost always achieved because hepatic metabolism via the First-Pass Effect is completely, 100% avoided.
- Absorption into the bloodstream is usually complete, highly measurable, and highly predictable.
- In emergency medicine, this method is unparalleled. If a patient is unconscious, seizing, uncooperative, or violently vomiting, parenteral therapy is absolutely necessary to save their life.
General Disadvantages of the Parenteral Route:
- It is highly expensive because all parenteral preparations require rigorous sterilization, specialized glass ampoules, and single-use syringes.
- Pain, fear, and psychological distress almost always accompany or follow the injection.
- It strictly requires the services of a professionally skilled personnel (nurses, doctors, paramedics) because it is technically difficult, dangerous, and physically awkward for a patient to safely perform a deep injection on themselves (with some exceptions like insulin pens).
Specific Parenteral Routes:
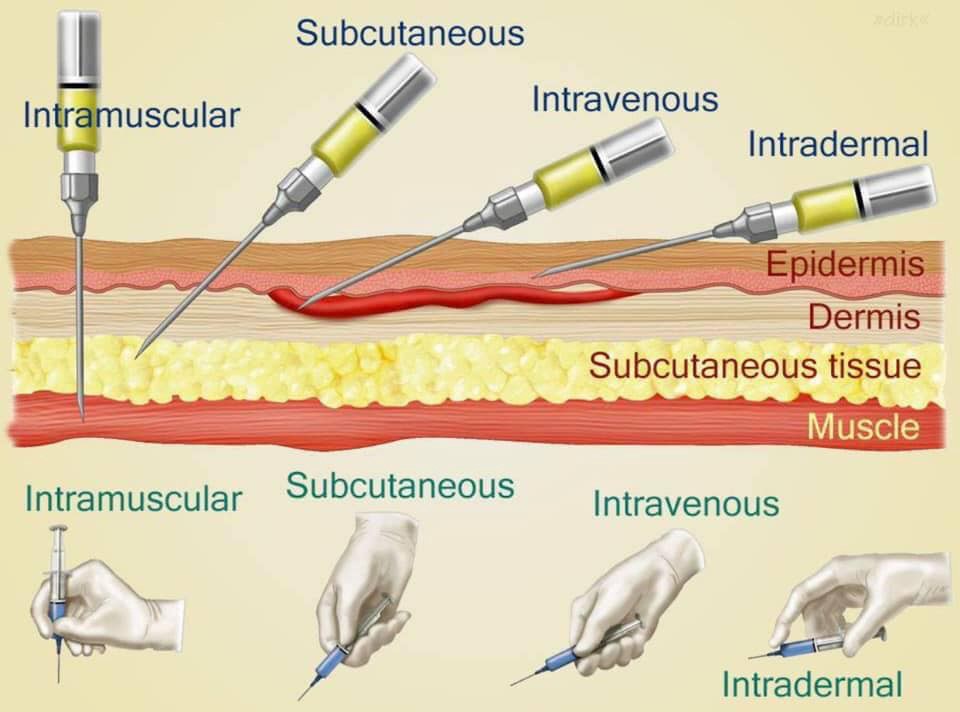
A. Subcutaneous (S.C.)
The drug is dissolved in a small volume of vehicle (liquid) and injected deep beneath the epidermis and dermis, directly into the fatty subcutaneous tissue.
- Because fat tissue has a relatively poor blood supply compared to muscle, absorption is slow and highly uniform.
- Because absorption is slow, the duration of drug action is heavily prolonged. This makes it incredibly useful when continuous, steady presence of the drug in tissues is needed over a long period.
- Depot Preparations: The usefulness is astronomically enhanced by "depot" preparations. These are chemically modified drugs that dissolve incredibly slowly in the fat, releasing the active drug over hours, days, or even months (e.g., long-acting basal insulins).
- Implants: An extreme form of SC delivery. A small incision is made in the skin, and a solid, sterile pellet or porous capsule is surgically slipped into the loose tissues and stitched up. It releases drugs for years (e.g., hormonal contraceptive implants like Nexplanon).
Caution: Substances causing chemical irritation to tissues must never be injected S.C., otherwise they will cause agonizing pain, sloughing, and severe necrosis (deadening/rotting of the tissues) at the injection site.
B. Intramuscular (I.M.)
The injection is made deep, straight down (usually at a 90-degree angle) directly into the belly of skeletal muscle tissue. The best and safest sites are the large, thick muscles: the deltoid muscle in the shoulder, or the gluteus muscles in the buttocks.
Advantages:
- Absorption is reasonably uniform.
- Rapid onset of action: Muscle tissue is highly vascularized (rich in blood vessels), meaning the drug is swept into the bloodstream much faster than a subcutaneous injection.
- Mild irritants can be given: Muscle tissue is much less sensitive to pain and chemical irritation than subcutaneous fat.
- Absorption is complete, predictable, and fully avoids gastric factors and the first-pass effect.
- The speed of absorption depends on the liquid vehicle: aqueous (water-based) solutions absorb very quickly, while oily preparations absorb slowly and act as a depot.
Disadvantages:
- Volume limits: Only up to about 10mL of drug can be forced into a muscle before it becomes dangerous and tearing occurs.
- Local pain, soreness, and potentially a sterile abscess can form.
- Risk of infection if the skin isn't cleaned properly.
- Nerve Damage: If injected in the wrong quadrant of the gluteus, the needle can strike and permanently sever or chemically burn the massive sciatic nerve, causing permanent leg paralysis.
C. Intravenous (I.V.)
The drug solution is injected directly through the wall of a vein into the lumen, where it instantly mixes and is diluted in the returning venous blood. The drug is carried straight to the Right side of the Heart, pumped to the lungs, and then circulated to all body tissues.
Advantages:
- 100% Bioavailability: Since it goes directly into the blood, the desired therapeutic concentration is achieved immediately, within seconds. This rapid onset is not possible by any other procedure.
- This is the only route for giving massive volumes of therapeutic fluids (e.g., 1-2 Liters of saline for dehydration, or whole Blood Transfusions).
- Certain drugs that are highly irritant can only be given IV. Why? Because the rapid flow of blood inside the vein dilutes the irritant instantly, protecting the vessel wall.
Disadvantages:
- No turning back: Once the drug is pushed into the vein, nothing can be done to physically retrieve it or prevent its action. An overdose here is a catastrophic emergency.
- Requires immense technical skill to find a vein, insert the needle correctly, and minimize the risk of the needle slipping out of the vein (extravasation). If an irritant drug leaks into the surrounding S.C. tissues, it causes severe necrosis.
- Air Embolism: If the syringe contains a large air bubble, injecting it into the vein can cause the air to travel to the heart or lungs, blocking blood flow and causing sudden death.
- Local vein complications: Irritation, cellulitis, and Thrombophlebitis (inflammation and blood clotting of the vein).
- Generally considered the "less safe" route simply due to the severity and speed of potential adverse reactions.
D. Intradermal (I.D.)
A very shallow injection where the drug is placed exactly into the papillary layer of the dermis (the thick layer of skin just beneath the very outer epidermis). It produces a small "bleb" or blister-like bump on the skin.
- It is highly painful because the dermis is packed with sensory pain nerves.
- Main uses:
- Inoculations: Administration of specific vaccines that require powerful local immune responses (e.g., the BCG vaccination for active immunization against Tuberculosis, or the historical smallpox vaccine).
- Sensitivity/Allergy Testing: Injecting minute amounts of a substance (like Penicillin, Anti-Tetanus Serum - ATS, or environmental allergens) to visually watch for a localized allergic skin reaction before giving a full systemic dose.
E. Intra-articular (Intra-synovial)
The needle is advanced directly into the joint cavity (the space between two bones filled with synovial fluid). This localizes the drug's intense action precisely at the site of administration without affecting the rest of the body.
- Example: Injecting strong corticosteroids (like Hydrocortisone acetate) directly into a swollen knee joint for the treatment of severe Rheumatoid Arthritis.
- Because joints are incredibly sensitive, a local anesthetic is almost always added to the syringe to minimize the agonizing pain of the fluid expansion.
- Strict asepsis (absolute sterility) must be maintained. Introducing even a single skin bacteria into a joint cavity can cause a devastating, cartilage-destroying joint infection.
F. Intra-cardiac
The needle is plunged through the chest wall, between the ribs, and directly into the muscular wall or chamber of the heart.
- Used almost exclusively in dramatic cardiac arrest scenarios where intra-cardiac injection of Adrenaline (Epinephrine) is made for emergency resuscitation to restart a stopped heart.
- Note: Very few modern case reports support this "Pulp Fiction" style injection in closed-chest CPR due to the risk of lacerating coronary arteries. It is largely reserved for use during an emergent open thoracotomy (chest is already cracked open).
G. Intra-arterial
The drug is injected directly into a high-pressure artery (which carries blood away from the heart to a specific organ).
- It is used to violently localize a drug's effects in one particular tissue, organ, or limb, intentionally starving the rest of the body of the drug.
- Examples: Potent, highly toxic anticancer drugs (chemotherapy) are shot directly into the artery feeding a tumor, destroying the tumor while sparing the patient systemic toxicity. Also used for injecting radio-opaque contrast dyes to diagnose peripheral vascular blockages via X-ray.
- Requires a highly competent, specialized physician.
- There is absolutely zero fear of the first-pass effect, as arterial blood goes straight to the organ tissues.
Inhalation and Topical Routes
A. Inhalation (Pulmonary Absorption)
Gaseous and highly volatile liquid drugs are inhaled deeply into the lungs. The lungs possess a massive surface area of pulmonary endothelium (millions of microscopic alveoli) surrounded by a dense web of capillaries.
- Because the blood-air barrier is incredibly thin, drugs are absorbed immediately and reach the systemic circulation and brain rapidly (e.g., general anesthetics like Isoflurane).
- Localized Inhalation: Drugs like Bronchodilators (e.g., Albuterol/Salbutamol for asthma) are given via metered-dose inhalers in aerosolized form. Modern inhalers allow the supply of accurately metered, microgram doses of drugs straight to the smooth muscle of the airways, minimizing systemic side effects like heart palpitations.
B. Topical Routes of Administration
Topical administration is the direct physical application of a drug strictly to the surface of the skin or a specific mucous membrane.
1. Skin (Epidermal / Transdermal)
Normally, drugs applied to healthy, unbroken skin are very poorly absorbed because the outer epidermis (stratum corneum) is a tough, dead, waterproof shield. However, the living layer beneath it (the dermis) is highly permeable to solutes.
- Local Action: Drugs are applied as creams, thick ointments, pastes, or poultices for local conditions (rashes, eczema).
- Enhanced Absorption: Systemic absorption happens rapidly and dangerously through abraded, burned, or denuded skin where the barrier is gone. Severe inflammation, which brings massive cutaneous blood flow to the skin, also radically promotes absorption.
- Inunction: The physical act of vigorously rubbing a drug suspended in a highly oily/lipid vehicle deep into the skin to force absorption.
- Transdermal Patches: A specialized adhesive patch that deliberately drives drug absorption entirely through the intact skin for a systemic action.
- Provides beautifully stable, flat-line blood levels of the drug for days.
- Completely bypasses hepatic first-pass metabolism.
- Limitation: The drug must be incredibly potent (active at microgram levels) and highly lipophilic (fat-soluble) to penetrate the skin. If a drug requires a large dose, the patch would have to be absurdly, impractically large. Examples include Nicotine patches, Fentanyl pain patches, and Scopolamine motion-sickness patches.
2. Mucous Membranes
Mucous membranes line all the wet, internal pathways of the body exposed to the outside. Drugs are applied here primarily for their local action.
- Mouth and Pharynx:
- Bitters: Foul tasting liquids applied to the tongue strictly for their neurological reflex action to stimulate saliva and gastric acid to improve sluggish digestion.
- Boroglycerine and Gentian Violet: Thick paints applied as astringents and antiseptics for localized mouth ulcers or oral thrush (fungal infections) directly on the buccal mucosa.
- Stomach & Intestine: While swallowing is usually "enteral," taking a liquid Antacid to chemically neutralize secreted stomach HCl, or an Emetic to locally irritate the stomach to induce violent vomiting after poisoning, are considered local topical actions within the gut tube.
- Respiratory Tract: For severe sinus infections or colds, Tincture of Benzoin in steam inhalations acts locally to soothe raw airways and give relief from chest congestion. Phenylephrine nasal drops physically shrink swollen local blood vessels to clear a blocked nose.
- Vagina: Drugs formulated as a solid pessary, cream, or dissolving tablet are inserted to treat aggressive local vaginal infections (like yeast infections or bacterial vaginosis). While some systemic absorption can occur due to the rich blood supply, this route is clinically restricted to local treatment.
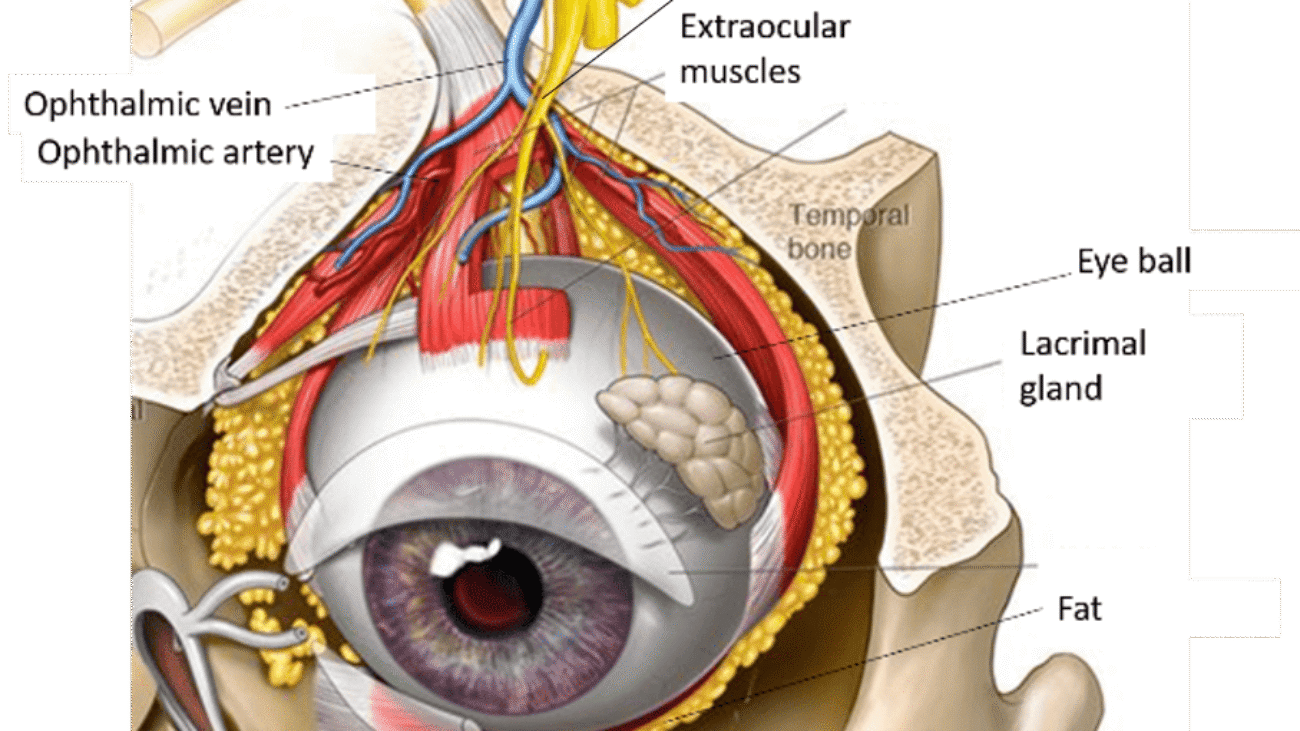
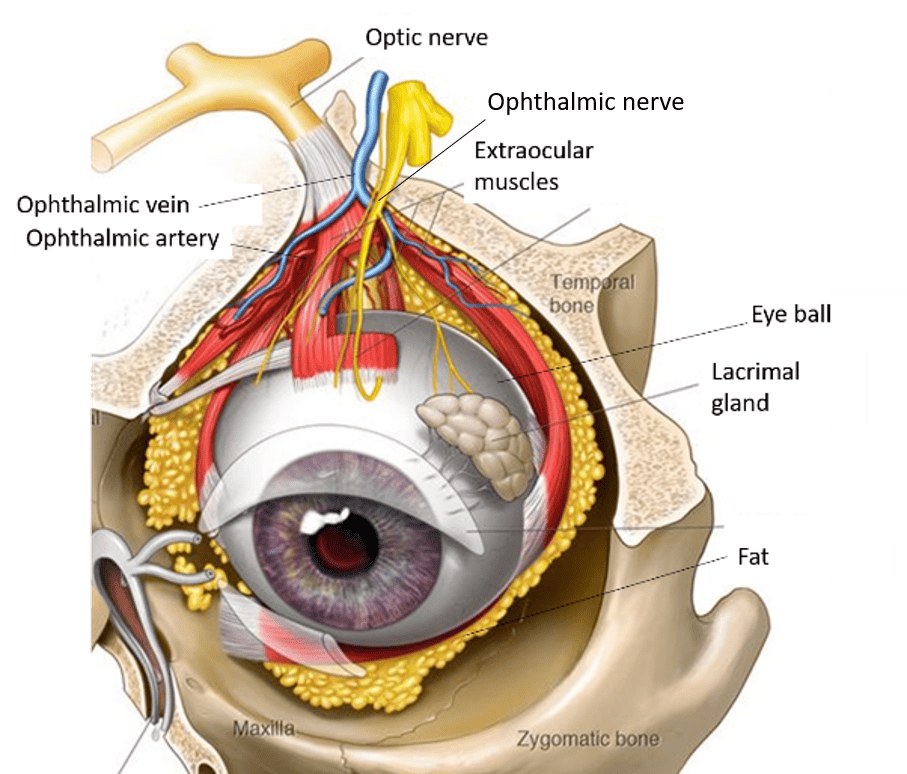
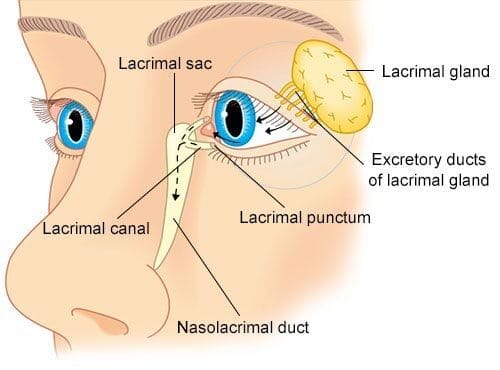
- Conjunctivae (The Eyes): The delicate, wet membrane lining the eyelids and covering the eyeball.
- Mydriatics: Eye drops forced to locally dilate the pupil (used by eye doctors to see into the back of the eye).
- Miotics: Drops used to aggressively constrict the pupil (often to treat Glaucoma).
- Local anesthetics, antiseptic drops, and antibiotic ointments are applied here strictly for superficial eye infections or surgeries.
Summary: Advantages & Disadvantages of Topical Routes
- Advantages: Provides spectacular local therapeutic effects directly where the problem is. Because it is poorly absorbed into the deeper layers and systemic blood, there is a massively lower risk of severe systemic side effects. The Transdermal sub-route offers the holy grail of steady-state drug levels without pills or needles.
- Disadvantages: Highly limited to localized problems (with the exception of specialized patches). Messy, can stain clothing (ointments/pastes), and is heavily dependent on the physical condition of the skin barrier.