Krebs / TCA Cycle Exam
Krebs Cycle Exam
Biochemistry: TCA/Krebs Cycle Exam
Test your knowledge with these 40 questions.
TCA/Krebs Cycle Exam
Question 1/40
Exam Complete!
Here are your results, .
Your Score
38/40
95%
Glycolysis Exam
Glycolysis Exam
Biochemistry: Glycolysis Exam
Test your knowledge with these 40 questions.
Glycolysis Exam
Question 1/40
Exam Complete!
Here are your results, .
Your Score
38/40
95%
Heme Metabolism Pathway
Heme : Metabolism Pathway
Heme Metabolism: Biosynthesis
Heme is a vital molecule. It acts as a "prosthetic group" (a permanent helper) for proteins like Hemoglobin (oxygen transport), Myoglobin (oxygen storage), and Cytochromes (drug detoxification and electron transport).
1. Structure & Definitions
What is a Porphyrin?
Porphyrins are large, cyclic compounds made of 4 Pyrrole Rings linked together by methenyl bridges.
They are famous for binding metal ions.
Example: Magnesium in Chlorophyll (plants).
Example: Iron in Heme (humans).
The Side Chains
The properties of the porphyrin depend on which "decorations" (side chains) are attached to the rings:
- A: Acetate (Acetyl)
- P: Propionate (Propionyl)
- M: Methyl
- V: Vinyl
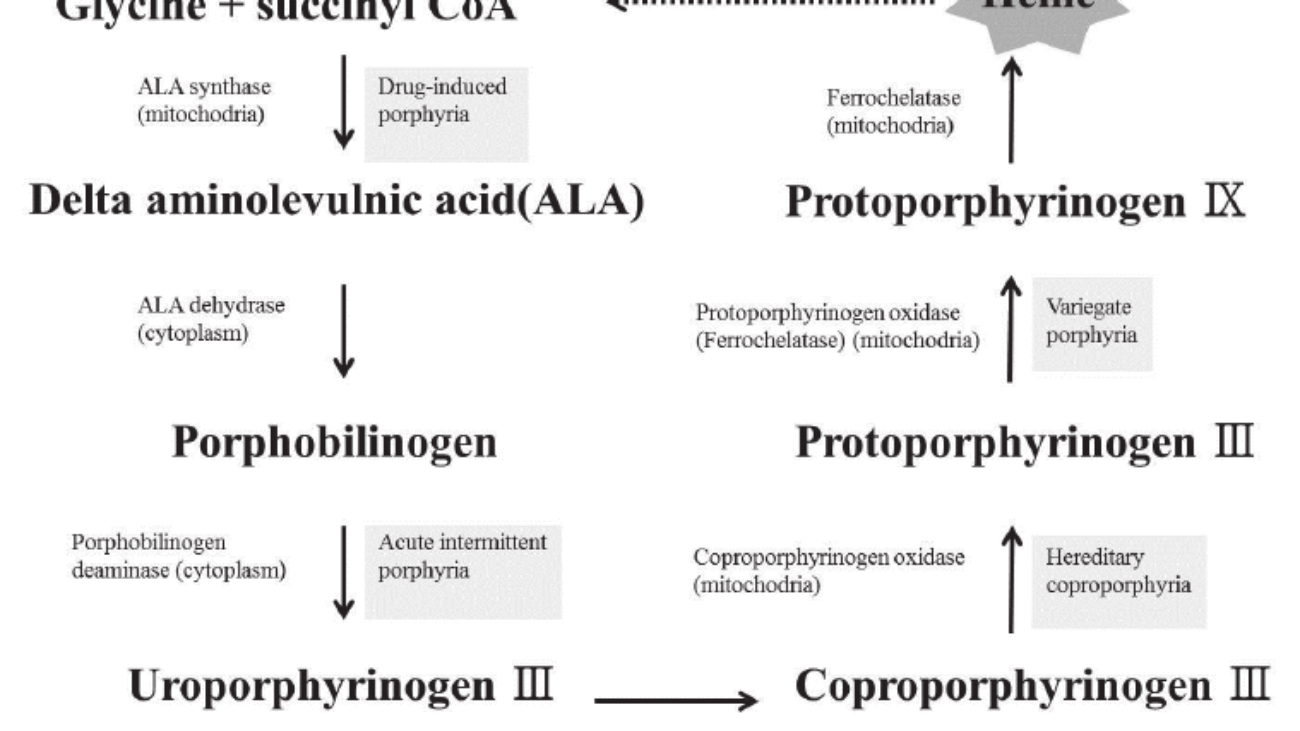
2. Steps of Heme Synthesis
This process is like a relay race. It starts in the Mitochondria, runs out to the Cytosol, and finishes back in the Mitochondria.
Step 1: Formation of ALA (The Rate-Limiting Step)
The Reaction:
Succinyl CoA (from TCA cycle) + Glycine → δ-Aminolevulinate (ALA) + CO₂
- Enzyme: ALA Synthase (ALAS).
- Coenzyme Required: Pyridoxal Phosphate (Vitamin B6).
- Significance: This is the Committed Step. Once this happens, the cell is committed to making Heme.
- ALAS-1: Found in the Liver (and all tissues).
- ALAS-2: Found in Bone Marrow (Erythroid cells).
Clinical Note: Mutation in ALAS-2 causes X-Linked Sideroblastic Anemia (Iron cannot be used, so it piles up).
Steps 2 to 5: Building the Ring in the Cytosol
Step 2: Formation of Porphobilinogen (PBG)
2 molecules of ALA condense to form 1 Ring (PBG).
- Enzyme: ALA Dehydratase (also called PBG Synthase).
- Requirement: This enzyme contains Zinc.
Result: ALA accumulates (Neurotoxic) causing brain damage and anemia.
Step 3: Formation of Hydroxymethylbilane (HMB)
4 molecules of PBG are linked together in a line (Linear Tetrapyrrole).
- Enzyme: HMB Synthase (PBG Deaminase).
Step 4: Ring Closure (Uroporphyrinogen III)
The linear chain is curled into a circle.
- Enzyme: Uroporphyrinogen III Synthase.
- Mechanism: It flips one of the rings to create an asymmetric "Type III" structure.
- Note: If this enzyme is missing, the ring closes spontaneously but incorrectly (Type I), which is useless to the body.
Step 5: Decarboxylation
Uroporphyrinogen III → Coproporphyrinogen III
- Enzyme: Uroporphyrinogen Decarboxylase.
- Action: Removes Carboxyl groups (CO₂). This makes the molecule less water-soluble (more hydrophobic) so it can re-enter the mitochondria.
Steps 6 to 9: The Final Touches
-
Step 6 & 7: Oxidation
Coproporphyrinogen III enters the mitochondria. It is oxidized to Protoporphyrinogen IX and then to Protoporphyrin IX.
Enzymes: Coproporphyrinogen Oxidase & Protoporphyrinogen Oxidase.
Key Detail: Step 8 creates double bonds, giving the molecule its red color. -
Step 9: Insertion of Iron (The Finale)
Protoporphyrin IX + Fe²⁺ (Ferrous) → HEME
- Enzyme: Ferrochelatase (Heme Synthase).
- Inhibitor: This enzyme is ALSO sensitive to Lead. Lead poisoning blocks the final insertion of iron.
3. Regulation of Heme Synthesis
The body carefully controls the first enzyme, ALA Synthase, to prevent overproduction.
Heme (the product) acts as a negative regulator.
- Repression: Heme stops the gene from making more ALA Synthase.
- Allosteric Inhibition: Hematin (Heme with Fe³⁺) binds directly to the enzyme to stop it.
Drugs like Barbiturates (sedatives) increase Heme synthesis.
- Barbiturates are metabolized by Cytochrome P450 in the liver.
- Cytochrome P450 contains Heme.
- Metabolizing the drug consumes the Heme.
- Free Heme levels drop.
- The "Brake" (Feedback Inhibition) is removed.
- ALA Synthase increases to replenish the lost Heme.
High concentrations of Glucose inhibit the induction of ALA Synthase.
Clinical Relevance: Giving glucose (IV sugar) is part of the treatment for acute attacks of Porphyria to try and slow down the pathway.
This is a Tuberculosis drug. It depletes Pyridoxal Phosphate (Vitamin B6).
Since Step 1 requires B6, INH can stop Heme synthesis and cause anemia.
Regulation of Heme Synthesis
The body must maintain a perfect balance of Heme.
Too Little: You get Anemia (no oxygen transport).
Too Much: Heme and its precursors are toxic to cells.
The main control switch is the very first enzyme: ALA Synthase (ALAS).
A. The Tale of Two Enzymes (ALAS1 vs. ALAS2)
Even though they do the same job, there are two different versions of this enzyme depending on where they live.
1. ALAS1 (The Housekeeper)
- Location: Found in All Tissues (Liver, etc.).
- Purpose: Makes heme for "Housekeeping" proteins like Cytochromes and Catalase.
- Regulation: Controlled by the amount of Heme present.
2. ALAS2 (The Specialist)
- Location: Found ONLY in Erythroid Cells (Red Blood Cell precursors in Bone Marrow).
- Purpose: Makes massive amounts of heme specifically for Hemoglobin.
- Regulation: Controlled by the amount of Iron present.
B. Regulation of ALAS1 (Liver)
The liver uses Negative Feedback Inhibition. Heme acts as the "Stop" signal. It attacks the enzyme at three different levels to shut it down.
What happens: High levels of "Free Heme" (heme not attached to proteins) travel to the nucleus.
The Effect: It tells the DNA to stop making the mRNA for ALAS1. This is the most important mechanism.
What happens: Heme makes the ALAS1 mRNA unstable.
The Effect: The mRNA is chopped up (degraded) before it can be used to build the enzyme.
Recall: ALAS1 is made in the Cytosol but must work in the Mitochondria.
The Effect: Heme blocks the door. It prevents the enzyme from entering the mitochondria. If it can't get in, it can't work.
C. Regulation of ALAS2 (Erythroid Cells)
Red blood cells don't care about free heme levels as much. They care about IRON. You cannot make Hemoglobin without Iron.
The IRE / IRP System
This acts like a physical switch on the mRNA.
- The Setup: The mRNA for ALAS2 has a special loop structure at the beginning (5' end) called the Iron-Responsive Element (IRE).
- Scenario A: Low Iron (Don't Build)
- A protein called IRP (Iron Regulatory Protein) sits on the loop (IRE).
- This acts like a roadblock. The ribosome cannot read the mRNA.
- Result: No ALAS2 is made. No Heme is made.
- Scenario B: High Iron (Build!)
- Iron binds to the IRP protein.
- This causes the IRP to fall off the mRNA.
- The roadblock is removed! Translation proceeds.
- Result: ALAS2 is made. Heme is produced to match the iron supply.
D. Other Factors Influencing Synthesis
Besides Heme and Iron, outside factors can speed up or slow down the process.
Mechanism: These drugs are metabolized by Cytochrome P450 (a heme protein).
The liver burns up its Heme supply to fight the drug. Low heme levels release the "brake" on ALAS1.
Result: Massive increase in Heme synthesis.
Mechanism: High glucose levels have a "calming" effect on ALAS1 (represses activity).
Clinical Use: We give IV Glucose (sugar) to patients having a Porphyria attack to stop the overproduction of toxic precursors.
Mechanism: Steroids (Estrogen, Androgens) induce ALAS1 synthesis.
This is why Porphyria attacks often happen during puberty or specific phases of the menstrual cycle.
Heme Degradation: The Disposal System
Making Heme is important, but getting rid of old Heme safely is just as critical. This process happens mainly in the Reticuloendothelial System (RES), specifically in the Spleen and Liver.
Phase 1: Breaking the Ring (Spleen)
- Globin (Protein): Broken down into Amino Acids and recycled.
- Iron (Fe): Removed and stored/recycled.
- Porphyrin Ring: This cannot be recycled. It must be degraded.
Step 1: Heme → Biliverdin (The Green Step)
- Substrate: Heme. (Note: The Iron must be oxidized from Fe²⁺ to Fe³⁺ first).
- Enzyme: Heme Oxygenase (HO).
- Action: It cuts the Porphyrin ring open at a specific bridge.
- The Products:
- Biliverdin: A linear tetrapyrrole with a GREEN color.
- Iron (Fe³⁺): Released for recycling.
- Carbon Monoxide (CO): This is the only time the body makes CO naturally. It acts as a signaling molecule.
- Significance: This is the Rate-Limiting Step of degradation.
Step 2: Biliverdin → Bilirubin (The Yellow Step)
- Substrate: Biliverdin (Green).
- Enzyme: Biliverdin Reductase.
- Requirement: Uses NADPH.
- Product: Bilirubin. This pigment is YELLOW-ORANGE.
Phase 2: Transport in the Blood
Step 3: The Albumin Taxi
The Bilirubin made in the spleen is called Unconjugated Bilirubin (UCB) or "Indirect Bilirubin."
UCB is Hydrophobic (Fat-soluble). It hates water. It cannot swim in the blood alone.
It binds tightly to Albumin (a protein in blood). Albumin acts as a "Taxi" to carry it to the liver.
Because Unconjugated Bilirubin is fat-soluble, if there is too much of it (and not enough Albumin), it can cross cell membranes. In babies, it can cross the Blood-Brain Barrier and deposit in the brain, causing permanent brain damage (Kernicterus).
Phase 3: Processing in the Liver
Step 4: Uptake
- The Albumin taxi drops Bilirubin off at the liver cell (hepatocyte).
- Carrier proteins (OATP) bring it inside.
- Ligandin: Inside the cell, it binds to Ligandin (or GST-B) so it doesn't slip back out.
Step 5: Conjugation (Making it Water-Soluble)
We need to make the bilirubin safe to excrete.
- Enzyme: UDP-glucuronosyltransferase (UGT1A1).
- Action: It attaches Glucuronic Acid molecules to the bilirubin.
- Product: Conjugated Bilirubin (Direct Bilirubin).
- Result: It is now Hydrophilic (Water-soluble). It can be mixed into bile.
Step 6: Secretion into Bile
- Transporter: MRP2 (Multidrug resistance-associated protein 2).
- Action: It pumps Conjugated Bilirubin against the gradient into the bile ducts.
- Significance: This is the Rate-Limiting Step for excretion. If this pump fails, Conjugated Bilirubin backs up into the blood.
Phase 4: The Intestine & Final Colors
Bile carries the Conjugated Bilirubin into the Intestine. Here, bacteria take over.
Gut bacteria remove the glucuronic acid (deconjugation) and convert bilirubin into Urobilinogen (Colorless).
Bacteria oxidize Urobilinogen into Stercobilin.
Color: BROWN
(This is why poop is brown).
Some is reabsorbed, goes to the kidney, and becomes Urobilin.
Color: YELLOW
(This is why pee is yellow).
Some is reabsorbed and goes back to the liver.
Enterohepatic Circulation
Visual Summary of Colors
Clinical Aspects: When Heme Metabolism Fails
We have learned how Heme is built and destroyed. Now we look at the diseases that happen when these processes break. We divide them into two main categories:
Defects in Synthesis.
Problem: Toxic precursors build up.
Defects in Degradation.
Problem: Bilirubin builds up.
Porphyrias: Disorders of Heme Synthesis
These are usually genetic (inherited). Depending on which enzyme is broken, different toxic chemicals accumulate. We classify them by their main symptoms: Nerve Pain (Acute) or Skin Blisters (Cutaneous).
1. Acute Intermittent Porphyria (AIP)
The "Nervous System" Porphyria
- Enzyme Defect: PBG Deaminase (Step 3).
- Accumulation: ALA and PBG.
- Symptoms (The 5 P's):
- Painful Abdomen (Severe, often confused for surgery).
- Polyneuropathy (Weakness, paralysis).
- Psychiatric (Anxiety, hallucinations).
- Port-wine Urine (Reddish-brown urine when standing).
- Precipitated by Drugs.
- Important: NO Skin Photosensitivity.
Triggers: Things that speed up Heme synthesis (Induce Cyt P450): Barbiturates, Alcohol, Sulfa drugs, Fasting/Dieting.
Treatment:
1. Stop the drug/alcohol.
2. IV Glucose (Sugar) or Hemin. (These inhibit ALAS1 to stop the production line).
2. Porphyria Cutanea Tarda (PCT)
The "Skin" Porphyria (Most Common)
- Enzyme Defect: Uroporphyrinogen Decarboxylase (Step 5).
- Accumulation: Uroporphyrinogen.
- Symptoms:
- Photosensitivity: The skin reacts to sunlight.
- Blisters: Fluid-filled bullae on hands/face.
- Hypertrichosis: Excessive hair growth.
- Tea-Colored Urine.
Triggered by: Chronic Alcoholism, Iron Overload, Hepatitis C.
Treatment:
1. Avoid Alcohol/Sun.
2. Phlebotomy: Drawing blood to reduce Iron levels.
3. Congenital Erythropoietic Porphyria (CEP)
Also known as Günther's Disease. This is extremely severe and rare.
- Defect: Uroporphyrinogen III Synthase.
- Symptoms: Severe mutilating skin blisters, Erythrodontia (Red/Brown teeth that glow under UV light), red urine.
- Treatment: Bone marrow transplant.
Jaundice: Disorders of Heme Degradation
Jaundice (Hyperbilirubinemia) is the yellowing of skin and eyes (sclera) when Bilirubin blood levels exceed 2–3 mg/dL. We classify it by where the traffic jam is.
| Type | The Problem | Bilirubin Type | Urine & Stool |
|---|---|---|---|
| 1. Pre-Hepatic (Hemolytic) |
Too much breakdown.
Hemolysis (Sickle cell, Malaria) produces bilirubin faster than the liver can handle. |
High Unconjugated (Indirect). |
Urine: Normal color (Unconjugated cannot enter urine).
Stool: Normal/Dark. |
| 2. Hepatic (Hepatocellular) |
Broken Factory.
Liver cells are damaged (Hepatitis, Alcohol) and cannot conjugate or excrete. |
High Mixed (Both).
Also high Liver Enzymes (ALT/AST). |
Urine: Dark (Conjugated leaks out).
Stool: Normal or Pale. |
| 3. Post-Hepatic (Obstructive) |
Blocked Pipe.
Gallstones or Cancer block the bile duct. Bile cannot leave. |
High Conjugated (Direct).
Also high ALP & GGT. |
Urine: Very Dark/Tea-colored (Bilirubinuria).
Stool: Pale/Clay (No stercobilin). Other: Pruritus (Itching). |
Genetic Disorders of Bilirubin
-
Gilbert's Syndrome:
Severity: Mild, Benign. Very common.
Cause: Enzyme works at 30% speed.
Trigger: Stress, Fasting. -
Crigler-Najjar Syndrome:
Severity: Severe/Fatal.
Cause: Enzyme is totally missing (Type I) or very low (Type II).
Risk: Kernicterus in babies.
-
Dubin-Johnson Syndrome:
Defect: MRP2 Transporter is broken. Liver can't pump bile out.
Sign: Black Liver (Pigment accumulation). Benign. -
Rotor Syndrome:
Defect: Similar to Dubin-Johnson but milder.
Sign: No black liver.
C. Neonatal Jaundice (Physiological)
Common in newborns (60%). Their liver machinery is immature.
- Fetal RBCs die quickly (Hemolysis).
- Liver UGT1A1 enzyme is slow (Immature).
- Gut flora is low (reabsorption increases).
Unconjugated Bilirubin is fat-soluble. It crosses the thin blood-brain barrier of the baby and deposits in the brain, causing permanent damage.
💡 Cure: PhototherapyBlue light converts bilirubin into a water-soluble shape (isomer) so the baby can pee it out without needing the liver.
Biochemistry: Heme Metabolism
Test your knowledge with these 40 questions.
Heme Metabolism Quiz
Question 1/40
Quiz Complete!
Here are your results, .
Your Score
38/40
95%
Nucleotide Metabolism Pathway
Nucleotide : Metabolism Pathway
Nucleotide Metabolism: Introduction & De Novo Purine Synthesis
To begin our journey, it is essential to clearly define and distinguish between nucleotides and nucleosides, understand their basic chemical structure, and appreciate their diverse and vital roles in biological systems.
I. Introduction to Nucleotides and Nucleosides
A. Definition and Components
1. Nucleoside
A molecule composed of two main parts:
- A Pentose Sugar: A 5-carbon sugar (either ribose or deoxyribose).
- A Nitrogenous Base: A heterocyclic ring structure containing nitrogen.
The Bond: The nitrogenous base is attached to the C1' carbon of the pentose sugar via a β-N-glycosidic bond.
2. Nucleotide
A Nucleotide is simply a Nucleoside with one or more Phosphate groups attached.
- Attachment: The phosphate group(s) are typically attached to the C5' carbon of the pentose sugar via an ester bond.
- Note: They can also be attached to the C3' carbon (less common, but important in RNA processing).
- • Monophosphate (NMP): One phosphate (e.g., AMP).
- • Diphosphate (NDP): Two phosphates (e.g., ADP).
- • Triphosphate (NTP): Three phosphates (e.g., ATP).
B. Pentose Sugars
The type of pentose sugar determines whether the nucleotide is for RNA or DNA.
- Found in Ribonucleosides and Ribonucleotides (RNA).
- Structure: It has a Hydroxyl (-OH) group at the C2' position.
- Found in Deoxyribonucleosides and Deoxyribonucleotides (DNA).
- Structure: It has a Hydrogen (-H) atom at the C2' position.
- Meaning: "Deoxy" literally means "lacking oxygen."
C. Nitrogenous Bases
These are cyclic, planar, relatively water-insoluble compounds that absorb UV light. They are categorized into two classes based on ring structure.
1. Purines (Double Ring)
Characterized by a double-ring structure (a six-membered pyrimidine ring fused to a five-membered imidazole ring).
The two major purine bases are:
- Adenine (A): Often designated with a single amino group.
- Guanine (G): Contains both an amino and a carbonyl group.
2. Pyrimidines (Single Ring)
Characterized by a single-ring structure (a six-membered heterocyclic ring).
The three major pyrimidine bases are:
- Cytosine (C): Contains an amino group.
- Thymine (T): Found only in DNA. Contains a methyl group at the C5 position.
- Uracil (U): Found only in RNA. Lacks the methyl group present in thymine.
D. Naming Conventions (Nomenclature)
| Base | Nucleoside (Ribose) | Nucleotide (Ribose-MP) | Nucleoside (Deoxyribose) | Nucleotide (Deoxyribose-MP) |
|---|---|---|---|---|
| Adenine (A) | Adenosine | Adenylate (AMP) | Deoxyadenosine | Deoxyadenylate (dAMP) |
| Guanine (G) | Guanosine | Guanylate (GMP) | Deoxyguanosine | Deoxyguanylate (dGMP) |
| Cytosine (C) | Cytidine | Cytidylate (CMP) | Deoxycytidine | Deoxycytidylate (dCMP) |
| Uracil (U) | Uridine | Uridylate (UMP) | - (rarely found in DNA) | - |
| Thymine (T) | Ribothymidine (rare) | Ribothymidylate (rTMP) | Deoxythymidine | Deoxythymidylate (dTMP) |
Note: For deoxyribonucleotides, the 'd' prefix is used (e.g., dATP, dGMP).
Note: Thymine is predominantly found in DNA. While "ribothymidine" exists, uracil is the primary pyrimidine in RNA.
E. Major Physiological Functions of Nucleotides
Nucleotides are far more than just building blocks for nucleic acids; they play incredibly diverse and crucial roles in almost every aspect of cellular life.
1. Building Blocks of Nucleic Acids
- DNA (Deoxyribonucleic Acid): Genetic material, stores and transmits hereditary information. dNTPs (dATP, dGTP, dCTP, dTTP) are polymerized to form DNA.
- RNA (Ribonucleic Acid): Involved in gene expression (mRNA, tRNA, rRNA), regulation, and catalysis. NTPs (ATP, GTP, CTP, UTP) are polymerized to form RNA.
2. Energy Currency of the Cell
- ATP (Adenosine Triphosphate): The primary energy-carrying molecule. Hydrolysis of its high-energy phosphate bonds releases energy to drive various cellular processes (muscle contraction, active transport, biosynthesis).
- GTP (Guanosine Triphosphate): Also an important energy source, particularly in protein synthesis (translation) and signal transduction.
3. Components of Coenzymes
Many essential coenzymes, critical for enzymatic reactions, are derivatives of nucleotides:
- NAD+ (Nicotinamide Adenine Dinucleotide): Derived from ATP. Involved in redox reactions (electron carrier).
- FAD (Flavin Adenine Dinucleotide): Derived from ATP. Involved in redox reactions.
- Coenzyme A (CoA): Derived from ATP. Involved in acyl group transfer reactions (e.g., fatty acid metabolism, TCA cycle).
4. Regulatory Molecules and Signal Transduction
- cAMP (cyclic Adenosine Monophosphate): A ubiquitous second messenger in signal transduction pathways, mediating the effects of many hormones (e.g., adrenaline).
- cGMP (cyclic Guanosine Monophosphate): Another important second messenger, involved in processes like vasodilation and vision.
- ADP, AMP: Allosteric regulators of many enzymes (e.g., in glycolysis, gluconeogenesis).
5. Activated Intermediates in Biosynthesis
- UDP-Glucose: Involved in glycogen synthesis.
- CDP-Diacylglycerol: Involved in lipid synthesis.
- S-Adenosylmethionine (SAM): A methyl group donor in numerous methylation reactions (not strictly a nucleotide but derived from ATP and methionine).
II. De Novo Synthesis of Purine Nucleotides
"De novo" means "from scratch," and indeed, the purine ring is constructed from small, simpler precursors in this pathway. This process primarily occurs in the liver, but also in other rapidly dividing cells.
A. Overall Pathway: Building the Purine Ring on PRPP
Unlike pyrimidine synthesis where the base is formed first and then attached to the sugar, purine synthesis begins with the sugar and builds the ring directly upon it.
α-D-Ribose-5-Phosphate (a product of the Pentose Phosphate Pathway).
- Ribose-5-phosphate is converted to 5-Phosphoribosyl-1-Pyrophosphate (PRPP).
- Enzyme: PRPP Synthetase (Ribose Phosphate Pyrophosphokinase).
- Energy Cost: ATP is consumed, and pyrophosphate (PPi) is released.
- Significance: PRPP is an activated pentose sugar that is a key precursor not only for purine synthesis but also for pyrimidine synthesis, NAD+ synthesis, and salvage pathways.
- The pyrophosphate group of PRPP is replaced by an amino group, forming 5-Phosphoribosyl-1-amine.
- Enzyme: Glutamine:PRPP Amidotransferase (this is the rate-limiting and committed step of purine synthesis).
- Nitrogen Source: The amino group comes from the amide nitrogen of Glutamine.
- Regulation: This enzyme is highly regulated (feedback inhibited by AMP, GMP, and IMP).
The purine ring (specifically the imidazole ring, followed by the pyrimidine ring) is built in a series of ten steps, consuming energy (ATP) and incorporating atoms from various small molecules.
Note: The intermediate after 5-phosphoribosyl-1-amine is called Glycinamide Ribonucleotide (GAR), as glycine is incorporated early on.
- The end product of this complex ten-step pathway is Inosine Monophosphate (IMP).
- IMP contains the complete purine ring structure. It is often referred to as hypoxanthine ribonucleotide.
B. Precursors for the Purine Ring Atoms
The atoms that make up the purine ring come from surprisingly diverse and simple sources. It is helpful to visualize the purine ring and where each atom originates:
- N1: From the amino group of Aspartate.
- C2: From N10-Formyl-Tetrahydrofolate (a folate derivative).
- N3: From the amide group of Glutamine.
- C4, C5, N7: From Glycine (the entire molecule of glycine provides these three atoms).
- C6: From CO₂ (bicarbonate).
- N9: From the amide group of Glutamine.
- C8: From N10-Formyl-Tetrahydrofolate (a folate derivative).
- Two Glutamines
- One Aspartate
- One Glycine
- One CO₂
- Two N10-Formyl-THF (tetrahydrofolate derivatives)
C. Formation of IMP as the Common Precursor
The series of reactions from 5-Phosphoribosyl-1-amine to IMP involves:
- Multiple steps of ATP hydrolysis: Providing the energy for the synthetic reactions.
- Two steps requiring N10-formyl-tetrahydrofolate: Donating single carbon units for the formation of C2 and C8 of the purine ring.
Clinical Relevance: This makes the pathway a target for folate antagonists in cancer chemotherapy (e.g., methotrexate). - Several enzyme-catalyzed reactions: Building up the ring structure sequentially.
D. Conversion of IMP to AMP and GMP
Once IMP is formed, it serves as a branch point for the synthesis of the two major purine ribonucleotides: Adenosine Monophosphate (AMP) and Guanosine Monophosphate (GMP). These two pathways are reciprocally regulated to ensure balanced production.
Synthesis of AMP from IMP
-
Step 1:
IMP is converted to Adenylosuccinate.
- Enzyme: Adenylosuccinate Synthetase.
- Energy Input: GTP is used (hydrolyzed to GDP + Pi). This is a crucial regulatory point: the synthesis of AMP requires GTP, linking the two purine pathways.
- Nitrogen Source: Aspartate is incorporated.
-
Step 2:
Adenylosuccinate is cleaved to AMP and Fumarate.
- Enzyme: Adenylosuccinase.
Synthesis of GMP from IMP
-
Step 1:
IMP is converted to Xanthosine Monophosphate (XMP).
- Enzyme: IMP Dehydrogenase.
- Redox Reaction: NAD+ is reduced to NADH.
-
Step 2:
XMP is converted to GMP.
- Enzyme: GMP Synthetase.
- Energy Input: ATP is used (hydrolyzed to AMP + PPi). This is another crucial regulatory point: the synthesis of GMP requires ATP.
- Nitrogen Source: Glutamine is incorporated.
E. Regulation of IMP, AMP, and GMP Synthesis
The synthesis of purine nucleotides is tightly regulated to match the cell's needs and to maintain a balanced pool of ATP and GTP.
Inhibited by both purine nucleotides (AMP, GMP) and pyrimidine nucleotides.
- Feedback Inhibited by: AMP, GMP, and IMP (the end products of the pathway).
- Activated by: PRPP (substrate availability).
- AMP Synthesis: Adenylosuccinate Synthetase is inhibited by AMP. Its activity is dependent on GTP (linking AMP synthesis to the availability of GMP).
- GMP Synthesis: IMP Dehydrogenase is inhibited by GMP. Its activity is dependent on ATP (linking GMP synthesis to the availability of AMP).
III. De Novo Synthesis of Pyrimidine Nucleotides
We just learned how to make Purines (the double ring). Now, we look at Pyrimidines (the single ring: C, T, and U).
Location: Like Purines, this happens in the Cytoplasm (fluid) of the cell. It is very active in the liver.
A. The Strategy: "Ring First, Sugar Later"
This is the opposite of Purine synthesis.
- Purines: We built the ring directly on top of the sugar (PRPP).
- Pyrimidines: We build the Ring FIRST, and then we attach it to the sugar.
B. The Ingredients (Precursors)
The Pyrimidine ring is simpler. It comes from just 3 sources:
This amino acid provides the bulk of the ring: N1, C4, C5, and C6.
- Glutamine: Provides Nitrogen N3 (Amide group).
- CO₂: Provides Carbon C2.
C. The 6-Step Pathway to UMP
The goal is to make UMP (Uridine Monophosphate). Once we have UMP, we can make all the others.
Step 1: The Committed Step (Rate-Limiting)
Glutamine + CO₂ + 2 ATP → Carbamoyl Phosphate
- Enzyme: Carbamoyl Phosphate Synthetase II (CPS-II).
- Location: Cytosol.
- CPS-I: Mitochondria, uses Ammonia, for Urea.
- CPS-II: Cytosol, uses Glutamine, for Pyrimidines.
Step 2: Formation of Carbamoyl Aspartate
Carbamoyl Phosphate + Aspartate → Carbamoyl Aspartate
Enzyme: Aspartate Transcarbamoylase (ATCase).
This step fuses the pieces together to start the ring.
Step 3: Ring Closure
Loss of water closes the ring to form Dihydroorotate.
Enzyme: Dihydroorotase.
Note: In humans, enzymes 1, 2, and 3 are combined in one big protein called "CAD".
Step 4: Oxidation (The Odd One Out)
Dihydroorotate → Orotate.
Enzyme: Dihydroorotate Dehydrogenase.
⚠️ Important Location Exception:
This is the ONLY enzyme in the pathway located on the Inner Mitochondrial Membrane. All others are in the cytosol. It uses FAD to pass electrons to the electron transport chain.
Step 5: Attachment to Sugar
Orotate + PRPP → Orotidine Monophosphate (OMP).
Enzyme: Orotate Phosphoribosyltransferase (OPRT).
This is the moment the Ring meets the Sugar (PRPP).
Step 6: Decarboxylation
OMP loses CO₂ → Uridine Monophosphate (UMP).
Enzyme: OMP Decarboxylase.
Goal Achieved! We have the first Pyrimidine Nucleotide.
D. Making Other Nucleotides (CTP, dUDP, dTMP)
We have UMP, but we need C, T, and the DNA versions ("d").
We take UTP and add an amino group.
- Reaction: UTP → CTP.
- Enzyme: CTP Synthetase.
- Donor: Glutamine provides the nitrogen. ATP provides energy.
We must remove the oxygen from the Ribose sugar.
- Enzyme: Ribonucleotide Reductase.
- Action: Reduces the OH group at Carbon-2' to just H.
- Requirement: Thioredoxin and NADPH.
3. Making dTMP (Thymine) - Clinical "Hot Spot"
DNA needs Thymine (T), not Uracil (U). We must convert dUMP to dTMP.
The Reaction:
dUMP + Methylene-Tetrahydrofolate → dTMP.
The Enzyme:
Thymidylate Synthase
Cancer cells divide fast and need lots of DNA (lots of Thymine). We can kill cancer by stopping this enzyme.
- 5-Fluorouracil (5-FU): A drug that directly blocks Thymidylate Synthase.
- Methotrexate: A drug that blocks the recycling of the Folate needed for this reaction.
E. Regulation: Controlling the Speed
| Enzyme | Activators (Go!) | Inhibitors (Stop!) |
|---|---|---|
| CPS-II (Step 1) | PRPP, ATP | UTP, CTP (The Products) |
| Ribonucleotide Reductase | Complex regulation to ensure a perfect balance of all 4 DNA blocks (dATP, dGTP, dCTP, dTTP). | |
V. Salvage Pathways for Nucleotides
Concept: "De Novo" synthesis is like cooking a meal from scratch (expensive). "Salvage" is like eating leftovers (cheap and efficient).
A. Why Salvage?
- Energy Saving: De novo synthesis costs 6-7 ATP. Salvage costs only 1 ATP.
- Vital Tissues: The Brain and Red Blood Cells (RBCs) cannot make purines from scratch. They must use salvage pathways to survive.
- Rapid Growth: Bone marrow and immune cells (lymphoid) need so much DNA they use both methods.
B. How Salvage Works
We take a free Base (Adenine, Guanine, etc.) and re-attach it to a sugar (PRPP).
C. Purine Salvage Enzymes
Adenine + PRPP → AMP.
Deficiency: Causes kidney stones (2,8-Dihydroxyadenine stones).
This enzyme does double duty:
- Hypoxanthine + PRPP → IMP
- Guanine + PRPP → GMP
🚑 Clinical Alert: Lesch-Nyhan Syndrome
Cause: Total deficiency of HGPRT.
If HGPRT is missing, the body cannot recycle Purines.
- Waste Buildup: Hypoxanthine and Guanine are degraded into massive amounts of Uric Acid (Hyperuricemia).
- Symptoms: Severe Gout (painful joints), kidney stones.
- Neurological: Severe intellectual disability and Self-Mutilation (biting off lips and fingers).
D. Pyrimidine Salvage Enzymes
This is less critical clinically, but still important.
- UPRT: Salvages Uracil → UMP.
- Thymidine Kinase (TK): Salvages Deoxythymidine → dTMP.
Note: This enzyme is very active in rapidly dividing cells. - Deoxycytidine Kinase (dCK): Salvages Deoxycytidine → dCMP.
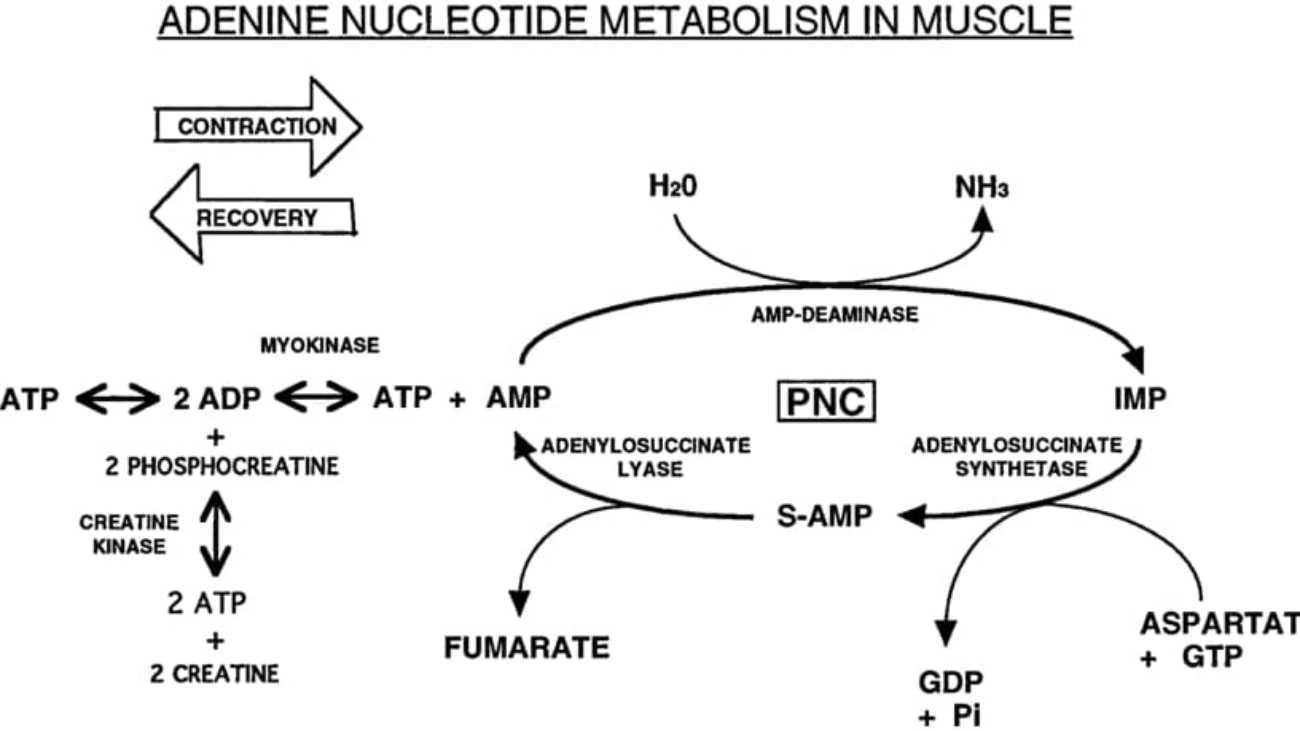
VI. Degradation of Purine Nucleotides
What happens to old DNA and RNA? The body must break them down safely.
For Purines (A and G), this process is critical because the final waste product is Uric Acid, which can cause disease if it builds up.
A. The General Strategy
The degradation involves three main phases:
- Dephosphorylation: Removing the phosphate groups (Triphosphate → Monophosphate → Nucleoside).
- Deamination: Removing the Nitrogen (Amino group).
- Oxidation: Turning the remaining ring into Uric Acid.
B. Degradation of AMP (Adenine)
AMP needs to be stripped down to Hypoxanthine.
AMP + H₂O → Adenosine + Pi
Enzyme: 5'-Nucleotidase.
(Alternate path in muscle: AMP Deaminase can turn AMP directly into IMP).
Adenosine + H₂O → Inosine + NH₃
Enzyme: Adenosine Deaminase (ADA)
Inosine + Pi → Hypoxanthine + Ribose-1-P
Enzyme: Purine Nucleoside Phosphorylase (PNP).
C. Degradation of GMP (Guanine)
GMP is stripped down to Xanthine.
- Step 1: GMP → Guanosine (Enzyme: 5'-Nucleotidase).
- Step 2: Guanosine → Guanine (Enzyme: PNP).
- Step 3: Guanine → Xanthine (Enzyme: Guanine Deaminase/Guanase).
D. The Common Pathway to Uric Acid
Both Hypoxanthine (from AMP) and Xanthine (from GMP) meet here. The goal is Oxidation.
The drug Allopurinol (used for Gout) works by inhibiting Xanthine Oxidase. This stops the production of Uric Acid.
E. Characteristics of Uric Acid
- Solubility: It is poorly soluble in water. It likes to turn into crystals (sodium urate).
- Excretion: We pee it out via the kidneys.
- The Danger: If levels get too high (Hyperuricemia), crystals form in joints (Gout) or kidneys (Stones).
- The Good Side: It is actually a strong antioxidant!
VII. Degradation of Pyrimidine Nucleotides
Unlike Purines, Pyrimidine degradation is "clean." The products are water-soluble.
A. The Products
The final products are simple molecules that dissolve easily:
1. Cytosine & Uracil Degradation
They share a pathway. Cytosine is converted to Uracil first.
- Step 1: CMP → UMP (Enzyme: Cytidine Deaminase).
- Step 2: UMP → Uracil.
- Step 3: Ring Opening by DPD (Dihydropyrimidine Dehydrogenase).
- End Product: β-Alanine (Used for Carnosine).
2. Thymine Degradation
Thymine (DNA only) has a methyl group, so its product is slightly different.
- Step 1: dTMP → Thymine.
- Step 2: Ring Opening by DPD.
- End Product: β-Aminoisobutyrate (Excreted in urine).
D. Clinical Relevance: DPD Deficiency
Dihydropyrimidine Dehydrogenase (DPD) is the rate-limiting enzyme for breaking down pyrimidines.
Patients with cancer are often given the drug 5-Fluorouracil (5-FU). This drug mimics Uracil.
The Danger: If a patient has a genetic DPD Deficiency, they cannot break down the drug. The drug builds up to toxic levels, causing death or severe side effects (neurotoxicity, bone marrow failure).
Note: Unlike Purines (Gout), there are no "accumulation diseases" for natural pyrimidines because they are water-soluble.
VII. Regulation of Nucleotide Metabolism
The body must balance these pools perfectly. Too little DNA means cells can't divide. Too much wastes energy.
This section explains the "Traffic Lights" (Regulation) and what happens when the traffic lights break (Disease).
A. General Regulatory Themes
-
🛑
Feedback Inhibition: The product (e.g., AMP) stops its own factory (Enzyme 1).
-
🔄
Reciprocal Regulation: "I'll scratch your back if you scratch mine." Making AMP requires GTP. Making GMP requires ATP. This ensures balance.
-
⚖️
Feed-forward Activation: If ingredients pile up (e.g., PRPP), they push the enzymes to work faster.
B. Regulation of Purine Synthesis
We control the flow at 3 main checkpoints.
- Go: Phosphate (Pi)
- Stop: Any Nucleotide (AMP, GMP, IMP)
Enzyme: Glutamine:PRPP Amidotransferase
- Go: High PRPP
- Stop: AMP, GMP, IMP
- Making AMP: Inhibited by AMP. Needs GTP.
- Making GMP: Inhibited by GMP. Needs ATP.
C. Regulation of Pyrimidine Synthesis
This enzyme makes ALL DNA building blocks (dATP, dGTP, dCTP, dTTP). Its regulation is complex.
- Global On/Off Switch:
ON = ATP (High energy = replicate DNA).
OFF = dATP (Too much DNA precursor = stop). - Fine Tuning: Different dNTPs bind to "Specificity Sites" to ensure the cell doesn't make too much of just one letter (e.g., dGTP stimulates making ADP).
VIII. Clinical Disorders & Pharmacology
1. Gout (Hyperuricemia)
What is it? High Uric Acid leads to sharp crystals depositing in joints (painful arthritis) and kidneys (stones).
- Underexcretion (90%): Kidneys fail to pee it out.
- Overproduction (10%):
- PRPP Synthetase Overactivity.
- High Cell Turnover (Cancer/Chemo).
- Partial HGPRT deficiency.
- Allopurinol / Febuxostat: Inhibits Xanthine Oxidase. Stops Uric Acid production.
- Probenecid: Helps kidneys excrete it.
- Colchicine/NSAIDs: For pain/inflammation.
2. Lesch-Nyhan Syndrome
X-Linked Recessive
Defect: Near total absence of HGPRT (Salvage Enzyme).
- Severe Hyperuricemia: Since purines cannot be salvaged, they are ALL degraded to Uric Acid (Severe Gout in children).
- Neurological (The Hallmark): Spasticity, Mental Retardation, and Compulsive Self-Mutilation (biting lips/fingers).
3. SCID (Bubble Boy Disease)
Adenosine Deaminase (ADA) Deficiency
- Mechanism: Without ADA, Adenosine accumulates. This turns into dATP.
- The Toxic Effect: High dATP turns OFF Ribonucleotide Reductase.
- Result: Cells cannot make DNA. Immune cells (B and T lymphocytes) cannot divide.
- Outcome: Severe Immunodeficiency (Fatal without bone marrow transplant or enzyme therapy).
4. Orotic Aciduria
Defect: Failure of UMP Synthase (OPRT + OMP Decarboxylase).
- Symptoms: Anemia (Megaoloblastic), Growth Retardation.
- Key Sign: Crystals of Orotic Acid in urine.
- Treatment: Oral Uridine. (It bypasses the block and inhibits CPS-II to stop Orotic Acid production).
Pharmacology: Targeting Nucleotides (Chemotherapy)
Cancer cells need nucleotides to grow. We use drugs to starve them.
Inhibits Dihydrofolate Reductase (DHFR). Prevents regeneration of THF (Folate). Stops Thymine and Purine synthesis.
"Suicide Inhibitor" of Thymidylate Synthase. Directly stops DNA from getting Thymine.
Inhibits Ribonucleotide Reductase. Stops conversion of RNA → DNA.
Inhibits De Novo Purine Synthesis (PRPP Amidotransferase).
IX. Additional Clinical & Pharmacological Notes
To complete our study of nucleotides, we must look at a few specific drugs and environmental factors that affect these pathways.
1. Mycophenolic Acid (Transplant Drug)
This is a powerful immunosuppressant drug used to prevent **Graft Rejection** (e.g., after a kidney transplant).
- It acts as a reversible, uncompetitive inhibitor of the enzyme IMP Dehydrogenase.
- Recall: IMP Dehydrogenase is needed to make GMP (Guanine) from IMP.
- The Result: It deprives rapidly dividing T-cells and B-cells of the Nucleic Acids they need to multiply. Without these immune cells, the body cannot attack the transplanted organ.
2. Sulfonamides (Sulfa Drugs)
These are antibiotics. They target bacteria by starving them of Nucleotides.
Bacteria must make their own Folic Acid (Folate) from scratch using a molecule called PABA (Para-aminobenzoic acid).
Sulfonamides look exactly like PABA (Structural Analogs). The bacteria try to use the drug instead of PABA, and their Folic Acid synthesis fails.
Humans cannot make Folic Acid. We must eat it in our diet. Therefore, Sulfa drugs kill bacteria but leave human purine synthesis alone.
3. Lead Poisoning & Gout ("Saturnine Gout")
Historically, Gout was often associated with "High Living" and alcohol. However, there is an environmental link.
- The Cause: In previous centuries, alcohol (especially port wine and moonshine) was often contaminated with Lead during storage or manufacturing.
- The Mechanism: Lead damages the kidney tubules.
- The Result: The damaged kidneys cannot excrete Uric Acid. The Uric Acid builds up, causing Secondary Gout.
4. Dietary Treatment for Orotic Aciduria
We learned that Orotic Aciduria causes Anemia because the body cannot make Pyrimidines (DNA).
Feeding a diet rich in Uridine results in:
- Improvement of Anemia: Uridine can be salvaged to make UMP, bypassing the broken enzyme block. This allows red blood cells to divide again.
- Decreased Orotate Excretion: The Uridine converts to UTP, which feedback-inhibits the first enzyme (CPS-II), stopping the production of the accumulated Orotic Acid.
Biochemistry: Nucleotide Metabolism
Test your knowledge with these 40 questions.
Nucleotide Metabolism Quiz
Question 1/40
Quiz Complete!
Here are your results, .
Your Score
38/40
95%
Amino Acids Metabolism Pathway
Amino Acids : Metabolism Pathway
Amino Acids & Protein Digestion/Absorption
Amino acids are the building blocks of proteins and play a central role in numerous metabolic pathways. Unlike carbohydrates and fats, the body has no dedicated storage form for amino acids. Instead, there's a dynamic "amino acid pool" that constantly receives and donates amino acids for various purposes.
The General Fates of Amino Acids
Once available in the body (either from diet, protein turnover, or de novo synthesis), amino acids follow several major metabolic pathways:
- Protein Synthesis (Anabolism): This is the primary and most vital role of amino acids. They are precisely assembled into new proteins (structural, enzymatic, hormonal, transport, etc.) within cells according to genetic instructions. This process is continuous, as proteins have finite lifespans and are constantly being synthesized and degraded (protein turnover).
-
Synthesis of Non-Protein Nitrogenous Compounds: Amino acids are precursors for a vast array of other essential nitrogen-containing molecules that are not proteins. These include:
- Neurotransmitters: e.g., dopamine, serotonin, GABA
- Hormones: e.g., thyroid hormones, adrenaline (epinephrine)
- Nucleotides: Components of DNA and RNA
- Heme: The iron-containing component of hemoglobin
- Creatine: Involved in energy storage in muscles
- Polyamines: Involved in cell growth and differentiation
-
Catabolism (Breakdown for Energy or Other Metabolites): When amino acids are in excess, or when energy stores (carbohydrates and fats) are insufficient, amino acids can be catabolized. This involves:
- Removal of the Amino Group: The nitrogen-containing amino group is removed (primarily as ammonia), which is then typically converted to urea for excretion.
- Metabolism of the Carbon Skeleton: The remaining carbon skeleton (α-keto acid) can be:
- Oxidized directly for energy (e.g., to Acetyl-CoA, TCA cycle intermediates).
- Converted into glucose (via gluconeogenesis).
- Converted into ketone bodies (via ketogenesis).
- Converted into fatty acids for storage.
Protein Digestion and Absorption
The body acquires amino acids primarily from the diet through the breakdown of ingested proteins. This process occurs in several stages:
In the Stomach:
- Denaturation: Dietary proteins first encounter the highly acidic environment of the stomach (pH 1.5-3.5) due to hydrochloric acid (HCl) secreted by parietal cells. This low pH causes proteins to denature, unfolding their complex three-dimensional structures and making them more accessible to enzymatic degradation.
- Pepsin Activity: Chief cells in the stomach secrete pepsinogen, a zymogen (inactive enzyme precursor). HCl cleaves pepsinogen to its active form, pepsin. Pepsin is an endopeptidase, meaning it hydrolyzes peptide bonds within the protein chain, preferentially cleaving bonds involving aromatic amino acids. This produces a mixture of smaller polypeptides and some oligopeptides.
In the Small Intestine (Duodenum):
- Neutralization: As the acidic chyme (partially digested food) moves from the stomach into the duodenum, its acidity stimulates the release of secretin and cholecystokinin (CCK). Secretin stimulates the pancreas to release bicarbonate, which neutralizes the stomach acid, raising the pH to around 7. This optimal pH is crucial for the activity of pancreatic proteases.
- Pancreatic Proteases: The pancreas secretes a cocktail of zymogens, including:
- Trypsinogen: Activated by enteropeptidase (also called enterokinase), an enzyme on the intestinal brush border, to form trypsin. Trypsin is a key enzyme because it then activates all other pancreatic zymogens.
- Chymotrypsinogen: Activated by trypsin to form chymotrypsin.
- Proelastase: Activated by trypsin to form elastase.
- Procarboxypeptidases A and B: Activated by trypsin to form carboxypeptidases A and B.
- Endopeptidases (Trypsin, Chymotrypsin, Elastase): These enzymes continue to hydrolyze internal peptide bonds within the polypeptides, breaking them down into smaller oligopeptides and tri- and di-peptides. Trypsin preferentially cleaves at basic amino acids (lysine, arginine), while chymotrypsin prefers aromatic amino acids (phenylalanine, tyrosine, tryptophan).
- Exopeptidases (Carboxypeptidases A and B): These enzymes remove amino acids one by one from the carboxyl (C-terminal) end of the polypeptide chains, producing free amino acids.
At the Intestinal Brush Border and Within Enterocytes:
- Brush Border Peptidases: The surface of the enterocytes (intestinal absorptive cells) contains various aminopeptidases and dipeptidases. Aminopeptidases cleave amino acids from the amino (N-terminal) end of oligopeptides. Dipeptidases and tripeptidases hydrolyze di- and tripeptides into free amino acids.
- Absorption into Enterocytes:
- Free Amino Acids: Absorbed by specific Na⁺-dependent co-transporters on the apical membrane (lumen side) of enterocytes. Different transporters exist for different classes of amino acids (e.g., neutral, basic, acidic).
- Di- and Tri-peptides: A significant portion of di- and tri-peptides are absorbed intact into the enterocytes via a separate proton-dependent cotransporter (PepT1).
- Intracellular Hydrolysis: Once inside the enterocyte, most absorbed di- and tri-peptides are further hydrolyzed into free amino acids by intracellular peptidases.
- Exit into Bloodstream: The free amino acids are then transported across the basolateral membrane (facing the bloodstream) into the portal circulation, primarily via facilitated diffusion and other transporters, and delivered to the liver.
Summary of Digestion Products for Absorption: The ultimate goal of protein digestion is to convert dietary proteins into free amino acids (the primary form absorbed into the blood), and to a lesser extent, di- and tri-peptides which are then broken down intracellularly.
Amino Acids & Amino Acid Pool/Nitrogen Balance
Differentiate Between Essential and Non-Essential Amino Acids
Amino acids are classified based on the human body's ability to synthesize them de novo (from scratch) or not. This classification is crucial for understanding nutritional requirements and metabolic pathways.
Essential Amino Acids (EAAs):
- Definition: These are amino acids that cannot be synthesized by the human body at all, or cannot be synthesized in sufficient quantities to meet physiological needs. Therefore, they must be obtained from the diet.
- Reason for Essentiality: The human body lacks the necessary enzymatic pathways to synthesize their carbon skeletons from simpler precursors, or it cannot synthesize them fast enough.
- List of Essential Amino Acids (PVT TIM HALL):
- Phenylalanine
- Valine
- Threonine
- Tryptophan
- Isoleucine
- Methionine
- Histidine (often considered essential, especially for infants and during growth, but some texts list it as semi-essential)
- Arginine (semi-essential; the body can synthesize it, but not always enough to meet the demands of rapid growth, especially in infants)
- Leucine
- Lysine
- Dietary Sources: Found in protein-rich foods, particularly "complete proteins" like meat, fish, eggs, dairy, soy, and quinoa, which contain all essential amino acids in adequate proportions.
Non-Essential Amino Acids (NEAAs):
- Definition: These are amino acids that the human body can synthesize de novo from intermediates of central metabolic pathways (like glycolysis, TCA cycle, and pentose phosphate pathway) or from other amino acids. They do not strictly need to be consumed in the diet.
- Reason for Non-Essentiality: The body possesses the necessary enzymatic machinery to synthesize their carbon skeletons and incorporate nitrogen.
- List of Non-Essential Amino Acids: Alanine, Asparagine, Aspartate, Cysteine, Glutamate, Glutamine, Glycine, Proline, Serine, Tyrosine.
- Conditional Essentiality: Some non-essential amino acids can become "conditionally essential" during specific physiological states or diseases. For example:
- Tyrosine becomes essential if dietary phenylalanine is insufficient or if the enzyme converting phenylalanine to tyrosine is deficient (e.g., in PKU).
- Cysteine becomes essential if dietary methionine is insufficient.
- Arginine and Glutamine can become conditionally essential during periods of rapid growth, severe illness, trauma, or stress.
Describe Amino Acid Pool and Nitrogen Balance
These concepts are fundamental to understanding the dynamic state of amino acid metabolism in the body.
The Amino Acid Pool:
- Concept: The "amino acid pool" refers to the total circulating and intracellular free amino acids available in the body at any given time. It's not a physical storage organ, but rather a conceptual reservoir.
- Sources of Amino Acids for the Pool:
- Dietary Protein Breakdown: Digestion and absorption of proteins from food.
- Tissue Protein Degradation (Protein Turnover): Continuous breakdown of existing body proteins.
- De Novo Synthesis: Synthesis of non-essential amino acids.
- Uses of Amino Acids from the Pool:
- Protein Synthesis: Rebuilding and repairing body proteins.
- Synthesis of Non-Protein Nitrogenous Compounds: As discussed earlier (nucleotides, hormones, neurotransmitters, etc.).
- Energy Production/Conversion: Catabolism of amino acids.
- Dynamic Equilibrium: The amino acid pool is in a constant state of flux, with amino acids continuously entering and leaving.
Nitrogen Balance:
- Concept: Nitrogen balance is a measure of the total nitrogen intake versus the total nitrogen excretion. It's used as a proxy for protein metabolism.
- Nitrogen Intake: Primarily from dietary protein. (Protein intake (g) / 6.25 = Nitrogen intake (g)).
- Nitrogen Excretion: Primarily as urea in urine, but also as ammonia, creatinine, uric acid, and small amounts in feces, sweat, and skin cells.
- Nitrogen Equilibrium (Zero Nitrogen Balance):
- Definition: Nitrogen intake equals nitrogen excretion.
- Physiological State: Healthy adults maintaining their body weight and muscle mass.
- Example: A non-growing adult consuming adequate protein.
- Positive Nitrogen Balance:
- Definition: Nitrogen intake is greater than nitrogen excretion. This indicates net protein synthesis and tissue growth.
- Physiological States: Growth (infants, children, adolescents), Pregnancy, Convalescence (recovery from illness), Bodybuilding.
- Example: A growing child who consumes enough protein for new tissue formation.
- Negative Nitrogen Balance:
- Definition: Nitrogen excretion is greater than nitrogen intake. This indicates net protein loss and tissue wasting.
- Physiological States: Inadequate Protein Intake (starvation), Severe Illness/Injury/Trauma (burns, infections), Cancer, Sepsis, Lack of Essential Amino Acids.
- Example: A patient with severe burns, where muscle protein is being broken down to provide amino acids for tissue repair and energy.
States of Nitrogen Balance:
General Reactions of Amino Acid Catabolism
When amino acids are in excess, or when the body needs to convert their carbon skeletons into other molecules, they undergo a series of catabolic reactions. The first and most critical step is the removal of the α-amino group, as this nitrogen cannot be stored and must be detoxified and excreted.
Transamination: Transfer of the Amino Group
- Definition: Transamination is the most common and initial step in the catabolism of most amino acids. It involves the transfer of an α-amino group from an amino acid to an α-keto acid. This reaction is reversible.
- Enzymes: Catalyzed by aminotransferases (also known as transaminases), such as Alanine Aminotransferase (ALT) and Aspartate Aminotransferase (AST).
- General Reaction:
Amino Acid 1 + α-Keto Acid 2 ⇌ α-Keto Acid 1 + Amino Acid 2 - Example:
Alanine + α-Ketoglutarate ⇌ Pyruvate + Glutamate - Coenzyme: All aminotransferases require pyridoxal phosphate (PLP), derived from Vitamin B6.
- Mechanism of PLP: PLP transiently accepts the amino group from the amino acid and then donates it to the α-keto acid.
- Key Players:
- α-Ketoglutarate: A central amino group acceptor, becoming Glutamate.
- Glutamate: Serves as a collecting point for amino groups.
- Significance: Collects amino groups, allows for interconversion of non-essential amino acids, and serves as a source of diagnostic markers (ALT/AST for liver damage).
Oxidative Deamination: Release of Ammonia
- Definition: Oxidative deamination is the process by which the amino group is removed from an amino acid, typically glutamate, and released as free ammonia (NH₃). This reaction is irreversible.
- Primary Enzyme: The key enzyme is Glutamate Dehydrogenase.
- Location: Found in the mitochondria, particularly high in the liver and kidney.
- Reaction:
Glutamate + NAD(P)⁺ + H₂O → α-Ketoglutarate + NH₄⁺ + NAD(P)H + H⁺ - Coenzymes: Can use either NAD⁺ or NADP⁺.
- Regulation: Glutamate dehydrogenase is allosterically regulated:
- Activated by: ADP, GDP (indicating low energy).
- Inhibited by: ATP, GTP (indicating high energy).
- Significance: This is the major source of ammonia destined for the urea cycle and links amino acid catabolism to the TCA cycle via α-ketoglutarate.
Fate of the Ammonia Produced from Deamination
Ammonia (NH₃) and ammonium ions (NH₄⁺) are highly toxic, especially to the central nervous system. Their detoxification and excretion are crucial.
- Transport to the Liver:
- Glutamine Synthetase: In most peripheral tissues, ammonia is "fixed" to glutamate to form glutamine, a non-toxic transport form.
- Glucose-Alanine Cycle: In muscle, amino groups are transferred to pyruvate to form alanine, which is then transported to the liver.
- Detoxification in the Liver (Urea Cycle): The liver is the primary site for converting toxic ammonia into non-toxic urea.
- Excretion: Urea is transported to the kidneys and excreted in the urine.
Fate of the α-Keto Acid Carbon Skeletons
After removal of the amino group, the remaining carbon skeleton can be channeled into various pathways:
- Glucogenic Amino Acids:
- Definition: Amino acids whose carbon skeletons can be converted into glucose via gluconeogenesis.
- Mechanism: Their α-keto acids are converted into intermediates of the TCA cycle (e.g., α-ketoglutarate, succinyl CoA) or directly into pyruvate.
- Ketogenic Amino Acids:
- Definition: Amino acids whose carbon skeletons can be converted into ketone bodies or fatty acids.
- Mechanism: Their α-keto acids are converted into Acetyl-CoA or Acetoacetyl-CoA.
- List: Only two amino acids are purely ketogenic: Leucine and Lysine.
- Mixed Amino Acids (Glucogenic and Ketogenic):
- Definition: Amino acids whose skeletons yield both glucogenic and ketogenic intermediates.
- List: Phenylalanine, Tyrosine, Tryptophan, Isoleucine, Threonine.
- Energy Production: The α-keto acids can also be directly oxidized in the TCA cycle to generate ATP, especially when amino acids are in excess or energy demands are high.
The Urea Cycle
The Urea Cycle (sometimes called the Ornithine Cycle) is the body's main safety system for handling nitrogen. It is a metabolic pathway (a series of chemical reactions) that occurs primarily in the Liver.
The Main Goal: To turn Ammonia (NH₃), which is highly toxic and dangerous to the brain, into Urea, which is much less toxic and safe to travel through the blood. The kidneys then filter the urea out into urine so it can leave the body.
🔑 Key Vocabulary (Read this first)
- Metabolic Pathway: A step-by-step chain of chemical reactions in the body.
- Mitochondria: The "power plant" inside a cell. This is a separate room inside the cell where the first steps happen.
- Cytosol: The liquid "main floor" of the cell that surrounds the mitochondria. The later steps happen here.
- Enzyme: A special protein that builds or breaks other molecules. Think of it as a worker or a machine.
- ATP: The energy currency of the cell. The body "pays" ATP to make reactions happen.
- Substrate/Reactant: The ingredients used at the start of a reaction.
- Product: The result made at the end of a reaction.
A. Steps and Intermediates of the Urea Cycle
The cycle has 5 distinct steps. It is unique because it happens in two different places within the liver cell. It starts in the Mitochondria and finishes in the Cytosol.
Phase 1: Mitochondrial Reactions (Inside the "Inner Room")
Steps 1 and 2 happen here.
Step 1: Carbamoyl Phosphate Synthesis
⚠️ This is the Rate-Limiting Step (The most critical step)
- Reactants (Ingredients): Ammonia (NH₃) + Bicarbonate (HCO₃⁻).
- Enzyme (The Worker): Carbamoyl Phosphate Synthetase I (CPS-I).
- Product (Result): Carbamoyl Phosphate.
- Energy Cost: Requires 2 ATP. This is an expensive step!
Detailed Note:
This enzyme, CPS-I, lives in the mitochondria. Do not confuse it with CPS-II, which lives in the cytosol and is used to make DNA building blocks (pyrimidines). This distinction is very important.
Step 2: Citrulline Synthesis
- Reactants: Carbamoyl Phosphate + Ornithine.
- Enzyme: Ornithine Transcarbamoylase (OTC).
- Product: Citrulline.
How it works:
Think of Ornithine as a "carrier vehicle." It picks up the Carbamoyl Phosphate to form Citrulline. Once Citrulline is formed, it is able to leave the mitochondria and travel out into the cytosol for the next phase.
Phase 2: Cytosolic Reactions (On the "Main Floor")
Steps 3, 4, and 5 happen here.
Step 3: Argininosuccinate Synthesis
Now that Citrulline has arrived in the cytosol, it meets a new ingredient.
- Reactants: Citrulline + Aspartate.
- Enzyme: Argininosuccinate Synthetase.
- Product: Argininosuccinate.
- Energy Cost: Requires 1 ATP (But it is hydrolyzed to AMP + PPi).
Important Details:
- The Nitrogen Source: The molecule Aspartate is very important because it donates the second nitrogen atom needed to build Urea.
- Energy Math: Even though only 1 ATP molecule is used, it is broken down deeply (into AMP), so the energy cost is equivalent to using 2 ATPs.
Step 4: Arginine Formation
- Reactant: Argininosuccinate.
- Enzyme: Argininosuccinase (also called Argininosuccinate Lyase).
- Products: Arginine + Fumarate.
The Connection:
The product Fumarate is a byproduct (a leftover). However, the body does not waste it. Fumarate enters the TCA Cycle (Krebs Cycle) to help make energy. This links the Urea Cycle to other energy cycles.
Step 5: Urea Cleavage (The Final Cut)
- Reactant: Arginine.
- Enzyme: Arginase.
- Products: Urea + Ornithine.
Completing the Cycle:
- Urea: This is the final safe waste product. It travels to the kidneys to be peed out.
- Ornithine: Notice that we made Ornithine again? This Ornithine is transported back into the mitochondria to start Step 2 again. This is why it is called a "Cycle."
B. Quick Reference: Enzyme Locations
Inside Mitochondria
- Carbamoyl Phosphate Synthetase I (CPS-I)
- Ornithine Transcarbamoylase (OTC)
Inside Cytosol
- Argininosuccinate Synthetase
- Argininosuccinase (Lyase)
- Arginase
C. Regulation: How the Body Controls the Speed
The body is smart. It does not run this cycle at full speed all the time. It regulates (controls) the speed based on how much protein you eat.
1. The "Master Switch": N-Acetylglutamate (NAG)
The enzyme CPS-I (from Step 1) is the rate-limiting enzyme. It acts like a gate. To open the gate, it needs a specific key.
- The Key: A molecule called N-Acetylglutamate (NAG).
- How it works (Allosteric Activation): When NAG attaches to CPS-I, it changes the shape of the enzyme, turning it "ON." Without NAG, CPS-I cannot work.
- Where does the Key (NAG) come from?
- NAG is made by an enzyme called NAG Synthase.
- NAG Synthase is stimulated by Arginine and Glutamate.
- The Logic: If you eat a lot of protein, your Arginine and Glutamate levels go up. This tells the body to make more NAG. More NAG turns on the Urea Cycle to clean up the waste from the protein.
2. Substrate Availability (Supply and Demand)
Simply put, if there is more "stuff" to process, the cycle goes faster. The rate increases if there are higher levels of Ammonia, Bicarbonate, or Aspartate available.
3. Long-Term Induction (Adaptation)
If you change your lifestyle for a long time, the body physically builds more of the urea cycle enzymes.
- High-Protein Diet: Eating lots of meat creates more nitrogen waste, so the liver builds more enzymes to cope.
- Starvation: During starvation, the body breaks down its own muscles (protein) for energy. This releases nitrogen, so the body must increase enzyme levels to handle the load.
D. Why is the Urea Cycle So Important?
- Detoxification (Safety): This is the #1 reason. Ammonia is toxic to neurons (brain cells). The cycle converts it into Urea, which is safe. Without this cycle, ammonia builds up (Hyperammonemia), leading to coma or death.
- Nitrogen Excretion: We cannot store excess nitrogen. Urea is the main vehicle for carrying nitrogen out of the body in urine.
- Balance (Homeostasis): It keeps the nitrogen levels in the body stable.
- Metabolic Connection: By producing Fumarate (in Step 4) and using Aspartate, it connects to the TCA cycle (energy production) and Gluconeogenesis (making sugar).
Summary: The "Math" of the Cycle
If we look at the Urea Cycle as one big equation, here is what goes in and what comes out.
Inputs (Cost)
- 2 Ammonia (NH₃): One is free ammonia, the second comes from Aspartate.
- 1 CO₂: Comes from Bicarbonate (HCO₃⁻).
- 3 ATP: This is the energy cost (used in Step 1 and Step 3).
Outputs (Result)
- 1 Urea: The waste product.
- 1 Fumarate: Sent to the TCA cycle.
- 2 ADP + 1 AMP: The leftovers of the used energy.
Overall Chemical Reaction:
NH₄⁺ + HCO₃⁻ + Aspartate + 3 ATP → Urea + Fumarate + 2 ADP + AMP + 4 Pi + H₂O
Classification & Metabolism of Amino Acids
Once the body removes the nitrogen (amino group) from an amino acid, what is left? We call the remaining part the "Carbon Skeleton."
The Big Question: What does the body do with this Carbon Skeleton?
The answer depends on the specific amino acid. It can be turned into Glucose (Sugar), Ketones/Fat, or Both.
1. Classifying Amino Acids by Their Products
We classify amino acids into three groups based on what they become after they are broken down (catabolized).
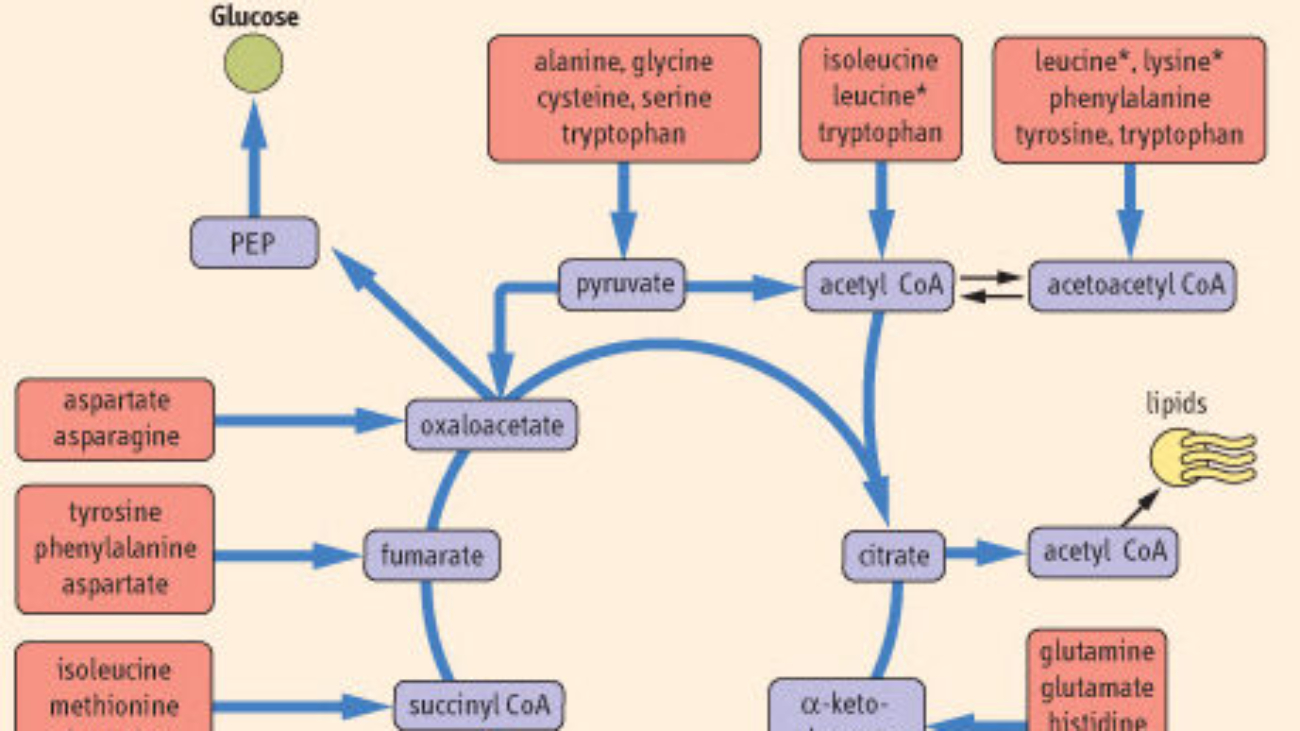
A. Glucogenic Amino Acids
"Gluco" = Glucose (Sugar) | "Genic" = Creating
Definition: These are amino acids whose carbon skeletons can be converted into Pyruvate or intermediates of the TCA Cycle (like α-ketoglutarate, succinyl CoA, fumarate, or oxaloacetate).
Why does this matter? (Significance):
- All these intermediates can be used to make new Glucose through a process called Gluconeogenesis.
- Scenario: Imagine you are starving or fasting. Your brain needs glucose to survive. The body breaks down these amino acids to make that vital sugar.
Examples (Sorted by what they enter):
- Enter as Pyruvate: Alanine, Cysteine, Glycine, Serine, Threonine, Tryptophan.
- Enter as α-Ketoglutarate: Arginine, Glutamate, Glutamine, Histidine, Proline.
- Enter as Succinyl CoA: Isoleucine, Methionine, Threonine, Valine.
- Enter as Fumarate: Aspartate, Phenylalanine, Tyrosine.
- Enter as Oxaloacetate: Asparagine, Aspartate.
B. Ketogenic Amino Acids
"Keto" = Ketones/Fat
Definition: These amino acids convert into Acetyl-CoA or Acetoacetyl-CoA.
Important Rule: These CANNOT make Glucose.
Why? Because in mammals, the step turning Pyruvate into Acetyl-CoA is irreversible (one-way only). Once you are Acetyl-CoA, you cannot go back up to become sugar.
Significance:
- They are used to make Ketone Bodies (alternative fuel for the brain during long starvation) or Fatty Acids (fat storage).
The "Exclusive" List (Only 2):
There are only two amino acids that are purely ketogenic:
- Leucine
- Lysine
(Mnemonic: The "L" amino acids differ from the rest).
C. Mixed Amino Acids
Glucogenic AND Ketogenic
Definition: These are flexible. When they break down, part of their skeleton becomes a precursor for glucose, and another part becomes a precursor for ketones/fat.
Examples:
- Phenylalanine
- Tyrosine
- Tryptophan
- Isoleucine
- Threonine
Note: You will see these names appear in the Glucogenic list as well because they fit both categories.
Visual Summary: Where do they go?
2. Metabolism of Specific Amino Acid Groups
While all amino acids undergo transamination (removing nitrogen), the path for their carbon skeletons is unique. We will look at three special groups.
A. Branched-Chain Amino Acids (BCAAs)
Who are they? Leucine, Isoleucine, Valine.
Unique Feature: Unlike most amino acids that go to the Liver, BCAAs are primarily metabolized in the Muscles (and other peripheral tissues).
Why? The liver lacks the first enzyme needed to break them down.
The Pathway:
Step 1: Transamination (Moving the Nitrogen)
The enzyme Branched-chain Aminotransferase (BCAT) removes the amino group.
- Location: Skeletal muscle, kidney, brain.
- Result: We are left with α-Keto Acids (specifically called BCKAs).
Step 2: Oxidative Decarboxylation (The Irreversible Step)
The BCKAs are processed by a massive enzyme complex called Branched-Chain α-Keto Acid Dehydrogenase (BCKD).
- Required Helpers (Coenzymes): It needs 5 friends to work: TPP, FAD, NAD+, Lipoic Acid, and Coenzyme A.
If a person is born without this BCKD enzyme complex, they cannot break down BCAAs. The "Keto Acids" build up in the blood and urine. The urine smells sweet like maple syrup/burnt sugar. This accumulation is toxic to the brain (neurotoxic) and can cause death if not treated.
Step 3: The End Products
- Leucine → Becomes Acetyl-CoA (Purely Ketogenic).
- Valine → Becomes Succinyl-CoA (Purely Glucogenic).
- Isoleucine → Becomes Acetyl-CoA AND Succinyl-CoA (Mixed).
Significance of BCAAs:
- Muscle Fuel: A key energy source during exercise.
- Building Muscle: Leucine signals the muscle to start building protein.
- Nitrogen Transport: They help form Alanine, which carries nitrogen safely to the liver.
B. Aromatic Amino Acids
These amino acids have a ring structure (benzene ring). They are Phenylalanine, Tyrosine, and Tryptophan.
1. Phenylalanine & Tyrosine
Phenylalanine is an Essential amino acid (you must eat it). Tyrosine is made from Phenylalanine.
Phenylalanine + O₂ + BH4 → Tyrosine + H₂O + BH2
- Enzyme: Phenylalanine Hydroxylase (PAH).
- Coenzyme: Tetrahydrobiopterin (BH4).
If the enzyme PAH is missing or broken:
- Phenylalanine cannot turn into Tyrosine.
- Phenylalanine builds up to dangerous levels.
- This is toxic to the brain and causes severe intellectual disability.
- Treatment: A lifelong diet with very low Phenylalanine.
What does Tyrosine become?
- Catabolism: Broken down into Fumarate (Glucogenic) and Acetoacetate (Ketogenic).
- Special Products: Tyrosine is the raw material for:
- Catecholamines: Dopamine, Norepinephrine, Epinephrine (Adrenaline).
- Thyroid Hormones: T3 and T4.
- Melanin: The pigment for skin and hair.
2. Tryptophan (Essential)
Tryptophan has a very complex breakdown path. It is a Mixed amino acid.
- End Products: Alanine (Glucogenic) and Acetyl-CoA (Ketogenic).
- Important Derivatives (What it makes):
- Serotonin: Regulates mood and appetite.
- Melatonin: Regulates sleep cycles.
- Niacin (Vitamin B3): We can make a small amount of this vitamin from Tryptophan.
C. Sulfur-Containing Amino Acids
These contain Sulfur atoms: Methionine and Cysteine.
1. Methionine (Essential)
Methionine is famous for being a "Donor." It gives away methyl groups (CH3) to help build other things.
The Cycle of Methionine (Step-by-Step):
- Activation: Methionine + ATP → SAM (S-Adenosylmethionine).
Think of SAM as "Super Active Methionine." - Donation: SAM gives away its Methyl group and becomes SAH.
- Hydrolysis: SAH is broken down into Homocysteine.
The Fate of Homocysteine (The Fork in the Road):
Homocysteine is dangerous if it stays. It must go somewhere. It has two choices:
Turn back into Methionine.
Needs: Vitamin B12 + Folate.
Turn into Cysteine.
Needs: Vitamin B6.
If the enzymes needed to clear Homocysteine don't work (genetic defect), Homocysteine levels rise. This causes heart problems, skeletal deformities, and eye issues.
2. Cysteine
Cysteine is usually made from Methionine. However, if you don't eat enough Methionine, Cysteine becomes essential.
- Catabolism: It breaks down into Pyruvate (Glucogenic) and Sulfate.
- Important Derivatives:
- Glutathione: The body's master antioxidant (detoxifier).
- Taurine: Found in bile.
- Coenzyme A: Vital for energy metabolism.
Interconnectedness of Metabolism
Amino acid metabolism does not happen in a lonely island. It is like a city with many roads connecting to other neighborhoods. It is tightly linked to Carbohydrates (Sugar) and Lipids (Fats).
Why is this important?
This connection gives the body "Metabolic Flexibility." It ensures you can survive different situations—whether you just ate a huge meal (feast) or haven't eaten for days (famine/starvation).
A. Connection to Carbohydrate (Sugar) Metabolism
1. Glycolysis and Gluconeogenesis
Many amino acids break down into Pyruvate. Pyruvate is a famous "crossroads" molecule. Once an amino acid becomes Pyruvate, it has three choices:
- Choice 1 (Energy): Turn into Acetyl-CoA and burn in the TCA cycle.
- Choice 2 (No Oxygen): Turn into Lactate (Lactic Acid).
- Choice 3 (Make Sugar): Turn into Oxaloacetate, which is then used to build Glucose (Gluconeogenesis).
2. The Glucose-Alanine Cycle (Muscle-Liver Link)
This is a specific transport system that connects your muscles to your liver. Think of Alanine as a "Taxi."
- In the Muscle: When muscles work, they make waste (Pyruvate) and breakdown amino acids (Nitrogen). They combine these to make Alanine.
- The Journey: Alanine travels through the blood to the Liver.
- In the Liver: The Liver separates them.
- The Nitrogen goes to the Urea Cycle (to be excreted).
- The Pyruvate is turned back into Glucose.
- Return Trip: The Glucose is sent back to the muscle to be used as fuel again.
B. Connection to Lipid (Fat) Metabolism
When amino acids break down into Acetyl-CoA, they enter the world of fats.
1. Making Fat (Storage)
If you have too much energy (you ate too much protein and carbs), the body uses the Acetyl-CoA from amino acids to synthesize Fatty Acids for storage.
2. Making Ketones (Survival)
If you are starving, the body turns Acetyl-CoA into Ketone Bodies. These serve as emergency fuel for the Brain and Heart.
Note: Acetyl-CoA is also used to make Cholesterol.
C. Connection to the TCA Cycle (Krebs Cycle)
The TCA cycle is the "Central Hub" or the "Roundabout" of metabolism.
Concept: Anaplerosis ("Topping Up")
Sometimes, the TCA cycle runs out of ingredients (intermediates) because they were taken away to build other things. Glucogenic amino acids can be broken down to refill these ingredients. This refilling process is called Anaplerosis.
Energy Production: Ultimately, the carbon skeletons of all amino acids can be fully burned in this cycle to produce ATP (Energy).
D. Nucleotide Metabolism
DNA and RNA need Nitrogen and Carbon to be built.
- Nitrogen Source: Supplied by Glutamine, Aspartate, and Glycine.
- Carbon Source: Supplied by Glycine.
E. Regulatory Cross-Talk
Hormones control these choices:
- Insulin (Fed State): Says "Build!" Promotes protein synthesis.
- Glucagon (Fasting State): Says "Break down!" Stimulates turning amino acids into glucose.
- ATP Levels: High ATP means "We are full," favoring synthesis. Low ATP means "We are hungry," favoring breakdown for energy.
Common Metabolic Disorders
These are "Inborn Errors of Metabolism." They are usually genetic (inherited from parents). A specific enzyme is broken or missing. This causes a traffic jam: Toxic precursors build up and Essential products run out.
1. Phenylketonuria (PKU)
Defect: Phenylalanine Hydroxylase (PAH)
The Mechanism:
The body cannot convert Phenylalanine into Tyrosine.
- Accumulation: Phenylalanine builds up. It turns into toxic acids (Phenylpyruvate) causing a "Mousy" (mouse-like) odor in urine.
- Deficiency: Tyrosine becomes essential (because we can't make it). Less melanin is made, leading to fair skin/hair.
🚨 Clinical Signs & Danger:
- Neurotoxicity: High Phenylalanine destroys the brain.
- Symptoms: Severe intellectual disability, microcephaly (small head), seizures.
Treatment: Lifelong diet restriction. No meat, dairy, or aspartame. Special formula required.
2. Maple Syrup Urine Disease (MSUD)
Defect: Branched-Chain α-Keto Acid Dehydrogenase (BCKD)
- The Problem: Cannot break down Leucine, Isoleucine, and Valine (BCAAs).
- Symptoms (Neonatal): Poor feeding, vomiting, coma, seizures.
- Outcome: Severe brain damage or death if not treated immediately.
- Treatment: Diet strictly limiting BCAAs.
3. Alkaptonuria (Black Urine Disease)
Defect: Homogentisate 1,2-Dioxygenase (HGD)
This is a defect in Tyrosine breakdown. A chemical called Homogentisic Acid (HGA) builds up.
4. Homocystinuria
Defect: Cystathionine β-Synthase (CBS)
The Problem: Methionine and Homocysteine levels are too high. Cysteine becomes essential.
Clinical Appearance (Marfan-like):
- Eyes: Dislocation of the lens (Ectopia Lentis).
- Skeleton: Tall, thin body with long limbs (Marfanoid habitus). Osteoporosis.
- Vascular (Critical): High risk of blood clots (Thrombosis), causing strokes or heart attacks at a young age.
Treatment: High doses of Vitamin B6 (if responsive), low methionine diet, and Betaine.
5. Urea Cycle Disorders (UCDs)
Defect: Any enzyme in the Urea Cycle
The Killer: Hyperammonemia (High Ammonia).
What happens? Ammonia is not removed. It reaches the brain and causes:
- Vomiting and Lethargy (tiredness).
- Cerebral Edema (Brain swelling).
- Coma and Death.
Treatment: Restrict protein intake. Use drugs to scavenge ammonia. Liver transplant may be needed.
Nitrogen Catabolism & Toxicity
While we know how the Urea Cycle works, we must understand why and when the body decides to break down proteins, and exactly why ammonia is so dangerous to the brain.
1. When does Protein Catabolism happen?
The body does not store protein like it stores fat. It breaks it down in three specific situations:
-
🔄
Normal Turnover: Old proteins are broken down to build new ones. Any extras are destroyed.
-
🍖
Dietary Surplus: If you eat more protein than you need, the body cannot store it. It breaks the surplus down for energy.
-
⚠️
Starvation or Diabetes: When sugar (carbohydrates) is unavailable, the body breaks down its own muscle protein to use as emergency fuel.
2. Mechanisms of Nitrogen Removal
Before we can burn the amino acid for energy, we must remove the nitrogen. This happens in two ways.
A. Transamination (The Swap)
We swap the Amino Group onto α-Ketoglutarate to form Glutamate.
- Enzymes: Aminotransferases (like AST and ALT).
- Coenzyme Required: PLP (Vitamin B6).
- Clinical Note: High levels of AST or ALT in the blood indicate Liver or Heart damage (the cells burst and leak the enzyme).
B. Deamination (The Removal)
Removing the amino group completely to release Ammonia (NH₄⁺).
Performed by Glutamate Dehydrogenase. It uses NAD+ or NADP+. This is the main way Glutamate releases ammonia in the liver.
Specific to Serine and Threonine (because they have an -OH group). Used enzymes called Dehydratases (e.g., Serine Dehydratase).
3. Transport: The Ammonia Taxi System
Ammonia is toxic. It cannot swim freely in the blood. It must be carried by safe "Taxi" molecules.
Taxi 1: Glutamine
From Brain & Kidney → To Liver
Ammonia + Glutamate → Glutamine.
Glutamine is neutral and non-toxic. It travels to the liver, where the enzyme Glutaminase breaks it back down to release the ammonia.
Taxi 2: Alanine
From Muscle → To Liver
Muscle waste (Pyruvate) + Nitrogen → Alanine.
Alanine travels to the liver. The liver takes the Nitrogen for Urea, and turns the Pyruvate back into Glucose (Glucose-Alanine Cycle).
4. Clinical Pathology: When things go wrong
Blood Urea Nitrogen (BUN)
- Normal Range: 20 – 40 mg/dL.
- Significance: High BUN usually means the Kidneys are not working (they aren't filtering the urea out).
- Causes of High BUN (Uremia):
- Pre-renal: Blood flow issue (heart failure).
- Renal: Kidney damage.
- Post-renal: Blockage (kidney stones/tumor).
Why is Ammonia Toxic to the Brain?
If the liver fails (Cirrhosis) or the Urea Cycle has a genetic defect, ammonia builds up. It causes tremors, slurred speech, coma, and death. But why?
To try and clean up the ammonia, the brain combines it with α-Ketoglutarate to make Glutamate.
The Problem: α-Ketoglutarate is needed for the Krebs Cycle (energy). If you use it all up to fight ammonia, the Krebs cycle stops. The brain runs out of ATP (Energy).
Excess Glutamate creates excess GABA, an inhibitory neurotransmitter. This slows down brain signals (causing lethargy/coma).
Accumulation of Glutamine inside brain cells pulls water in (osmosis). This causes Cerebral Edema (Brain Swelling), which can be fatal.
Treatment Note: Lactulose
Hepatic Encephalopathy (Brain damage from liver ammonia) is often treated with Lactulose, which helps pull ammonia into the gut to be pooped out.
Biosynthesis of Amino Acids
Biosynthesis (Anabolism) is the process of the body building complex molecules from simple ones. In this section, we explore how the body creates Amino Acids, which are the building blocks of proteins, nucleotides, and lipids.
Introduction & Key Concepts
- Ancient Pathways: These chemical pathways are very old in evolutionary history.
- Shared Roads: Building (Anabolism) often uses the same ingredients as Breaking Down (Catabolism).
- Source of Carbon: The "backbones" of amino acids come from three main places:
- Glycolysis
- Citric Acid Cycle (TCA)
- Pentose Phosphate Pathway
- Stereochemistry: Our body specifically makes L-Amino Acids. This shape is enforced during the Transamination step.
1. Nitrogen Fixation: Getting Nitrogen
Before we can build an amino acid, we need Nitrogen. The air is 80% Nitrogen Gas (N₂), but our bodies cannot use gas. It must be "fixed" (turned into a solid/liquid form like Ammonia, NH₃).
Who fixes Nitrogen?
- 60% - Microorganisms: Specific bacteria (Diazotrophs) do the heavy lifting. They use ATP and a protein called Ferredoxin.
- 15% - Nature's Power: Lightning and UV radiation have enough energy to break nitrogen bonds.
- 25% - Industrial: Humans do it chemically.
The Industrial Method (Haber Process)
Fritz Haber discovered how to do this in a factory.
Equation: N₂ + 3H₂ → 2NH₃
The Biological Machine: Nitrogenase Complex
Bacteria use a complex enzyme system to turn N₂ into NH₃. This system has two distinct parts working together.
Part 1: The Reductase (The "Fe Protein")
Function: This is the power supply. It gathers electrons.
- Contains a 4Fe-4S center (Iron-Sulfur cluster).
- It hydrolyzes (burns) ATP.
- This burning causes a shape change (conformational change) that pushes electrons to Part 2.
Part 2: The Nitrogenase (The "MoFe Protein")
Function: This is the factory where the chemistry happens.
- Structure: It is an α2β2 tetramer (4 subunits) weighing 240 kD.
- The P-Cluster: Where electrons enter.
- The Cofactor: It contains an Iron-Molybdenum (FeMo) cofactor. This specific metal cluster is what binds to Nitrogen (N₂) and reduces it to Ammonia (NH₃).
2. Assimilation: Bringing Ammonia into the Body
Once we have Ammonia (NH₄⁺), we must attach it to a carbon molecule to start making amino acids. This happens through two main "Gatekeeper" enzymes: Glutamate and Glutamine.
Gate 1: Glutamate Dehydrogenase
This enzyme combines Ammonia with α-Ketoglutarate (from the TCA cycle).
Significance: Most other amino acids get their α-amino group (their nitrogen) from Glutamate via Transamination.
Gate 2: Glutamine Synthetase
This enzyme adds a second nitrogen to Glutamate to make Glutamine.
Significance: The sidechain nitrogen of Glutamine is used to build complex amino acids like Tryptophan and Histidine.
3. The Amino Acid Families
Amino acids are grouped into "Families" based on which carbon skeleton they come from.
| Origin (Parent) | Amino Acids Produced (Children) |
|---|---|
| Oxaloacetate | Aspartate → Asparagine, Methionine, Threonine, Lysine |
| Pyruvate | Alanine, Valine, Leucine, Isoleucine |
| α-Ketoglutarate | Glutamate → Glutamine, Proline, Arginine |
| 3-Phosphoglycerate | Serine → Glycine, Cysteine |
| PEP + Erythrose-4P | Phenylalanine, Tyrosine, Tryptophan (Aromatic) |
| Ribose-5-Phosphate | Histidine |
Essential vs. Non-Essential
Non-Essential (We make them)
These pathways are simple (few steps).
Examples: Alanine, Glutamate, Aspartate.
Essential (Must eat them)
These pathways are complex (many steps). We lost the ability to make them.
Examples: Histidine, Lysine, Methionine, Valine.
Observation: The graph in the slides shows a direct link—Essential amino acids require many more enzymatic steps to create than non-essential ones.
4. Details of Specific Pathways
A. Aspartate and Alanine (Transamination)
These are made by simply swapping the oxygen group for an amino group using Glutamate.
- Oxaloacetate + Glutamate ↔ Aspartate + α-Ketoglutarate
- Pyruvate + Glutamate ↔ Alanine + α-Ketoglutarate
B. Asparagine (Amidation)
We take Aspartate and add another nitrogen.
Aspartate + ATP + Glutamine (Donor) → Asparagine + Glutamate + AMP + PPi
C. Proline and Arginine
Both are made from Glutamate.
- Glutamate is reduced to Glutamic γ-semialdehyde.
- This intermediate cyclizes (forms a ring) to eventually become Proline.
- Or, through the urea cycle (involving Ornithine), it becomes Arginine.
D. Serine and Glycine
- Start: 3-Phosphoglycerate (from glycolysis).
- Oxidation: Converted to 3-Phosphohydroxypyruvate.
- Transamination: Converted to 3-Phosphoserine.
- Hydrolysis: Converted to Serine.
How to make Glycine?
The enzyme Serine Transhydroxymethylase removes a carbon from Serine to make Glycine. This requires Tetrahydrofolate.
5. One-Carbon Metabolism (The Carriers)
The body often needs to move single carbon atoms (methyl groups) around to build things. It uses two main "Postmen" for this.
Carrier 1: Tetrahydrofolate (THF)
Derived from Folic Acid (Vitamin B9).
- It carries 1-carbon groups on its Nitrogen atoms (N5 or N10).
- It can carry them in different "Oxidation States" (Methyl, Methylene, Formyl, etc.).
- Limit: It is not strong enough to donate methyl groups for some hard reactions (like DNA methylation).
Carrier 2: S-Adenosylmethionine (SAM)
The "Super" Donor.
- Made from Methionine + ATP.
- It has a high "Methyl Transfer Potential" (it really wants to give away its methyl group).
- Use: Used for DNA methylation and other difficult synthesis tasks.
The Activated Methyl Cycle:
Methionine → SAM → (Donates CH3) → S-Adenosylhomocysteine → Homocysteine → (Regenerates) → Methionine
6. Aromatic Amino Acids
These are the amino acids with rings: Phenylalanine, Tyrosine, and Tryptophan.
The Shikimate & Chorismate Pathway
Plants and bacteria use this pathway (humans don't—that's why these are essential for us).
- Key Intermediate: Chorismate.
- Chorismate branches out to form Phenylalanine and Tyrosine (via Prephenate).
- Chorismate also converts to Anthranilate to eventually form Tryptophan (using PRPP).
The weedkiller Glyphosate works by inhibiting the enzyme that makes Chorismate. Because humans do not have this enzyme, Roundup is toxic to plants but relatively safe for humans.
7. Regulation: Controlling the Factory
The body doesn't waste energy. If we have enough amino acids, we stop making them. This is done via Feedback Inhibition.
Basic Feedback Inhibition
The final product (Z) goes back and inhibits the first enzyme (A → B).
A → B → C → D → E → Z (Z blocks A)
Example: Serine
Serine inhibits the enzyme 3-phosphoglycerate dehydrogenase.
Complex Regulation Strategies
- Enzyme Multiplicity: Having 3 versions of the same enzyme (isozymes). One is inhibited by Lysine, one by Methionine, one by Threonine. This allows fine-tuning (seen in Aspartokinase).
- Cumulative Feedback: The enzyme is only partially stopped by one product. To stop it completely, ALL products must be present (Example: Glutamine Synthetase).
- Cascade Control (Glutamine Synthetase): This enzyme is so important it has a "Master Switch." It is controlled by Adenylylation (adding AMP).
- Adenylylated = Less Active.
- Deadenylylated = More Active.
- This switch is controlled by regulatory proteins (Pa/Pd) sensing ATP and α-Ketoglutarate levels.
8. Amino Acid Derivatives
Amino acids are not just for proteins. They are precursors for many vital biomolecules.
Made from Glutamate + Cysteine + Glycine. It is the body's main antioxidant and sulfhydryl buffer.
Made from Arginine. It is a short-lived signal molecule (vasodilator).
Made from Glycine + Succinyl-CoA. Essential for blood (Hemoglobin).
Tyrosine → Dopamine/Adrenaline.
Tryptophan → Serotonin.
Histidine → Histamine.
Amino Acid Carbon Skeleton Catabolism
Introduction: When we break down amino acids, we first remove the Nitrogen (Amino group). What is left is called the "Carbon Skeleton" (the Alpha-Keto Acid).
The Main Goal:
To turn these skeletons into energy. They must be converted into one of the 7 molecules that can enter the central energy pathways (TCA Cycle or Glycolysis).
1. Classification: What do they become?
We categorize amino acids based on their final product.
A. Glucogenic
Makes Glucose (Sugar)
These turn into Pyruvate or TCA cycle intermediates (like Oxaloacetate).
- Alanine, Arginine
- Asparagine, Aspartate
- Cysteine, Glutamate
- Glutamine, Glycine
- Proline, Serine, Histidine
- Methionine, Valine
B. Ketogenic
Makes Ketones/Fat
These turn into Acetyl-CoA. They cannot become sugar.
- Leucine
- Lysine
C. Mixed
Makes Both
Part of the molecule becomes sugar, part becomes fat.
- Tyrosine
- Isoleucine
- Phenylalanine
- Tryptophan
- Threonine
2. Metabolism of Glycine & Threonine
Glycine Degradation
Glycine has 3 pathways to be broken down:
- Pathway 1 (Conversion to Serine):
Enzyme: Serine Hydroxymethyltransferase.
Requires: Tetrahydrofolate (Folate) and Pyridoxal Phosphate (Vitamin B6). - Pathway 2 (Major Animal Pathway):
Oxidative cleavage breaks Glycine into CO₂, Ammonia (NH₄⁺), and a methylene group (-CH₂-). - Pathway 3: Does not lead to Pyruvate (less common).
Threonine Degradation
Threonine has two roads it can take:
Threonine is turned into Glycine first, then into Pyruvate. This accounts for only 10-30% of breakdown in humans.
This is the primary way humans handle Threonine. It yields Propionyl-CoA, which eventually becomes Succinyl-CoA.
3. Amino Acids Forming Acetyl-CoA
Seven amino acids break down into Acetyl-CoA. We will focus on the most clinically important pathway: Phenylalanine and Tyrosine.
The Phenylalanine → Tyrosine Pathway
Step 1: Hydroxylation
Phenylalanine is converted to Tyrosine by the enzyme Phenylalanine Hydroxylase.
Tetrahydrobiopterin (BH4)
BH4 donates electrons to the reaction and becomes BH2. It must be recharged back to BH4 to work again.
Phenylketonuria (PKU) occurs if Phenylalanine Hydroxylase is missing. Phenylalanine builds up and damages the brain.
Step 2: Tyrosine Breakdown
Tyrosine is further broken down to produce Fumarate and Acetoacetate.
If the enzyme Homogentisate oxidase is missing, Homogentisate accumulates. This causes Alkaptonuria (Black Urine Disease).
4. Amino Acids Forming α-Ketoglutarate
Five amino acids enter the cycle here: Proline, Glutamate, Glutamine, Arginine, Histidine.
-
1. Glutamine:
Uses the enzyme Glutaminase to donate its amide nitrogen, becoming Glutamate.
-
2. Proline:
Proline is a ring. The ring is opened (oxidized) to form a Schiff base, then hydrolyzed to form Glutamate γ-semialdehyde, which becomes Glutamate.
-
3. Arginine:
Converted to Ornithine (in the Urea Cycle). Ornithine is then converted to Glutamate γ-semialdehyde.
-
4. Histidine:
Follows a complex multistep path. Key detail: One carbon is removed using Tetrahydrofolate as a cofactor.
5. Amino Acids Forming Succinyl-CoA
These are Methionine, Isoleucine, Threonine, and Valine.
The Propionyl-CoA Pathway
All four of these amino acids eventually turn into Propionyl-CoA (a 3-carbon unit). The body must turn this into Succinyl-CoA (a 4-carbon unit) to use it.
The Critical Conversion Steps:
- Carboxylation: Propionyl-CoA adds a carbon to become Methylmalonyl-CoA. (Needs Biotin).
- Epimerization: The molecule is rearranged.
- Isomerization (The Mutase Step): Methylmalonyl-CoA is turned into Succinyl-CoA.
Important: This enzyme (Methylmalonyl-CoA Mutase) requires Vitamin B12 (Cobalamin).
If the B12-dependent mutase enzyme is missing, Methylmalonyl-CoA builds up. This causes severe metabolic acidosis.
6. Branched-Chain Amino Acids (BCAAs)
The BCAAs are Leucine, Isoleucine, and Valine.
Where does this happen?
Muscle, Adipose, Kidney, Brain.
NOT in the Liver. The Liver is missing the first enzyme (Aminotransferase) needed for BCAAs.
The BCKD Complex
After the amino group is removed, we are left with Alpha-Keto Acids. These are processed by a massive enzyme called the Branched-Chain α-Keto Acid Dehydrogenase (BCKD) Complex.
This complex performs "Oxidative Decarboxylation" (removing carbon as CO₂).
- Defect: The BCKD complex is broken.
- Result: Alpha-Keto acids accumulate in the blood and urine.
- Symptom: Urine smells sweet like Maple Syrup or burnt sugar.
- Danger: Causes mental retardation and death in infancy if untreated.
- Treatment: Strict diet restricting Valine, Isoleucine, and Leucine.
7. Asparagine and Aspartate
Destination: Oxaloacetate
These ultimately enter the cycle as Oxaloacetate.
- Asparagine is hydrolyzed by the enzyme Asparaginase. It releases NH₄⁺ and becomes Aspartate.
- Aspartate undergoes transamination (swaps Nitrogen) to become Oxaloacetate.
Biochemistry: Amino Acid Metabolism
Test your knowledge with these 30 questions.
Amino Acid Metabolism Quiz
Question 1/30
Quiz Complete!
Here are your results, .
Your Score
27/30
90%
Cholesterol Metabolism Pathway
Cholesterol Metabolism : Pathway
Cholesterol Metabolism
Cholesterol often gets a bad rap due to its association with heart disease, but it's crucial to understand that it is an essential molecule for life. Our bodies need cholesterol to function properly. The problem arises when its levels are imbalanced or when it's handled improperly within the body.
Importance and Physiological Functions of Cholesterol
Cholesterol is a lipid belonging to the steroid family. Its unique amphipathic structure (a polar hydroxyl group and a nonpolar steroid ring system and hydrocarbon tail) allows it to insert into cell membranes, giving it critical structural and signaling roles.
- Essential Component of Cell Membranes:
- Cholesterol is a major constituent of virtually all animal cell membranes.
- It modulates membrane fluidity, permeability, and stability, acting as a "buffer": at high temperatures, it stiffens the membrane, while at low temperatures, it prevents rigidity.
- It is particularly abundant in myelin sheaths, enhancing nerve signal transmission.
- Precursor for Steroid Hormones:
- Cholesterol is the obligate precursor for all five major classes of steroid hormones: Glucocorticoids (e.g., Cortisol), Mineralocorticoids (e.g., Aldosterone), Androgens (e.g., Testosterone), Estrogens (e.g., Estradiol), and Progestogens (e.g., Progesterone).
- Precursor for Bile Acids (and Bile Salts):
- In the liver, cholesterol is converted into primary bile acids.
- Bile acids emulsify dietary fats in the small intestine, facilitating their absorption. This is the primary way the body eliminates excess cholesterol.
- Precursor for Vitamin D Synthesis:
- 7-Dehydrocholesterol, a precursor in the cholesterol synthesis pathway, is converted to pre-vitamin D3 in the skin upon exposure to UV light.
- This is then converted to the active hormone, calcitriol, essential for calcium homeostasis.
Sources of Cholesterol
The body acquires cholesterol from two main sources:
- Endogenous Synthesis (De Novo Synthesis):
- The vast majority of cholesterol (about 80%) is synthesized internally, primarily in the liver (~50% of total synthesis), but also in the intestine, adrenal cortex, and reproductive organs.
- Dietary Intake (Exogenous Cholesterol):
- Cholesterol is consumed in the diet, found exclusively in animal products (meat, eggs, dairy). Plant foods do not contain cholesterol.
- The amount absorbed can vary significantly among individuals.
Absorption of Dietary Cholesterol
The process of dietary cholesterol absorption primarily occurs in the small intestine:
- Emulsification: Dietary cholesterol esters are emulsified by bile salts into smaller micelles.
- Hydrolysis: Cholesterol Esters (CE) are hydrolyzed into free cholesterol (FC) by pancreatic cholesterol esterase. Only free cholesterol can be absorbed.
- Micelle Formation: Free cholesterol and other digested lipids become incorporated into mixed micelles with bile salts.
- Uptake by Enterocytes:
- Mixed micelles diffuse to the brush border of the intestinal enterocytes.
- The primary transporter responsible for cholesterol uptake is the Niemann-Pick C1-Like 1 (NPC1L1) protein. This transporter is the target of the drug ezetimibe.
- Intracellular Processing and Re-esterification:
- Once inside the enterocyte, free cholesterol can be either effluxed back into the lumen via ABC G5/G8 transporters or re-esterified to cholesterol esters by the enzyme Acyl-CoA Cholesterol Acyltransferase 2 (ACAT2).
- Chylomicron Assembly and Secretion:
- The newly formed cholesterol esters and re-formed triacylglycerols are packaged with apolipoproteins (primarily apoB-48) into large lipoprotein particles called chylomicrons.
- Chylomicrons are then released into the lymphatic system, which eventually drains into the bloodstream.
Summary
So, to summarize, cholesterol is a vital molecule for cell structure, hormones, bile acids, and Vitamin D. We get it from both our diet and internal synthesis. Dietary cholesterol is absorbed in the small intestine via NPC1L1, processed, and then packaged into chylomicrons for transport.
Pathways of Cholesterol Synthesis (De Novo Synthesis)
Cholesterol is an indispensable molecule, and while we obtain some from our diet, the human body possesses the remarkable ability to synthesize nearly all the cholesterol it requires through a complex process known as de novo synthesis. This internal production ensures a constant supply for vital cellular functions.
Sites of Synthesis:
While virtually all nucleated cells can synthesize cholesterol, certain tissues are particularly active:
- The liver is the predominant site, responsible for approximately 50% of the body's synthesis.
- Other significant contributors include the intestine, the adrenal cortex, and the testes and ovaries.
Cellular Location of Enzymes:
The enzymatic machinery is distributed between two key cellular compartments:
- Enzymes for the initial stages are found in the cytoplasm.
- Enzymes for later stages are located within the membranes of the endoplasmic reticulum.
Requirements for Cholesterol Biosynthesis:
The synthesis of cholesterol is an energetically demanding process:
- Carbon Atoms: All 27 carbon atoms are derived from Acetyl-CoA. A total of 18 molecules are consumed.
- Reducing Equivalents: The process requires significant reducing power, supplied by NADPH (approx. 16 moles).
- Energy: The process requires considerable energy from ATP (approx. 36 moles).
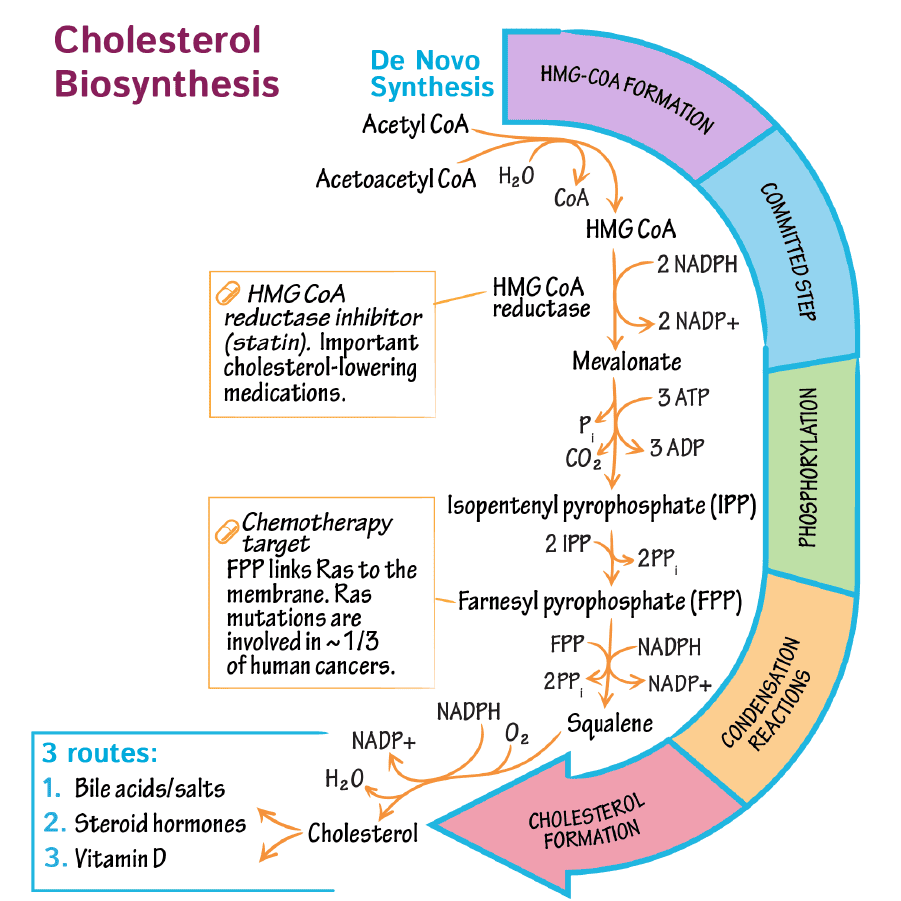
Steps of Cholesterol Biosynthesis: A Detailed Pathway
The complex pathway can be delineated into five principal stages:
Step 1: Formation of HMG-CoA
The synthesis initiates with the condensation of Acetyl-CoA units:
- Two molecules of Acetyl-CoA combine to form Acetoacetyl-CoA (catalyzed by thiolase).
- Acetoacetyl-CoA condenses with a third molecule of Acetyl-CoA to yield β-hydroxy-β-methylglutaryl-CoA (HMG-CoA), catalyzed by HMG-CoA synthase.
It is crucial to note the distinction from ketone body synthesis: the cytosolic HMG-CoA synthase produces HMG-CoA for cholesterol synthesis, while the mitochondrial HMG-CoA synthase participates in ketogenesis. This segregation ensures the pathways operate independently.
Step 2: Conversion of HMG-CoA to Mevalonate
This stage represents the rate-limiting and committed step in cholesterol biosynthesis:
- HMG-CoA is reduced to mevalonate in a reaction catalyzed by HMG-CoA reductase.
- This endoplasmic reticulum-bound enzyme requires two molecules of NADPH.
- HMG-CoA reductase is the primary therapeutic target for statins, a class of drugs that lower plasma cholesterol.
Step 3: Production of Activated Isoprenoid Units
Mevalonate is subsequently processed to generate activated 5-carbon units:
- Mevalonate undergoes a series of three phosphorylation steps, utilizing ATP.
- This is followed by decarboxylation to produce isopentenyl pyrophosphate (IPP), a 5-carbon isoprenoid unit.
- IPP can be isomerized to its structural cousin, dimethylallyl pyrophosphate (DMAPP).
Step 4: Synthesis of Squalene
The activated 5-carbon isoprenoid units are progressively linked:
- DMAPP condenses with IPP to form the 10-carbon geranyl pyrophosphate (GPP).
- GPP condenses with another IPP to yield the 15-carbon farnesyl pyrophosphate (FPP).
- Finally, two molecules of FPP condense head-to-head, mediated by squalene synthase and requiring NADPH, to form the 30-carbon linear molecule squalene.
Step 5: Conversion of Squalene to Cholesterol
The linear squalene molecule undergoes cyclization and a series of modifications:
- Squalene is first oxidized to squalene epoxide by squalene monooxygenase.
- Squalene epoxide undergoes a complex cyclization reaction to form lanosterol, the first true steroid compound in the pathway.
- From lanosterol, a multi-step process involving ~19 enzymatic reactions ensues, involving:
- Demethylation: Removal of three methyl groups, reducing the carbon count from 30 to 27.
- Double Bond Rearrangement: Shifts in the positions of double bonds.
- Reduction: Reduction of a double bond in the side chain.
These precise modifications culminate in the formation of cholesterol.
Regulation of Cholesterol Synthesis
The synthesis of cholesterol is a highly regulated process. The primary point of control is the enzyme HMG-CoA reductase, the rate-limiting step in the pathway. Regulation occurs through several sophisticated mechanisms:
Transcriptional Control (Feedback Inhibition via SREBP Pathway):
- This is the most critical long-term regulatory mechanism.
- When intracellular cholesterol levels are high, they reduce the activity of SREBP-2 (Sterol Regulatory Element-Binding Protein 2).
- This leads to a reduction in the transcription of genes encoding HMG-CoA reductase and the LDL receptor, thus diminishing cholesterol synthesis.
- Conversely, low cellular cholesterol levels activate SREBP-2, promoting gene transcription and increasing both cholesterol synthesis and uptake.
Translational Control:
High concentrations of cholesterol also exert an inhibitory effect on the translation of HMG-CoA reductase mRNA, directly reducing the quantity of enzyme synthesized.
Enzyme Degradation (Proteolysis):
High sterol levels induce a conformational change in the reductase enzyme, making it more susceptible to ubiquitination and subsequent degradation by the proteasome. This shortens the enzyme's lifespan, leading to a quick reduction in its activity.
Covalent Modification (Hormonal Regulation and Energy Status):
HMG-CoA reductase exists in two interconvertible forms:
- Dephosphorylated form: More active.
- Phosphorylated form: Less active (inactive).
Phosphorylation is primarily mediated by AMP-activated protein kinase (AMPK), which is activated when cellular ATP is low. By phosphorylating and inactivating HMG-CoA reductase, AMPK conserves cellular energy.
Hormonal Influence:
- Insulin and Thyroid Hormones: Increase HMG-CoA reductase activity by promoting dephosphorylation (activation).
- Glucagon and Glucocorticoids: Decrease HMG-CoA reductase activity by promoting phosphorylation (inactivation).
Direct Inhibition by Drugs (Statins):
- Drugs such as lovastatin, simvastatin, and atorvastatin are competitive inhibitors of HMG-CoA reductase.
- They structurally resemble HMG-CoA and bind to the active site, blocking its ability to convert HMG-CoA to mevalonate and directly reducing the rate of cholesterol synthesis.
Inhibition by Bile Acids:
Bile acids, which are derivatives of cholesterol, can also contribute to feedback regulation by inhibiting HMG-CoA reductase activity.
Connecting to Cholesterol Transport:
While not a direct regulatory mechanism for synthesis, the major players in cholesterol transport are intrinsically linked to overall cholesterol homeostasis:
- Low-Density Lipoproteins (LDL): Primarily transport cholesterol from the liver to peripheral tissues. Often called "bad cholesterol."
- High-Density Lipoproteins (HDL): Transport excess cholesterol from peripheral tissues back to the liver for excretion or recycling (reverse cholesterol transport). Often called "good cholesterol."
Formation and Excretion of Bile Acids and Bile Salts
Bile acids are a family of steroid acids that represent the major catabolic products of cholesterol in the body. Their primary physiological function is to facilitate the digestion and absorption of dietary fats and fat-soluble vitamins in the small intestine. They also play a crucial role in cholesterol homeostasis by being the principal route for cholesterol excretion.
What is Bile?
Bile is a complex, watery, yellowish-green fluid produced by the liver. It consists of a watery mixture of organic and inorganic compounds.
The quantitatively most important organic components of bile are phosphatidylcholine (lecithin) and conjugated bile salts.
Bile can either pass directly from the liver into the duodenum (the first part of the small intestine) via the common bile duct, or it can be stored and concentrated in the gallbladder when not immediately needed for digestion.
A. Synthesis of Primary Bile Acids
The synthesis of bile acids, known as cholic acid and chenodeoxycholic acid, occurs exclusively in the liver. This multi-step pathway converts the hydrophobic cholesterol molecule into more polar, amphipathic bile acids, making them water-soluble.
Initiation - The Rate-Limiting Step:
The synthesis pathway involves the insertion of hydroxyl groups at specific positions on the steroid structure of cholesterol. The hydrocarbon chain is also shortened by three carbons.
The first and rate-limiting step in bile acid synthesis is the introduction of a hydroxyl group at carbon 7 of cholesterol, forming 7α-hydroxycholesterol.
This reaction is catalyzed by the enzyme cholesterol 7α-hydroxylase (CYP7A1).
CYP7A1 is a cytochrome P450 enzyme, requiring molecular oxygen (O₂) and NADPH.
Regulation: The activity of CYP7A1 is highly regulated. It is inhibited by bile acids (a feedback mechanism) and induced by cholesterol (when cholesterol levels are high). This ensures that bile acid synthesis is responsive to both bile acid demand and cholesterol availability.
Subsequent Reactions:
Following the initial hydroxylation, 7α-hydroxycholesterol undergoes a series of additional modifications. These steps involve:
- Further hydroxylations (e.g., at C-12 to form cholic acid, which is a triol - having three hydroxyl groups).
- Epimerization of the 3β-hydroxyl group to a 3α-hydroxyl group.
- Reduction of the double bond in the B ring.
- Oxidation of the side chain (carbon atoms 24, 25, 26, and 27) and its cleavage to introduce a carboxyl group at C-24, shortening the side chain from 8 to 5 carbons.
Formation of Primary Bile Acids:
These reactions ultimately lead to the formation of the two primary bile acids:
- Cholic acid: (a triol) Has hydroxyl groups at C-3α, C-7α, and C-12α.
- Chenodeoxycholic acid: (a diol) Has hydroxyl groups at C-3α and C-7α.
B. Conjugation of Primary Bile Acids to Form Bile Salts
To significantly improve their ability to emulsify fat and enhance their water solubility, primary bile acids are further modified in the liver through a process called conjugation. They are joined with either the amino acid glycine or taurine.
Mechanism:
The carboxyl group (–COOH) at the end of the bile acid side chain forms an amide bond with the amino group (–NH₂) of glycine or taurine.
This reaction is catalyzed by bile acid-CoA ligase (which activates the bile acid by forming a CoA thioester) and bile acid-CoA:amino acid N-acyltransferase.
Resulting Conjugated Bile Acids (Bile Salts):
This generates the conjugated bile acids:
- Taurocholic acid and Taurochenodeoxycholic acid
- Glycocholic acid and Glycocholic acid
These conjugated forms are all necessary to give bile its essential function in fat digestion.
At physiological pH, these conjugated bile acids exist as anions (negatively charged) due to the low pKa of their conjugates. Therefore, they are referred to as bile salts (e.g., taurocholate, glycocholate). The term "bile salts" specifically refers to these ionized forms.
Physiological Significance of Conjugation:
- Increased Solubility & Emulsification: Conjugation makes bile acids much more soluble and improves their amphipathic nature, crucial for emulsifying dietary fats.
- Effective Detergency: The salts are large, negatively charged ions that are not readily absorbed by passive diffusion in the upper region of the small intestine, ensuring sustained activity.
- PKA Reduction: Conjugation lowers the pKa of the bile acids, ensuring that they remain ionized (charged) even in the acidic environment of the upper small intestine.
C. Enterohepatic Circulation of Bile Salts
Bile salts are essential for fat digestion, but the body has a highly efficient system to conserve them rather than synthesizing new ones for every meal. This system is called the enterohepatic circulation.
Secretion:
Synthesized and conjugated bile salts are secreted from the liver, stored in the gallbladder, and released into the duodenum after a fatty meal.
Function in Small Intestine:
In the duodenum and jejunum, bile salts emulsify dietary fats and form mixed micelles.
Reabsorption:
A remarkable 95% of bile salts are reabsorbed in the ileum (the final part of the small intestine). This reabsorption occurs via a specialized, active transport system known as the apical sodium-dependent bile acid transporter (ASBT) in the ileal enterocytes. Some passive reabsorption of unconjugated bile acids can also occur in the jejunum and colon.
Portal Vein Transport:
Once reabsorbed, bile salts enter the portal venous blood and are transported back to the liver, mostly bound to albumin.
Hepatic Uptake:
The liver efficiently extracts the bile salts from the portal blood via specific transporters.
Recycling:
The liver then re-secretes these reabsorbed bile salts into the bile, completing the circulation. This cycle can occur 4-12 times a day.
D. Formation and Excretion of Secondary Bile Acids
Not all bile acids are reabsorbed directly. Bacterial action in the gut leads to the formation of secondary bile acids.
Bacterial Deconjugation:
As bile salts travel through the colon, intestinal bacteria can deconjugate them, removing glycine or taurine.
Bacterial Dehydroxylation:
These free primary bile acids can then be further metabolized by gut bacteria, specifically undergoing 7α-dehydroxylation. This results in the formation of secondary bile acids:
- Deoxycholic acid (from cholic acid)
- Lithocholic acid (from chenodeoxycholic acid)
Fate of Secondary Bile Acids:
Most secondary bile acids are also reabsorbed and return to the liver. In the liver, deoxycholic acid can be re-conjugated. Lithocholic acid, which is less soluble, is often sulfonated before being secreted back into bile, which aids in its excretion.
E. Excretion of Cholesterol
The excretion of cholesterol from the body primarily occurs via two main routes:
- Conversion to Bile Acids and Excretion: A small fraction of bile salts (about 5%, or 0.2-0.6 grams per day) is not reabsorbed and is instead excreted in the feces. This represents a net loss and is the most significant route for cholesterol elimination.
- Direct Secretion of Unesterified Cholesterol into Bile: The liver can also secrete free, unesterified cholesterol directly into the bile. A portion of this is reabsorbed, but a significant amount is excreted. If the concentration of cholesterol in bile exceeds the solubilizing capacity of bile salts, it can precipitate, leading to cholesterol gallstones.
Synthesis of Steroid Hormones
Cholesterol is not merely a structural component of cell membranes or a precursor for bile acids; it is also the obligate precursor for all steroid hormones. These powerful signaling molecules regulate a vast array of physiological processes, including metabolism, inflammation, immune responses, salt and water balance, sexual development, and reproduction.
A. General Pathway for Steroid Hormone Synthesis
The synthesis of all steroid hormones follows a common, fundamental pathway that begins with cholesterol. This process primarily occurs in the mitochondria and endoplasmic reticulum of steroidogenic tissues.
Tissue-Specific Synthesis:
While virtually all cells contain cholesterol, steroid hormone synthesis is restricted to specialized endocrine tissues, including:
- Adrenal cortex: Produces glucocorticoids, mineralocorticoids, and some adrenal androgens.
- Gonads (Testes and Ovaries): Produce androgens, estrogens, and progestins.
- Placenta: Produces estrogens and progestins during pregnancy.
Rate-Limiting Step: Transport into Mitochondria:
- The first and rate-limiting step is the transport of cholesterol from the cytoplasm into the inner mitochondrial membrane.
- This transport is mediated by the Steroidogenic Acute Regulatory protein (StAR). StAR's activity is rapidly regulated by tropic hormones (e.g., ACTH, LH).
Initial Conversion: Cholesterol to Pregnenolone:
- Once inside the inner mitochondrial membrane, cholesterol is converted to pregnenolone.
- This is catalyzed by the cholesterol desmolase complex, also known as P450scc (cytochrome P450 side-chain cleavage enzyme), which requires NADPH and O₂.
- Pregnenolone is the universal precursor for all other steroid hormones.
B. Major Classes of Steroid Hormones
From pregnenolone, the pathway diverges. The specific hormones produced depend on the enzymatic machinery present in the particular tissue.
Progestins (C21 Steroids):
- Progesterone is derived directly from pregnenolone.
- Function: Crucial for maintaining pregnancy and regulating the menstrual cycle.
- Primary site of synthesis: Ovaries (corpus luteum), adrenal cortex, placenta.
Glucocorticoids (C21 Steroids):
- Cortisol is the primary human glucocorticoid.
- Pathway: Pregnenolone → Progesterone → ... → Cortisol.
- Function: Regulates metabolism, suppresses immune responses, reduces inflammation, and helps adapt to stress.
- Primary site of synthesis: Adrenal cortex (zona fasciculata).
Mineralocorticoids (C21 Steroids):
- Aldosterone is the most potent human mineralocorticoid.
- Pathway: Pregnenolone → Progesterone → ... → Aldosterone.
- Function: Regulates electrolyte balance by promoting sodium reabsorption and potassium excretion, thus influencing blood pressure.
- Primary site of synthesis: Adrenal cortex (zona glomerulosa).
Androgens (C19 Steroids):
- Dehydroepiandrosterone (DHEA), Androstenedione, and Testosterone are key androgens.
- Function: Responsible for the development of male secondary sexual characteristics and libido in both sexes.
- Primary site of synthesis: Testes, adrenal cortex, ovaries.
Estrogens (C18 Steroids):
- Estradiol is the most potent and abundant human estrogen.
- Pathway: Estrogens are synthesized from androgens (testosterone or androstenedione) through a reaction catalyzed by the enzyme aromatase.
- Function: Responsible for the development of female secondary sexual characteristics and regulation of the menstrual cycle.
- Primary site of synthesis: Ovaries, placenta, adipose tissue, testes (to a lesser extent).
C. Regulation of Steroid Hormone Synthesis
The synthesis is tightly regulated by the hypothalamic-pituitary-adrenal/gonadal axes.
- Tropic Hormones: Specific peptide hormones from the anterior pituitary stimulate target endocrine glands:
- Adrenocorticotropic hormone (ACTH): Stimulates the adrenal cortex (cortisol).
- Luteinizing hormone (LH): Stimulates testosterone production in testes and progesterone in ovaries.
- Follicle-stimulating hormone (FSH): Stimulates estrogen production by ovarian follicles.
- Feedback Inhibition: High levels of circulating steroid hormones typically exert negative feedback on the hypothalamus and pituitary gland.
- Enzyme Specificity: The expression and activity of specific steroidogenic enzymes (e.g., 21-hydroxylase, aromatase) within different tissues dictate which hormones are produced. Genetic deficiencies can lead to disorders like congenital adrenal hyperplasia.
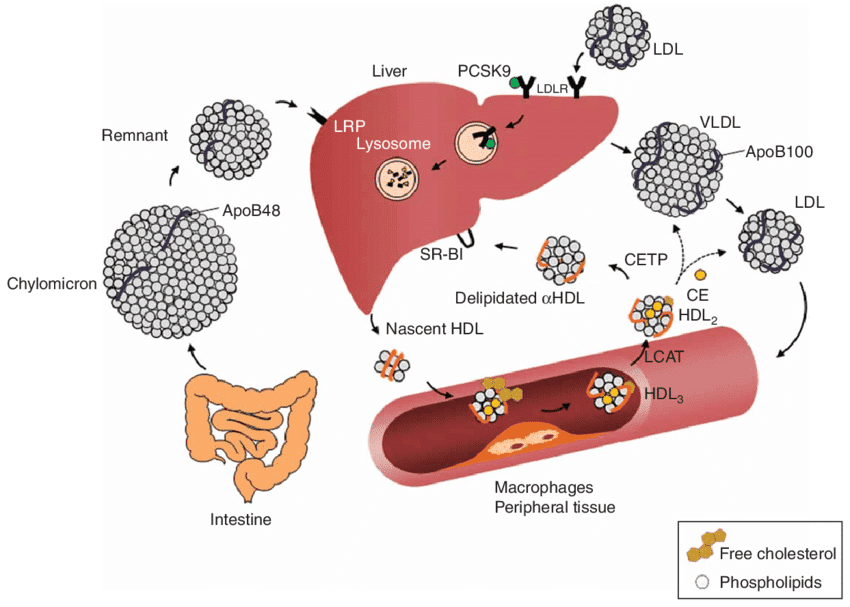
Transport of Cholesterol by Lipoproteins
Cholesterol, being a lipid, is largely insoluble in the aqueous environment of blood plasma. To be efficiently transported between tissues for synthesis, utilization, and excretion, cholesterol (along with other lipids like triglycerides and phospholipids) is packaged into complex particles called lipoproteins. These molecular transporters have a hydrophilic exterior and a hydrophobic core, allowing them to carry lipids through the bloodstream.
Cholesteryl ester in the diet is hydrolyzed to cholesterol, which is then absorbed by the intestine together with dietary unesterified cholesterol and other lipids. It is then incorporated into chylomicrons.
Ninety-five percent of the chylomicron cholesterol is delivered to the liver in chylomicron remnants. Most of the cholesterol secreted by the liver in VLDL is retained during the formation of IDL and ultimately LDL, which is taken up by the LDL receptor in liver and extrahepatic tissues.
A. Structure of Lipoproteins
All lipoproteins share a common structural organization:
- Hydrophobic Core: Contains the most water-insoluble lipids:
- Triglycerides (TGs)
- Cholesteryl esters (CEs)
- Hydrophilic Shell: Surrounds the core and allows the particle to be soluble in blood:
- Phospholipids
- Free (unesterified) Cholesterol
- Apolipoproteins: Proteins integral to the shell that provide structural integrity, act as enzyme cofactors (e.g., ApoC-II), and serve as ligands for cell surface receptors (e.g., ApoB-100).
B. Classes of Lipoproteins
Lipoproteins are classified based on their density (more lipid = less dense). From largest/least dense to smallest/most dense, the main classes are:
Chylomicrons (CM):
- Origin: Intestine.
- Primary Lipid: Dietary triglycerides (>80%).
- Main Apolipoproteins: ApoB-48, ApoC-II, ApoE.
- Function: Transport dietary (exogenous) lipids from the intestine to peripheral tissues and then to the liver.
- Metabolism: Nascent chylomicrons acquire ApoC-II and ApoE from HDL. ApoC-II activates lipoprotein lipase (LPL) in capillaries, which hydrolyzes TGs. The resulting chylomicron remnants, enriched in cholesteryl esters and ApoE, are taken up by the liver.
Very Low-Density Lipoproteins (VLDL):
- Origin: Liver.
- Primary Lipid: Endogenously synthesized triglycerides (50-60%).
- Main Apolipoproteins: ApoB-100, ApoC-II, ApoE.
- Function: Transport endogenously synthesized lipids from the liver to peripheral tissues.
- Metabolism: Similar to chylomicrons, LPL hydrolyzes TGs from VLDL. As VLDL loses TGs, it becomes smaller and denser, first forming intermediate-density lipoproteins (IDL).
Intermediate-Density Lipoproteins (IDL):
- Origin: Formed from VLDL after triglyceride hydrolysis.
- Primary Lipid: Roughly equal amounts of TGs and cholesteryl esters.
- Main Apolipoproteins: ApoB-100, ApoE.
- Function: An intermediate in the conversion of VLDL to LDL. About half are taken up by the liver, and the rest are metabolized to LDL.
Low-Density Lipoproteins (LDL):
- Origin: Primarily from the catabolism of VLDL and IDL.
- Primary Lipid: Cholesteryl esters (~45-50%).
- Main Apolipoprotein: ApoB-100.
- Function: Transport cholesterol from the liver to peripheral tissues. Often called "bad cholesterol."
- Metabolism: Cells needing cholesterol take up LDL particles via the LDL receptor, which specifically recognizes ApoB-100.
High-Density Lipoproteins (HDL):
- Origin: Liver and intestine.
- Primary Lipid: Relatively rich in protein (~50%) and phospholipids.
- Main Apolipoproteins: ApoA-I (major), ApoC-II, ApoE.
- Function: Often called "good cholesterol." HDL plays a crucial role in reverse cholesterol transport, collecting excess cholesterol from peripheral cells and transporting it back to the liver. It also serves as a reservoir for ApoC-II and ApoE.
- Metabolism: Nascent HDL particles acquire free cholesterol from peripheral cells. This cholesterol is esterified by lecithin-cholesterol acyltransferase (LCAT). Mature HDL can exchange cholesteryl esters for TGs with other lipoproteins (via CETP) and is finally taken up by the liver (via SR-B1).
C. Exogenous vs. Endogenous Pathways of Lipid Transport
- Exogenous Pathway: Deals with dietary lipids. Chylomicrons are the key lipoproteins.
- Endogenous Pathway: Deals with endogenously synthesized lipids. VLDL, IDL, and LDL are the main players for distribution, while HDL mediates reverse transport.
D. Role of Key Enzymes and Receptors
- Lipoprotein Lipase (LPL): Hydrolyzes TGs in chylomicrons and VLDL in capillaries.
- Hepatic Lipase (HL): Hydrolyzes TGs and phospholipids in IDL and HDL.
- Lecithin-Cholesterol Acyltransferase (LCAT): Esterifies free cholesterol to cholesteryl esters within HDL.
- Cholesteryl Ester Transfer Protein (CETP): Facilitates the exchange of cholesteryl esters and TGs between lipoproteins.
- LDL Receptor: Mediates the uptake of LDL (and IDL) via recognition of ApoB-100.
- Scavenger Receptor Class B Type 1 (SR-B1): Mediates selective uptake of cholesteryl esters from HDL by the liver.
Regulation of Cholesterol Synthesis, Metabolism, and Transport
Maintaining cholesterol homeostasis is critical. The body employs an intricate network of regulatory mechanisms, with the primary point of control being the enzyme HMG-CoA reductase.
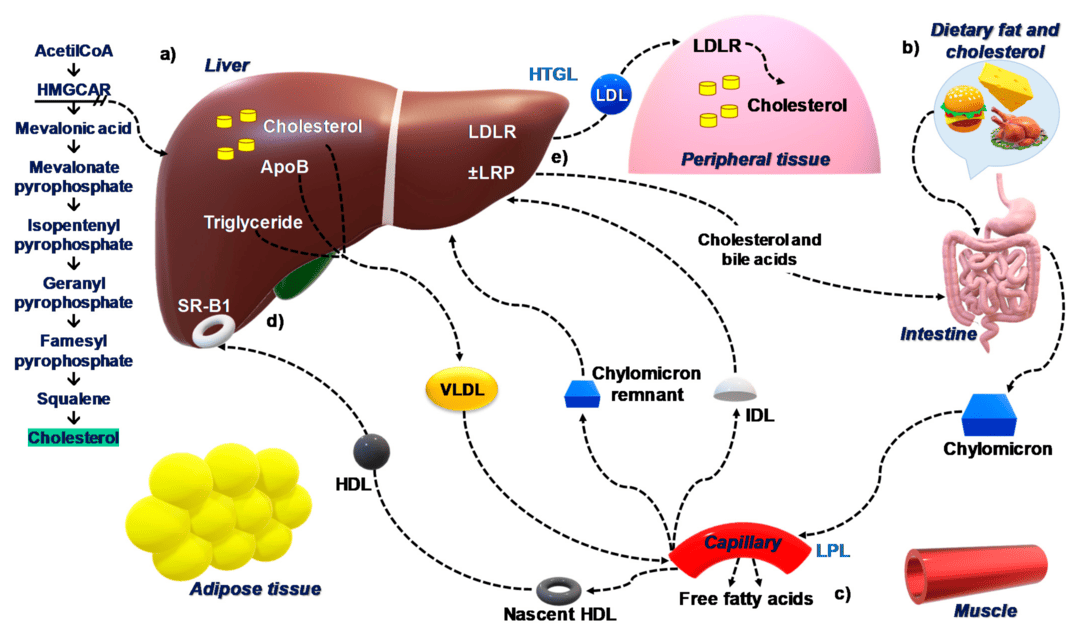
A. Regulation of Cholesterol Synthesis
Transcriptional Control (Gene Expression):
- SREBP-2 (Sterol Regulatory Element-Binding Protein-2): This is the master regulator.
- Low Cellular Cholesterol: SREBP-2 is cleaved, moves to the nucleus, and binds to Sterol Regulatory Elements (SREs). This activates transcription of genes for HMG-CoA reductase and the LDL receptor, increasing both cholesterol production and uptake.
- High Cellular Cholesterol: SREBP-2 is retained in the ER, and transcription is suppressed.
Post-Translational Control (Enzyme Activity & Degradation):
- Phosphorylation (Covalent Modification):
- AMP-activated protein kinase (AMPK): When cellular energy is low, AMPK phosphorylates and inactivates HMG-CoA reductase.
- Insulin: Promotes dephosphorylation, activating the enzyme.
- Glucagon: Promotes phosphorylation, inactivating the enzyme.
- Proteasomal Degradation: High levels of cholesterol cause HMG-CoA reductase to be degraded by the proteasome.
B. Regulation of Bile Acid Synthesis
- Cholesterol 7α-hydroxylase (CYP7A1): This is the rate-limiting enzyme.
- Negative Feedback by Bile Acids: High levels of bile acids returning to the liver inhibit the transcription of the CYP7A1 gene.
- Positive Regulation by Cholesterol: Increased cholesterol induces CYP7A1 activity, ensuring excess cholesterol can be eliminated.
C. Regulation of Cholesterol Transport
LDL Receptor Regulation:
- The number of LDL receptors on the cell surface is the primary determinant of LDL clearance from the blood.
- High Cellular Cholesterol: Downregulates LDL receptor synthesis (via the SREBP-2 mechanism).
- Low Cellular Cholesterol: Upregulates LDL receptor synthesis.
- PCSK9: This enzyme promotes the degradation of the LDL receptor. Inhibitors of PCSK9 are a new class of drugs for lowering LDL.
Dietary and Hormonal Factors:
- Saturated and Trans Fats: Increase LDL cholesterol.
- Soluble Fiber: Lowers LDL cholesterol by interfering with bile acid reabsorption.
- Insulin: Promotes cholesterol synthesis.
- Estrogens: Tend to lower LDL and raise HDL.
D. Importance of Cholesterol Homeostasis
The tight regulation is vital because both insufficient (hypocholesterolemia) and excessive (hypercholesterolemia) cholesterol levels are detrimental. Excess cholesterol, particularly carried by LDL, can lead to its deposition in arterial walls, causing atherosclerosis.
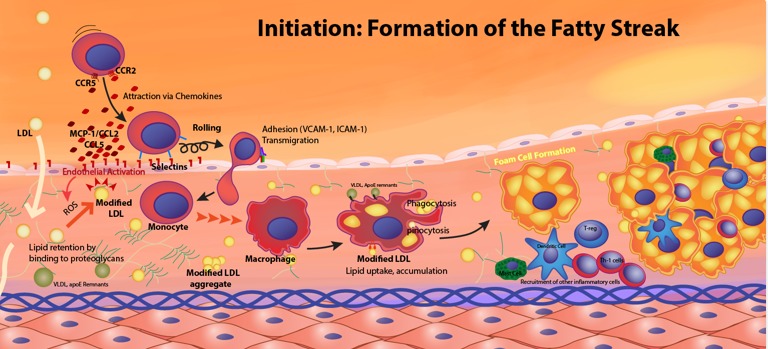
Clinical Significance of Cholesterol: Atherosclerosis
Atherosclerosis is a chronic inflammatory disease characterized by the buildup of fatty plaques within the arterial walls, leading to hardening and narrowing of the arteries.
A. Hypercholesterolemia and Dyslipidemia
- Hypercholesterolemia: Abnormally high levels of cholesterol in the blood.
- Dyslipidemia: A broader term for abnormal lipid levels, including high LDL ("bad cholesterol"), low HDL ("good cholesterol"), and high triglycerides.
B. The Role of Lipoproteins in Atherosclerosis
- Low-Density Lipoprotein (LDL) - The Primary Atherogenic Particle:
- High LDL levels lead to its infiltration into the arterial wall, where it becomes oxidized (oxLDL).
- Macrophages ingest oxLDL in an uncontrolled manner, transforming into foam cells.
- Accumulations of foam cells form fatty streaks, the earliest lesions of atherosclerosis.
- High-Density Lipoprotein (HDL) - The Anti-Atherogenic Particle:
- HDL is crucial for reverse cholesterol transport, removing excess cholesterol from arterial walls and transporting it to the liver.
- HDL also has antioxidant and anti-inflammatory properties. High HDL levels are associated with reduced CVD risk.
C. Pathogenesis of Atherosclerosis
The development of atherosclerotic plaques is a multi-stage process:
- Endothelial Dysfunction: Damage to the artery's inner lining.
- LDL Infiltration and Oxidation: LDL enters the arterial wall and becomes oxidized.
- Macrophage Recruitment and Foam Cell Formation: Immune cells are recruited and become lipid-laden foam cells.
- Smooth Muscle Cell Migration and Proliferation: These cells contribute to the bulk of the plaque.
- Fibrous Cap Formation: A cap of collagen and smooth muscle cells forms over the lipid core.
- Plaque Progression and Complications: Over time, plaques can grow, develop a necrotic core, and become unstable. Plaque rupture exposes the core to blood, leading to rapid thrombus (blood clot) formation, which can cause a heart attack or stroke.
D. Management of Dyslipidemia and CVD Risk
- Lifestyle Modifications: Diet, physical activity, weight management, and smoking cessation.
- Pharmacological Interventions:
- Statins (HMG-CoA Reductase Inhibitors): The most effective drugs for lowering LDL.
- Ezetimibe: Inhibits cholesterol absorption.
- PCSK9 Inhibitors: Prevent LDL receptor degradation.
- Bile Acid Sequestrants: Increase cholesterol excretion via bile acids.
Comprehensive Summary: Cholesterol Metabolism and Its Clinical Significance
Cholesterol is a vital lipid molecule with diverse metabolic fates, playing crucial roles in maintaining cellular structure and serving as a precursor for essential biomolecules. Its metabolism is tightly regulated, and dysregulation can lead to significant health consequences, particularly cardiovascular disease.
I. Metabolic Fates of Cholesterol
Cholesterol is the obligate precursor for several critical compounds:
- Steroid Hormones: Progestins, Glucocorticoids, Mineralocorticoids, Androgens, and Estrogens.
- Bile Acids [Salts]: Catabolized in the liver to form cholic and chenodeoxycholic acid, which are conjugated to form bile salts for fat digestion. This is the main route of cholesterol excretion via the stool.
- Vitamin D3: A precursor for the synthesis of Vitamin D3 upon exposure to UV light in the skin.
II. Transport of Cholesterol: Lipoproteins
Cholesterol is transported in lipoproteins, classified by density:
- Chylomicrons (CM): Transport dietary lipids from the intestine.
- VLDL: Transport endogenous lipids from the liver.
- IDL: Transitional particles.
- LDL ("bad cholesterol"): Delivers cholesterol to peripheral tissues.
- HDL ("good cholesterol"): Collects excess cholesterol and returns it to the liver (Reverse Cholesterol Transport).
III. Regulation of Cholesterol Metabolism
Regulation involves transcriptional control (via SREBP-2), post-translational control (phosphorylation of HMG-CoA reductase by AMPK), feedback inhibition, and LDL receptor modulation.
IV. Clinical Significance of Cholesterol
A. Atherosclerosis:
Increased plasma cholesterol, particularly elevated LDL, is a major risk factor. High LDL leads to cholesterol accumulation in arteries, oxidation, foam cell formation, and plaque development, increasing the risk of heart attacks and strokes. HDL is protective.
B. Variation of Serum Cholesterol Levels (Normal: 150-220 mg/dl):
- Hypercholesterolemia (High): Associated with diabetes mellitus, nephrotic syndrome, hypothyroidism, obstructive jaundice, familial hypercholesterolemia.
- Hypocholesterolemia (Low): Associated with hyperthyroidism, malnutrition, malabsorption, and anemia.
C. Gallstones (Cholelithiasis):
Primarily composed of crystalline cholesterol. Linked to the liver secreting bile that is saturated with cholesterol, coupled with abnormally low levels of bile salts and lecithin, which leads to cholesterol precipitation.
Biochemistry: Cholesterol Metabolism
Test your knowledge with these 40 questions.
Cholesterol Metabolism Quiz
Question 1/40
Quiz Complete!
Here are your results, .
Your Score
38/40
95%
Integrated Metabolism and Fuel Homeostasis
Integrated Metabolism & : Fuel Homeostasis
Integrated Metabolism and Fuel Homeostasis
Fuel Homeostasis refers to the dynamic equilibrium and finely tuned regulation of energy substrates (glucose, fatty acids, ketone bodies, amino acids) in the body. Its primary goal is to ensure a continuous and adequate supply of fuel to all tissues, particularly the brain, under varying physiological conditions.
It is crucial for survival, allowing the body to adapt to fluctuations in nutrient availability and energy demand. Disruptions lead to metabolic diseases like diabetes, obesity, and metabolic syndrome.
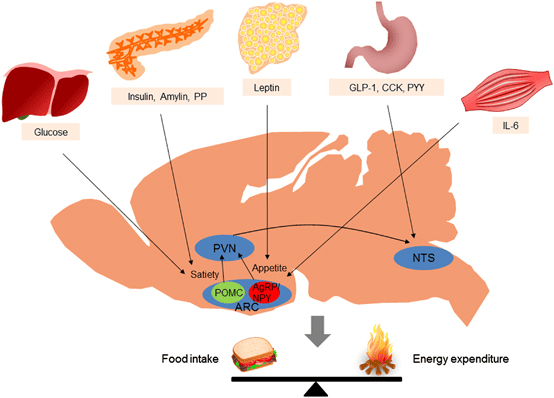
Key Metabolic Organs and Their Specialized Roles
The human body is a highly integrated system where different organs specialize in fuel storage, production, and utilization.
Liver (Hepatocytes): The Metabolic Hub
- Glucose Homeostasis: Central to maintaining blood glucose levels.
- Fed State: Takes up excess glucose, converting it to glycogen (glycogenesis) or fatty acids (lipogenesis).
- Fasting State: Releases glucose into the blood via glycogenolysis and gluconeogenesis.
- Lipid Metabolism: Site of de novo fatty acid synthesis, cholesterol synthesis, and VLDL assembly. It is also the primary site for ketogenesis during prolonged fasting.
- Amino Acid Metabolism: Site for amino acid uptake, protein synthesis, deamination, and the urea cycle.
- Lack of Ketone Body Utilization: Cannot use ketone bodies as fuel due to the absence of thiophorase.
Adipose Tissue (Adipocytes): The Energy Storehouse
- Storage: Primary site for the long-term storage of energy as triacylglycerols (TAGs).
- Mobilization: Releases free fatty acids and glycerol via lipolysis during fasting.
- Synthesis: Can synthesize TAGs from fatty acids and glycerol-3-phosphate.
- Endocrine Organ: Produces adipokines (e.g., leptin, adiponectin).
Skeletal Muscle (Myocytes): The Major Energy Consumer
- Fuel Utilization: Highly versatile; can use glucose, fatty acids, and ketone bodies.
- Glycogen Storage: Stores significant amounts of glycogen, but only for its own use (lacks glucose-6-phosphatase).
- Fatty Acid Oxidation: Major site for fatty acid oxidation, particularly during exercise and fasting.
- Protein Reservoir: A significant protein reserve that can be catabolized during prolonged fasting.
Brain (Neurons and Glial Cells): Obligate Glucose User, Adaptable in Fasting
- Primary Fuel: Under normal conditions, relies almost exclusively on glucose.
- Adaptation in Fasting: During prolonged fasting, the brain can adapt to utilize ketone bodies as a significant alternative fuel, sparing muscle protein.
- Cannot use Fatty Acids: Fatty acids cannot cross the blood-brain barrier.
Pancreas (Islets of Langerhans): The Endocrine Regulator
- Insulin (Beta Cells): Released in response to high blood glucose (fed state). Promotes fuel storage.
- Glucagon (Alpha Cells): Released in response to low blood glucose (fasting state). Promotes fuel mobilization.
- Somatostatin (Delta Cells): Inhibits secretion of both insulin and glucagon.
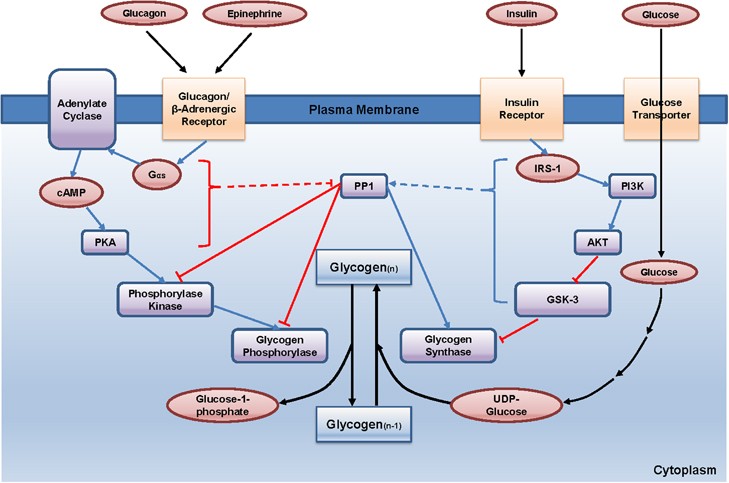
Major Hormones Orchestrating Fuel Homeostasis
These hormones act synergistically and antagonistically to maintain metabolic balance.
Insulin (Anabolic Hormone)
- Source: Pancreatic β-cells.
- Stimulus: High blood glucose, amino acids.
- Overall Effect: Promotes fuel storage; lowers blood glucose.
- Actions:
- Liver: Increases glycogenesis, lipogenesis; inhibits glycogenolysis, gluconeogenesis.
- Muscle: Increases glucose uptake (via GLUT4), glycogenesis, protein synthesis.
- Adipose: Increases glucose uptake (via GLUT4), TAG synthesis; inhibits lipolysis (inhibits HSL).
Glucagon (Catabolic Hormone)
- Source: Pancreatic α-cells.
- Stimulus: Low blood glucose.
- Overall Effect: Promotes fuel mobilization; raises blood glucose.
- Actions (primarily liver): Increases glycogenolysis, gluconeogenesis, ketogenesis; inhibits glycogenesis, lipogenesis.
Catecholamines (Epinephrine, Norepinephrine - Stress Hormones)
- Source: Adrenal medulla, sympathetic nervous system.
- Stimulus: Stress, exercise, hypoglycemia.
- Overall Effect: "Fight or flight"; rapid mobilization of energy stores.
- Actions:
- Liver & Muscle: Increases glycogenolysis.
- Adipose: Potent activator of HSL, promoting lipolysis.
Cortisol (Glucocorticoid - Stress Hormone)
- Source: Adrenal cortex.
- Stimulus: Stress (chronic), low blood glucose.
- Overall Effect: Sustained glucose production; catabolic.
- Actions:
- Liver: Increases gluconeogenesis (by increasing enzyme synthesis).
- Muscle: Increases protein breakdown.
- Adipose: Increases lipolysis.
- Decreases peripheral glucose utilization.
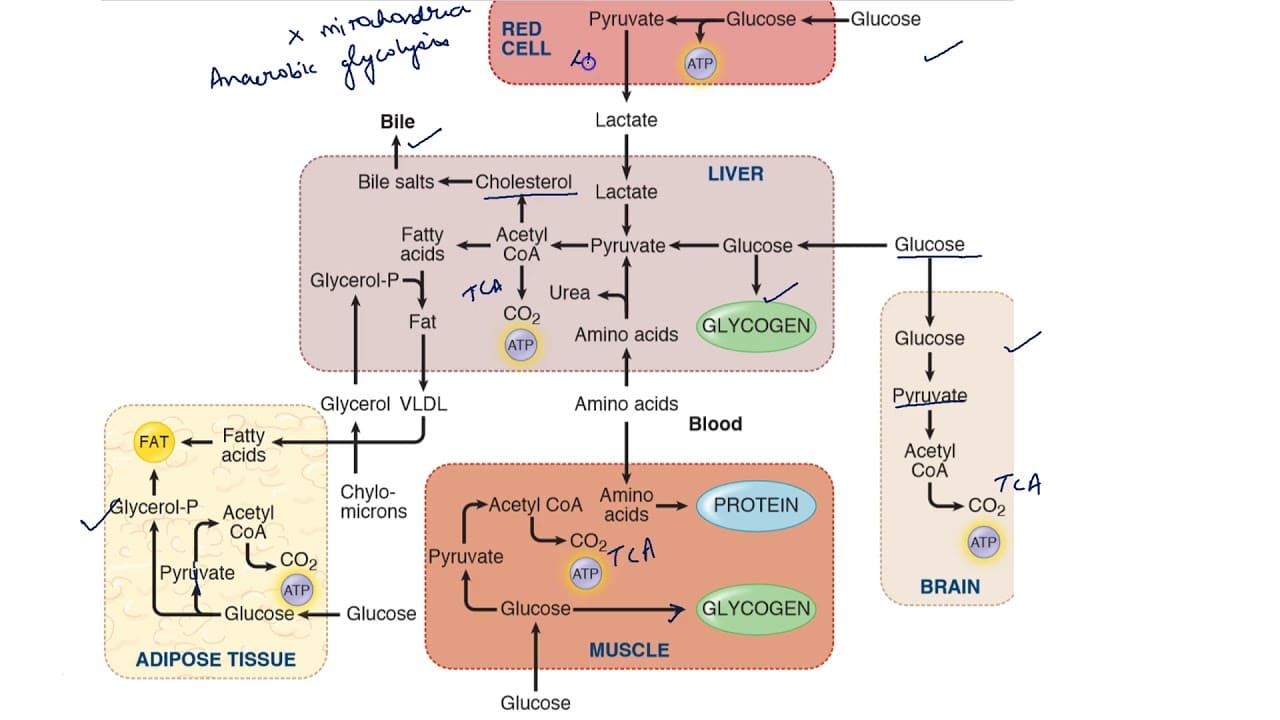
The Fed State (Post-prandial Metabolism)
The fed state is characterized by nutrient absorption from the gastrointestinal tract, leading to elevated levels of glucose, amino acids, and triacylglycerols in the blood. The body's primary response is to store these excess nutrients and utilize glucose as the main fuel.
A. High Insulin:Glucagon Ratio:
- Following a meal, especially one rich in carbohydrates, blood glucose levels rise.
- This rise in glucose stimulates the pancreatic β-cells to release insulin.
- Simultaneously, high glucose inhibits the pancreatic α-cells, suppressing glucagon secretion.
- The resulting high insulin:glucagon ratio orchestrates the anabolic (storage) and glucose-utilizing responses.
B. Carbohydrate Metabolism: Glucose as the Primary Fuel and for Storage
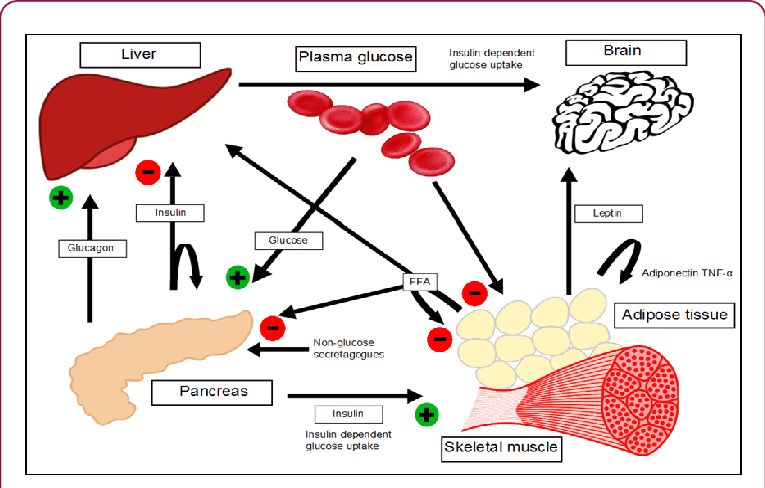
Tissue-Specific Glucose Uptake and Utilization:
Liver:
- High Priority Uptake: Glucose enters hepatocytes via GLUT2 transporters.
- Phosphorylation: Glucokinase rapidly phosphorylates glucose to Glucose-6-Phosphate, trapping it inside.
- Glycogenesis (Glycogen Synthesis): G6P is directed towards glycogen synthesis. Insulin activates glycogen synthase.
- Glycolysis and Pyruvate Oxidation: Excess G6P enters glycolysis, and the resulting pyruvate is converted to Acetyl-CoA.
- Lipogenesis (Fatty Acid Synthesis): When energy and glycogen stores are full, Acetyl-CoA is channeled into de novo fatty acid synthesis. Insulin stimulates this process by activating Acetyl-CoA Carboxylase (ACC).
- VLDL Synthesis: Newly synthesized fatty acids are esterified to form TAGs, which are packaged into Very-Low-Density Lipoproteins (VLDL) and secreted into the bloodstream.
Adipose Tissue (Adipocytes):
- Insulin-Dependent Glucose Uptake: Insulin stimulates the translocation of GLUT4 transporters to the cell membrane.
- Glycerol-3-Phosphate Production: Glucose undergoes glycolysis to produce glycerol-3-phosphate, essential for esterifying fatty acids into TAGs.
- Fatty Acid Uptake: Adipose tissue takes up fatty acids from chylomicrons and VLDL via the action of Lipoprotein Lipase (LPL), which is activated by insulin.
Skeletal Muscle:
- Insulin-Dependent Glucose Uptake: Insulin stimulates GLUT4 translocation, increasing glucose uptake.
- Glycogenesis: Muscle cells synthesize glycogen for their own energy reserves.
- Glycolysis and Oxidation: Glucose is used as a primary fuel source for ATP production.
Brain:
- Insulin-Independent Glucose Uptake: Glucose uptake occurs via GLUT1 and GLUT3 transporters, ensuring a constant supply.
- High Glucose Utilization: The brain consumes a significant amount of glucose (about 120g/day).
C. Lipid Metabolism: Storage and Transport
- Dietary Fat Absorption and Chylomicron Formation: Dietary TAGs are hydrolyzed, absorbed, and then re-esterified within enterocytes. These TAGs are packaged into chylomicrons and released into the lymph and then the bloodstream.
- Chylomicron Metabolism: As chylomicrons circulate, their TAGs are hydrolyzed by Lipoprotein Lipase (LPL), an enzyme activated by insulin. This promotes the uptake of fatty acids into adipose tissue (for storage) and muscle (for use).
- Hepatic VLDL Production: As mentioned, the liver converts excess glucose into fatty acids, which are packaged as TAGs into VLDL particles and secreted. Like chylomicrons, VLDL TAGs are acted upon by LPL.
D. Amino Acid Metabolism: Protein Synthesis
- Amino Acid Absorption: Dietary proteins are digested into amino acids and transported to the liver via the portal circulation.
- Tissue-Specific Utilization:
- Liver: Uses amino acids for liver protein synthesis, synthesis of plasma proteins (e.g., albumin), and synthesis of non-protein nitrogenous compounds. Excess amino acids can be deaminated and their carbon skeletons used for energy or lipogenesis.
- Skeletal Muscle: Insulin promotes the uptake of amino acids. The primary fate is protein synthesis, to repair and build muscle mass.
- Other Tissues: Amino acids are taken up for the synthesis of new proteins and other molecules.
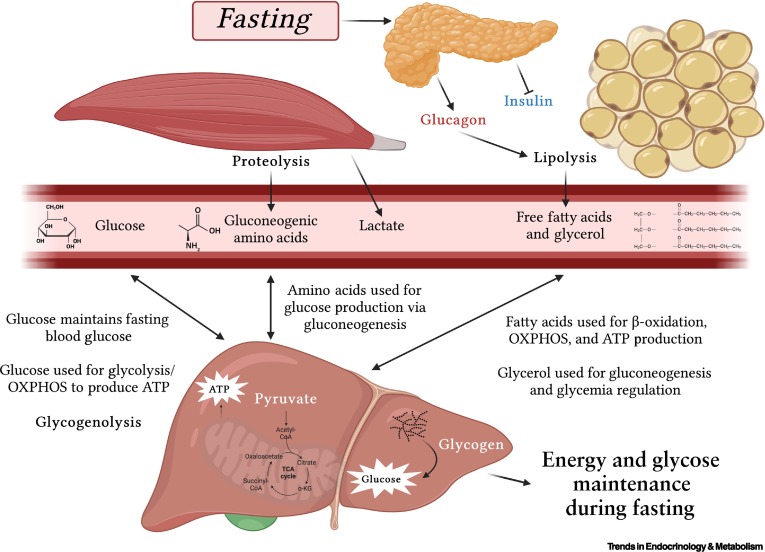
The Fasting State (Early Fasting, Overnight Fast)
The fasting state is characterized by the absence of nutrient intake. The body must now shift from storing fuels to mobilizing its endogenous reserves to maintain a steady supply of energy, especially for the brain. This transition is orchestrated by a low insulin:glucagon ratio.
A. Low Insulin:High Glucagon Ratio:
- As blood glucose levels fall, pancreatic β-cells reduce insulin secretion.
- Concurrently, falling glucose stimulates pancreatic α-cells to increase glucagon secretion.
- The resulting low insulin:high glucagon ratio is the primary signal that triggers the mobilization of stored fuels and the production of new glucose.
- Catecholamines (epinephrine, norepinephrine) and cortisol also play supportive roles.
B. Carbohydrate Metabolism: Glucose Production and Sparing
The primary goal is to maintain blood glucose levels for the brain and other glucose-dependent tissues.
Glycogenolysis (Glycogen Breakdown):
- Liver Glycogen: This is the first line of defense. Hepatic glycogen is rapidly mobilized, stimulated by glucagon and epinephrine. The resulting glucose-6-phosphate is dephosphorylated by glucose-6-phosphatase (present only in the liver) to release free glucose into the blood.
- Duration: Liver glycogen can maintain blood glucose for about 12-24 hours.
- Muscle Glycogen: Muscle glycogen is used only by the muscle itself for energy and cannot be released into the blood.
Gluconeogenesis (New Glucose Synthesis):
- As liver glycogen is depleted, gluconeogenesis becomes the primary mechanism for maintaining blood glucose. This is highly active in the liver.
- Substrates for Gluconeogenesis:
- Lactate: From anaerobic glycolysis in red blood cells.
- Glycerol: Released from the breakdown of TAGs in adipose tissue.
- Glucogenic Amino Acids: Derived from protein breakdown, primarily in skeletal muscle.
- Hormonal Regulation: Glucagon and cortisol are major stimulators.
Glucose Sparing:
To conserve glucose for the brain, other tissues switch their fuel preference to fatty acids and ketone bodies.
C. Lipid Metabolism: Mobilization of Stored Fat
Lipolysis in Adipose Tissue:
- Hormone-Sensitive Lipase (HSL): Glucagon and catecholamines activate HSL in adipocytes via a cAMP-dependent cascade.
- HSL hydrolyzes stored TAGs into free fatty acids (FFAs) and glycerol.
- FFAs: Released into the bloodstream, bind to albumin, and are transported to tissues for β-oxidation.
- Glycerol: Released into the bloodstream and travels to the liver to serve as a substrate for gluconeogenesis.
Fatty Acid Oxidation (β-Oxidation):
- Liver: Becomes a major site of fatty acid oxidation, providing ATP for gluconeogenesis. Excess Acetyl-CoA fuels ketogenesis.
- Skeletal Muscle, Heart, Kidneys: Utilize fatty acids as their primary fuel, thereby sparing glucose.
Ketogenesis (Ketone Body Formation):
- Location: Liver mitochondria.
- Stimulus: High rate of fatty acid oxidation in the liver produces large amounts of Acetyl-CoA. When the TCA cycle is saturated (due to OAA being diverted for gluconeogenesis), the excess Acetyl-CoA is diverted to ketone body synthesis.
- Products: Acetoacetate and β-hydroxybutyrate.
- Purpose: Ketone bodies are water-soluble fuels that can be transported to extrahepatic tissues, particularly the brain, muscle, and heart.
D. Amino Acid Metabolism: Protein Breakdown for Glucose Production
Protein Breakdown in Muscle:
- As fasting continues, skeletal muscle protein becomes a significant source of amino acids for gluconeogenesis. Cortisol promotes this breakdown.
- Glucogenic Amino Acids: Released into the bloodstream and transported to the liver (e.g., alanine, glutamine).
- Alanine Cycle (Cahill Cycle): Pyruvate in muscle is transaminated to alanine, which travels to the liver. In the liver, alanine is converted back to pyruvate for gluconeogenesis.
- Glutamine: Plays a major role in transporting amino groups from muscle to the liver and kidneys.
Urea Cycle:
The amino groups removed from amino acids are converted to ammonia, which is detoxified in the liver via the urea cycle, producing urea for excretion. The rate of the urea cycle increases during fasting.
In summary, the early fasting state is a period of catabolism driven by a low insulin:glucagon ratio. The body prioritizes maintaining blood glucose through glycogenolysis and gluconeogenesis, while other tissues shift to fatty acid oxidation. Ketone body production begins to ramp up, setting the stage for their increased utilization in prolonged starvation.
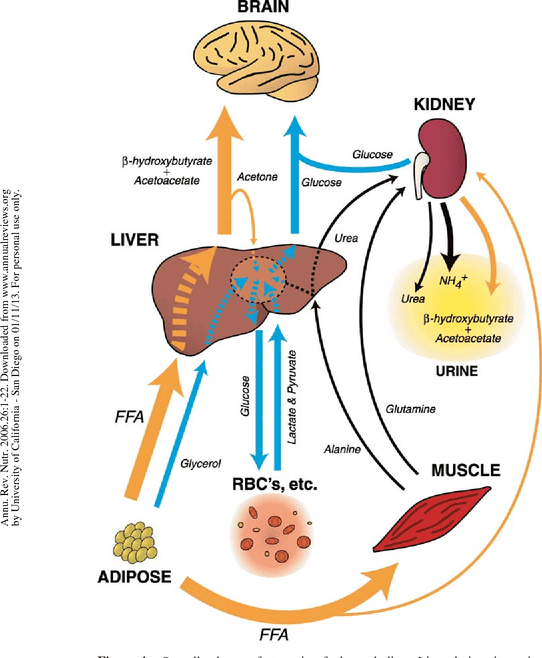
The Starved State (Prolonged Fasting/Starvation)
The starved state represents an extended period of nutrient deprivation, pushing the body's metabolic adaptations to their limits. The primary goals shift to:
- Glucose Sparing: Minimizing the use of glucose by peripheral tissues.
- Protein Sparing: Reducing the breakdown of essential muscle protein.
- Increased Reliance on Fat and Ketone Bodies: Maximizing energy production from abundant fat stores.
A. Continued Low Insulin:High Glucagon Ratio (and elevated stress hormones):
- The hormonal profile established in early fasting persists and may even intensify.
- Insulin levels remain very low, while glucagon, cortisol, and epinephrine remain elevated, reinforcing the catabolic drive.
B. Carbohydrate Metabolism: Extreme Glucose Sparing and Gluconeogenesis Adaptation
Liver Glycogen Depletion:
By the time the starved state is reached (typically after 24-48 hours), liver glycogen stores are almost completely depleted. The body can no longer rely on glycogenolysis.
Sustained Gluconeogenesis (but with changing substrates):
- Gluconeogenesis remains the sole source of new glucose, with kidney gluconeogenesis becoming increasingly significant (up to 40-50% of total production).
- Shift in Substrates:
- Glycerol: Becomes a relatively constant source due to ongoing lipolysis.
- Amino Acids: The rate of muscle protein breakdown decreases significantly after several days/weeks. This is a crucial adaptation to preserve essential lean body mass. The contribution of amino acids to gluconeogenesis gradually declines.
- Lactate: Continues to contribute to a minor extent.
Brain's Adaptation to Ketone Bodies (Glucose Sparing):
- This is the most critical adaptation in the starved state. The brain significantly increases its utilization of ketone bodies (β-hydroxybutyrate and acetoacetate) for energy.
- Mechanism: Ketone bodies cross the blood-brain barrier and are converted back to Acetyl-CoA for the TCA cycle.
- Impact: By shifting to ketone bodies, the brain dramatically reduces its demand for glucose (from ~120g/day to as low as 30-40g/day). This reduces the need for gluconeogenesis from amino acids, thereby sparing muscle protein.
C. Lipid Metabolism: Maximized Mobilization and Ketone Body Production
Maximized Lipolysis:
Lipolysis in adipose tissue continues at a very high rate, providing a continuous supply of fatty acids (for fuel) and glycerol (for gluconeogenesis). Fat stores are the largest energy reserve.
Massive Ketogenesis:
The liver's production of ketone bodies reaches its peak. The high influx of fatty acids, coupled with the low insulin state, promotes maximal β-oxidation and subsequent conversion of Acetyl-CoA into acetoacetate and β-hydroxybutyrate. Blood ketone body levels rise to very high concentrations, serving as the primary fuel for the brain, heart, and skeletal muscle.
D. Amino Acid Metabolism: Protein Sparing and Reduced Nitrogen Excretion
Reduced Muscle Protein Breakdown:
After an initial period of high protein catabolism, the body adapts to significantly reduce muscle protein breakdown. This is directly linked to the brain's increased use of ketone bodies, as less glucose needs to be synthesized from amino acids. This adaptation is critical for long-term survival.
Decreased Urea Production:
As amino acid catabolism decreases, the amount of nitrogen released also decreases. Consequently, the liver's production of urea via the urea cycle significantly declines. This is reflected in a reduced excretion of urea in the urine, signifying the shift to protein-sparing metabolism.
Summary of the Starved State: The starved state is characterized by extreme adaptations aimed at survival. The body shifts almost entirely to fat and ketone body metabolism to preserve its vital protein reserves. The brain becomes a major consumer of ketone bodies, dramatically reducing its glucose requirement and allowing for a significant reduction in the breakdown of muscle protein. This allows individuals to survive for extended periods without food.
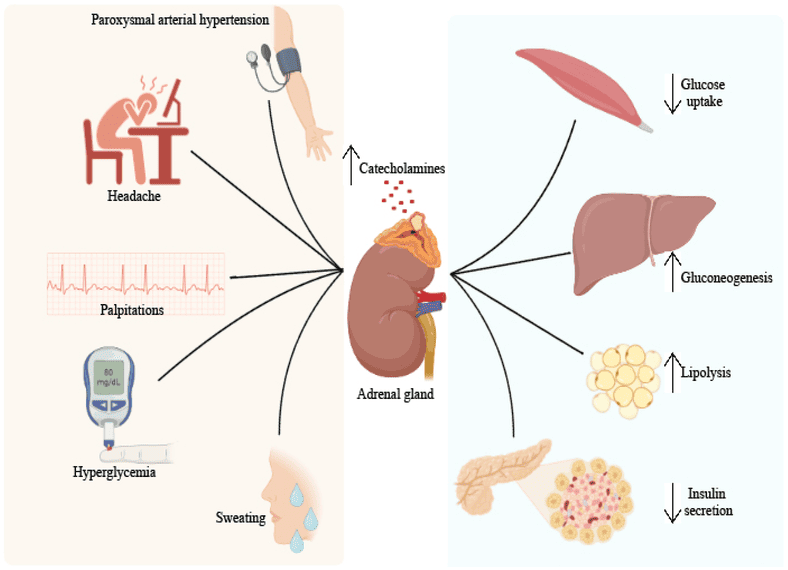
Diabetes Mellitus as a Disorder of Fuel Homeostasis
Diabetes Mellitus (DM) is a group of metabolic diseases characterized by hyperglycemia (high blood glucose) resulting from defects in insulin secretion, insulin action, or both. This chronic hyperglycemia is associated with long-term damage and failure of various organs.
The core problem is a breakdown in the body's ability to regulate glucose, leading to a state that inappropriately resembles a constant "fasted" or even "starved" state in some tissues, despite abundant glucose in the blood.
A. Overview of Types of Diabetes:
Type 1 Diabetes Mellitus (T1DM): Absolute Insulin Deficiency
- Cause: Autoimmune destruction of the pancreatic β-cells, leading to an absolute deficiency of insulin production.
- Onset: Typically in childhood or adolescence.
- Metabolic State: Resembles a perpetual, severe starved state because glucose cannot enter insulin-dependent cells.
Type 2 Diabetes Mellitus (T2DM): Insulin Resistance with Relative Insulin Deficiency
- Cause: A combination of insulin resistance (target cells fail to respond to insulin) and progressive pancreatic β-cell dysfunction.
- Onset: Typically in adulthood, but increasingly seen in adolescents.
Gestational Diabetes Mellitus (GDM):
- Cause: Insulin resistance that develops during pregnancy, often resolving after childbirth but increasing future risk of T2DM.
B. Metabolic Consequences of Absolute Insulin Deficiency (Type 1 Diabetes)
This leads to a profound metabolic crisis, an exaggerated fasted state, if untreated.
Hyperglycemia (High Blood Glucose):
- Increased Hepatic Glucose Production: Unchecked glycogenolysis and gluconeogenesis due to unopposed glucagon.
- Decreased Glucose Utilization: Insulin-dependent tissues (muscle, adipose) cannot take up glucose due to the lack of GLUT4 translocation.
- Result: Blood glucose soars, leading to osmotic diuresis (excessive urination) and thirst (polydipsia).
Increased Lipolysis and Hypertriglyceridemia:
- Unchecked Lipolysis: The absence of insulin means Hormone-Sensitive Lipase (HSL) is constantly active, leading to massive breakdown of stored TAGs.
- Increased Fatty Acids & VLDL: High levels of free fatty acids are released, and the liver continuously synthesizes VLDL, leading to high blood triglycerides.
Exaggerated Ketogenesis and Diabetic Ketoacidosis (DKA):
- This is a life-threatening complication of uncontrolled T1DM.
- Mechanism: A high influx of fatty acids to the liver, coupled with their rapid β-oxidation, generates huge amounts of Acetyl-CoA. Uninhibited ketogenesis converts this Acetyl-CoA into ketone bodies.
- Metabolic Acidosis: The ketone bodies (acetoacetate and β-hydroxybutyrate) are strong acids. Their overproduction overwhelms the body's buffering capacity, causing blood pH to drop.
- Symptoms: Nausea, fruity breath (due to acetone), Kussmaul respiration (deep, labored breathing), confusion, and coma.
Protein Catabolism and Muscle Wasting:
The absence of insulin inhibits protein synthesis and promotes muscle protein breakdown. The released amino acids contribute to hepatic gluconeogenesis, exacerbating hyperglycemia and leading to significant weight loss.
C. Metabolic Consequences of Insulin Resistance (Type 2 Diabetes):
Hyperglycemia:
- Insulin Resistance in Muscle/Adipose: Reduced glucose uptake.
- Insulin Resistance in Liver: Fails to suppress hepatic glucose production.
- β-cell Dysfunction: Eventually, insulin secretion becomes inadequate to overcome resistance.
Dyslipidemia:
Insulin resistance leads to increased lipolysis, increased VLDL production, low HDL cholesterol, and the formation of small, dense LDL particles, increasing cardiovascular disease risk.
Less Prone to Ketoacidosis:
Patients with T2DM usually produce some insulin, which is often enough to suppress massive ketogenesis. A more common acute complication is Hyperosmolar Hyperglycemic State (HHS), characterized by extreme hyperglycemia and dehydration without significant ketoacidosis.
D. Key Principles of Treatment:
Type 1 Diabetes:
- Insulin Replacement: Essential for survival.
- Diet and Exercise: Crucial for managing blood glucose.
Type 2 Diabetes:
- Lifestyle Modifications: Diet and exercise are foundational.
- Oral Medications: Metformin (reduces hepatic glucose production), Sulfonylureas (stimulate insulin secretion), and others.
- Insulin Therapy: May be required as the disease progresses.
Biochemistry: Integrated Metabolism
Fuel Homeostasis
Test your knowledge with these 30 questions.
Integrated Metabolism Quiz
Question 1/30
Quiz Complete!
Here are your results, .
Your Score
27/30
90%
Fatty Acid Metabolism
Fatty Acid : Metabolism
Fatty Acid Metabolism
Fatty acids are fundamental molecules in biology, playing roles in energy, structure, and signaling. Their metabolism is highly regulated and central to energy homeostasis in the body. For more details on Fatty Acids, Click Here.
Briefly, What are Fatty Acids?
Fatty acids are long hydrocarbon chains with a carboxyl group (-COOH) at one end. This makes them amphipathic molecules, meaning they have both hydrophobic (the hydrocarbon chain) and hydrophilic (the carboxyl group) regions. They are found esterified to glycerol in triacylglycerols (TAGs) or as components of phospholipids and sphingolipids. In their free form, they are called free fatty acids (FFAs).
Classification of Fatty Acids
A. Based on Saturation:
- Saturated Fatty Acids (SFAs): Contain no carbon-carbon double bonds. Examples: Palmitic acid (16:0), Stearic acid (18:0). Tend to be solid at room temperature.
- Unsaturated Fatty Acids (UFAs): Contain one or more carbon-carbon double bonds.
- Monounsaturated (MUFAs): Have one double bond (e.g., Oleic acid).
- Polyunsaturated (PUFAs): Have two or more double bonds (e.g., Linoleic acid).
B. Based on Chain Length:
- Short-Chain (SCFAs): 2 to 4 carbons.
- Medium-Chain (MCFAs): 6 to 12 carbons.
- Long-Chain (LCFAs): 14 to 20 carbons (most common).
- Very Long-Chain (VLCFAs): >20 carbons.
C. Based on Essentiality:
- Non-Essential Fatty Acids: Can be synthesized by the body.
- Essential Fatty Acids (EFAs): Cannot be synthesized and must be obtained from the diet.
- Linoleic Acid (Omega-6): Precursor to arachidonic acid.
- α-Linolenic Acid (Omega-3): Precursor to EPA and DHA.
Major Physiological Roles of Fatty Acids
Fatty acids are multifaceted molecules critical for life.
Energy Storage
Stored as triacylglycerols (TAGs), they are the body's most concentrated and efficient form of long-term energy storage, yielding more ATP per gram than carbohydrates.
Structural Components
They are integral components of phospholipids and sphingolipids, which form the fundamental structure of all biological membranes.
Signaling & Precursors
Essential fatty acids are precursors to powerful local signaling molecules called eicosanoids (prostaglandins, thromboxanes, leukotrienes) involved in inflammation, pain, and blood clotting.
Insulation & Absorption
Adipose tissue provides thermal insulation and protection for organs. Dietary fats are also necessary for the absorption of fat-soluble vitamins (A, D, E, K).
Primary Metabolic States: Fed vs. Fasted
The body meticulously regulates fatty acid metabolism based on energy availability.
Fed State (High Energy / Insulin Dominant)
After a meal, excess carbohydrates and proteins are converted into fatty acids (Lipogenesis) and stored as TAGs in adipose tissue. The goal is to store energy.
Fasted State (Low Energy / Glucagon Dominant)
When nutrient intake is low, stored TAGs are broken down, releasing fatty acids. These are then broken down for energy (Beta-Oxidation). The goal is to release stored energy.
Major Pathways Involved in Fatty Acid Metabolism
- Fatty Acid Synthesis (Lipogenesis): The process of building fatty acids from Acetyl-CoA. Occurs primarily in the cytosol.
- Fatty Acid Oxidation (Beta-Oxidation): The pathway that breaks down fatty acids into Acetyl-CoA to generate energy. Occurs primarily in the mitochondrial matrix.
- Triacylglycerol (TAG) Synthesis and Degradation: The processes of storing (esterification) and mobilizing (lipolysis) fatty acids.
- Ketone Body Metabolism:
- Ketogenesis: The liver converts excess Acetyl-CoA into ketone bodies during prolonged fasting.
- Ketolysis: Other tissues use ketone bodies as an alternative fuel source.
Fatty Acid Mobilization and Transport
When energy is needed, stored triacylglycerols (TAGs) in adipose tissue must be broken down, and the resulting fatty acids transported to other tissues for oxidation.
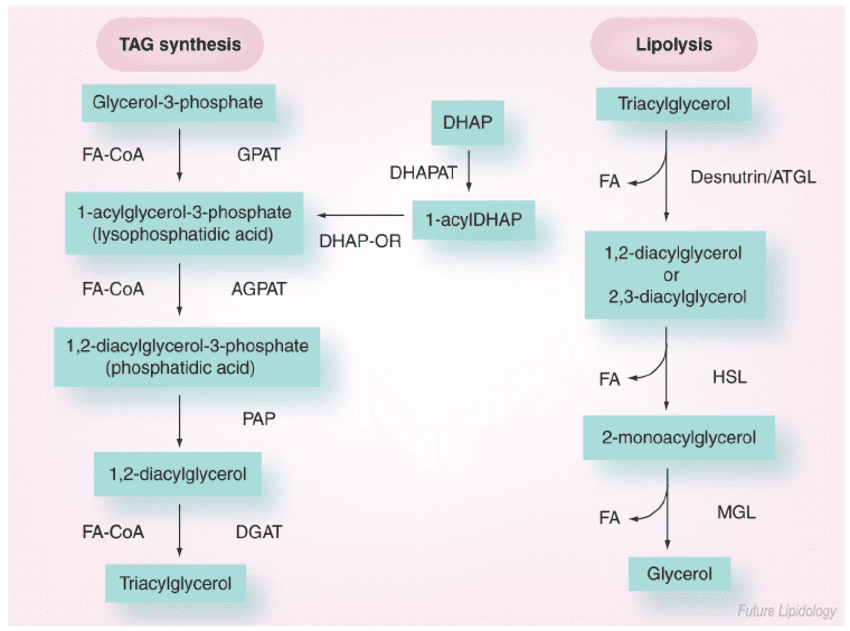
1. Triacylglycerol (TAG) Mobilization (Lipolysis)
Lipolysis is the process of breaking down stored TAGs into fatty acids and glycerol, occurring in adipocytes.
- Stimuli: Hormones like epinephrine, norepinephrine, and glucagon signal a low-energy state and activate lipolysis. Insulin inhibits it.
- Key Players (Lipases):
- Hormone-Sensitive Lipase (HSL): The rate-limiting enzyme, activated by phosphorylation via a PKA-dependent pathway.
- Adipose Triglyceride Lipase (ATGL): Initiates the first step, converting TAGs to DAGs.
- Monoacylglycerol Lipase (MAGL): Catalyzes the final step.
- Products of Lipolysis: Free Fatty Acids (FFAs) and Glycerol are released into the bloodstream.
- Fate of Glycerol: Travels to the liver, where it can enter glycolysis or gluconeogenesis. Adipocytes lack the enzyme (glycerol kinase) to re-utilize it.
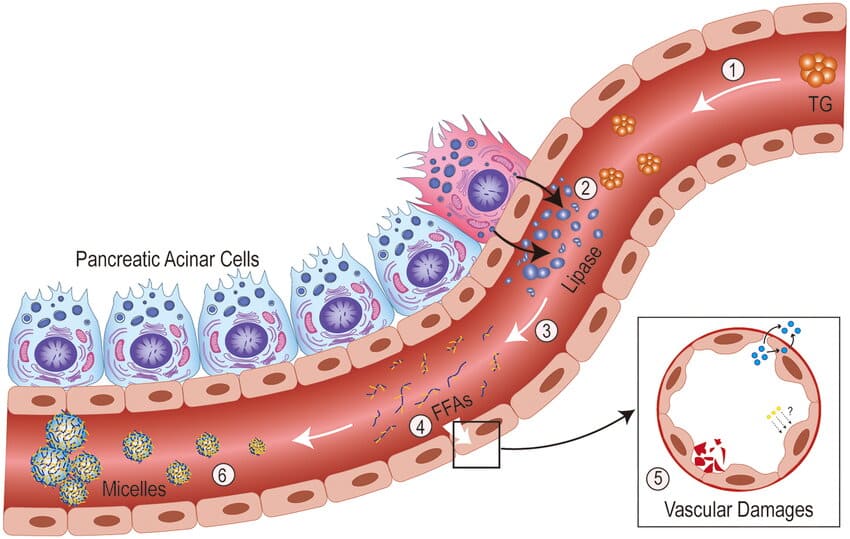
2. Transport of Free Fatty Acids (FFAs) in Blood
Long-chain fatty acids are hydrophobic and require a carrier in the blood.
- Carrier Protein: Albumin, the most abundant plasma protein, serves as the primary carrier for FFAs.
- Mechanism: FFAs bind non-covalently to hydrophobic pockets on the albumin molecule.
- Delivery to Tissues: Fatty acid-albumin complexes deliver FFAs to tissues like muscle and heart, where they are taken up by specific fatty acid transporters.
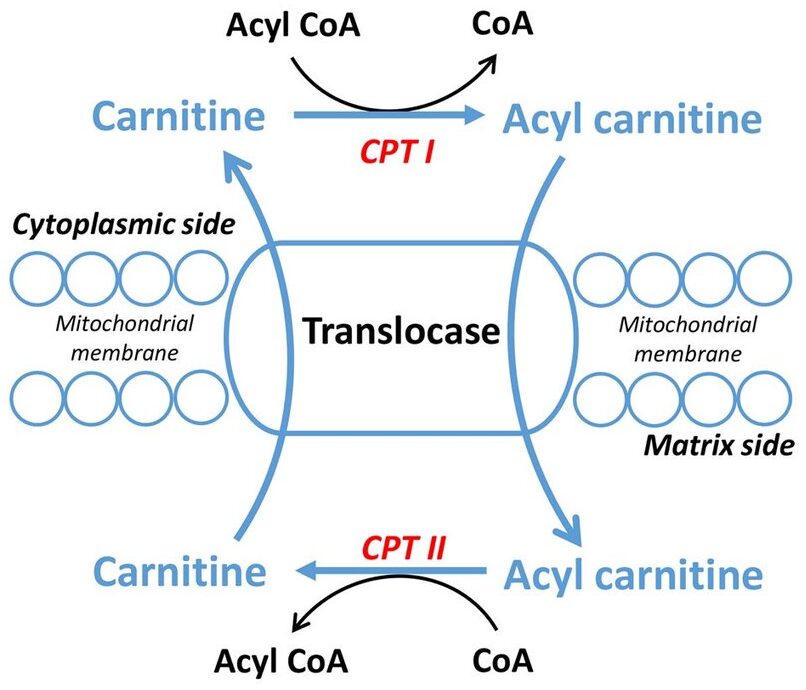
3. Transport into Mitochondria (The Carnitine Shuttle)
Long-chain fatty acids (LCFAs) cannot directly cross the inner mitochondrial membrane. They require the Carnitine Shuttle to enter the mitochondrial matrix for beta-oxidation.
Steps of the Shuttle:
- Activation (Cytosol): The FFA is first activated to a fatty acyl-CoA by Fatty Acyl-CoA Synthetase, consuming 2 ATP equivalents.
- Transfer to Carnitine (Outer Membrane): The fatty acyl group is transferred from CoA to carnitine by Carnitine Palmitoyltransferase I (CPT-I), forming fatty acylcarnitine. CPT-I is the rate-limiting step and is inhibited by malonyl-CoA.
- Translocation (Inner Membrane): Carnitine-Acylcarnitine Translocase (CACT) transports fatty acylcarnitine into the matrix while simultaneously transporting a free carnitine out.
- Transfer Back to CoA (Matrix): Inside the matrix, Carnitine Palmitoyltransferase II (CPT-II) transfers the fatty acyl group back to a mitochondrial CoA, regenerating fatty acyl-CoA (now ready for beta-oxidation) and freeing carnitine for reuse.
Now, with the fatty acyl-CoA ready in the mitochondrial matrix, we can move on to the actual breakdown process: Fatty Acid Oxidation (Beta-Oxidation).
Fatty Acid Oxidation (Beta-Oxidation)
Once long-chain fatty acids (as fatty acyl-CoA) have successfully entered the mitochondrial matrix via the carnitine shuttle, they are ready for a cyclic process called β-oxidation. This pathway systematically cleaves two-carbon units from the carboxyl end of the fatty acyl-CoA, generating acetyl-CoA, NADH, and FADH₂, which then feed into the citric acid cycle and oxidative phosphorylation for ATP production.
- Primary Location: Mitochondrial matrix.
- Purpose: To generate energy (ATP) from stored fatty acids.
The Sequential Steps of β-Oxidation for Saturated Fatty Acyl-CoAs
Beta-oxidation is a four-step cyclic process. Each cycle shortens the fatty acyl-CoA by two carbons and produces one molecule of Acetyl-CoA, one NADH, and one FADH₂.
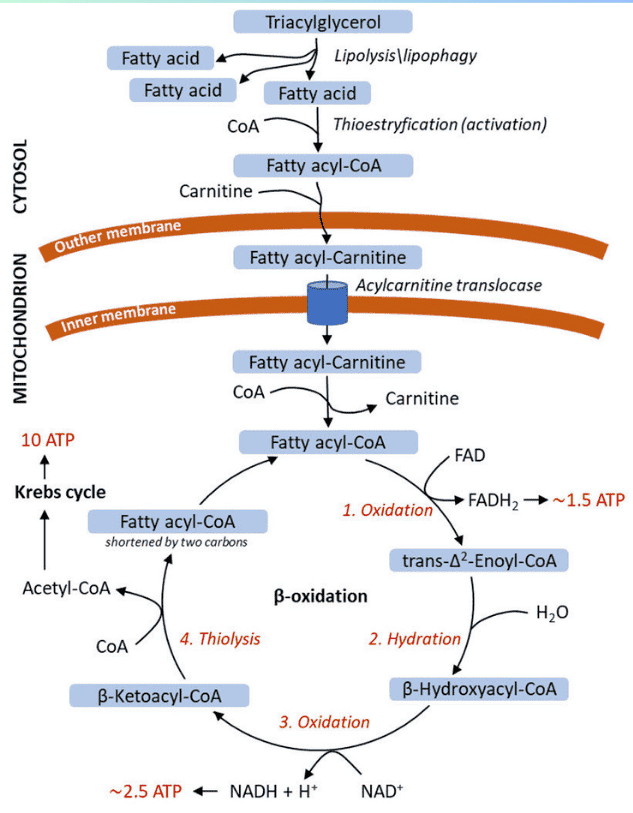
The Four Steps of One Cycle:
-
Oxidation (by FAD):
- Enzyme: Acyl-CoA Dehydrogenase (specific for chain length, e.g., VLCAD, LCAD, MCAD, SCAD).
- Reaction: Introduces a trans double bond between the α (C-2) and β (C-3) carbons of the fatty acyl-CoA, producing a trans-Δ²-enoyl-CoA.
- Product: FADH₂ (reduced flavin adenine dinucleotide). This FADH₂ then donates its electrons to Coenzyme Q in the electron transport chain, yielding ~1.5 ATP.
-
Hydration:
- Enzyme: Enoyl-CoA Hydratase (also known as Crotonase).
- Reaction: Adds water across the double bond of the trans-Δ²-enoyl-CoA, forming a hydroxyl group on the β-carbon. This produces L-β-hydroxyacyl-CoA.
-
Oxidation (by NAD⁺):
- Enzyme: β-hydroxyacyl-CoA Dehydrogenase.
- Reaction: Oxidizes the hydroxyl group on the β-carbon to a ketone group, producing β-ketoacyl-CoA.
- Product: NADH (reduced nicotinamide adenine dinucleotide). This NADH then donates its electrons to Complex I of the electron transport chain, yielding ~2.5 ATP.
-
Thiolytic Cleavage (Thiolysis):
- Enzyme: β-ketoacyl-CoA Thiolase (also known as Acyl-CoA Acetyltransferase).
- Reaction: Cleaves the bond between the α and β carbons. A molecule of Coenzyme A (CoA-SH) attacks the β-keto carbon, releasing one molecule of Acetyl-CoA and a new fatty acyl-CoA that is two carbons shorter than the original.
- Products: Acetyl-CoA (enters the Citric Acid Cycle) and a shortened fatty acyl-CoA (which re-enters the β-oxidation cycle).
Summary of One Cycle of β-Oxidation:
Input: Fatty Acyl-CoA (n carbons) → Output: 1 Acetyl-CoA + 1 FADH₂ + 1 NADH + Fatty Acyl-CoA (n-2 carbons)
Calculating the Net ATP Yield from Palmitate (16:0)
- Number of carbons: 16
- Number of Acetyl-CoA units produced: 16 / 2 = 8 Acetyl-CoA.
- Number of β-oxidation cycles needed: 8 - 1 = 7 cycles.
- ATP Yield Calculation:
- From β-oxidation cycles:
- 7 cycles × 1 FADH₂/cycle = 7 FADH₂
- 7 FADH₂ × 1.5 ATP/FADH₂ = 10.5 ATP
- 7 cycles × 1 NADH/cycle = 7 NADH
- 7 NADH × 2.5 ATP/NADH = 17.5 ATP
- Total from cycles = 10.5 + 17.5 = 28 ATP
- From Acetyl-CoA entering the Citric Acid Cycle (TCA Cycle):
- 8 Acetyl-CoA × (1 FADH₂ + 3 NADH + 1 GTP)/Acetyl-CoA
- 8 FADH₂ × 1.5 ATP/FADH₂ = 12 ATP
- 8 NADH × 2.5 ATP/NADH = 20 ATP
- 8 GTP × 1 ATP/GTP = 8 ATP
- Total from Acetyl-CoA = 12 + 20 + 8 = 40 ATP
- Initial Activation Cost:
- Activating the fatty acid consumes 2 ATP equivalents.
- Cost = -2 ATP
- From β-oxidation cycles:
- Net ATP Yield: (28 from cycles) + (40 from Acetyl-CoA) - 2 (activation) = 106 ATP.
Modifications for Unsaturated and Odd-Chain Fatty Acids
A. Unsaturated Fatty Acids:
- Problem: The presence of double bonds interferes with the standard pathway.
- Solutions:
- Enoyl-CoA Isomerase: For cis double bonds, this enzyme converts them to the trans form, bypassing the FADH₂-producing step in that cycle.
- 2,4-Dienoyl-CoA Reductase: For polyunsaturated fatty acids, this reductase (requiring NADPH) helps handle conjugated double bonds.
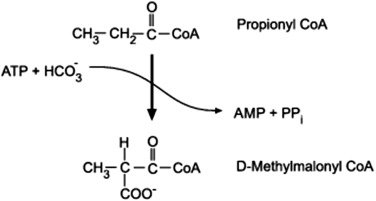
B. Odd-Chain Fatty Acids:
- Problem: The final cycle yields one Propionyl-CoA (3 carbons), which cannot enter the TCA cycle directly.
- Solution (Propionyl-CoA Pathway): Propionyl-CoA is converted to Succinyl-CoA (a TCA cycle intermediate) in a multi-step process requiring Biotin and Vitamin B12.
- Significance: This makes odd-chain fatty acids the only fatty acids that can yield a net glucose precursor.
Other Forms of Fatty Acid Oxidation
A. α-Oxidation:
- Location: Peroxisomes and Endoplasmic Reticulum.
- Purpose: Degrades fatty acids with a methyl group on the β-carbon (e.g., phytanic acid).
- Process: Removes one carbon at a time from the carboxyl end.
- Clinical Significance: A defect causes Refsum disease, leading to neurological damage.
B. ω-Oxidation:
- Location: Endoplasmic Reticulum of the liver and kidneys.
- Purpose: A minor pathway that becomes more important when β-oxidation is defective.
- Process: Oxidizes the methyl (ω) carbon at the opposite end of the chain, creating a dicarboxylic acid that can then undergo β-oxidation from both ends.
- Products: Succinate (4 carbons) and Adipate (6 carbons).
Ketone Body Metabolism (Ketogenesis and Ketolysis)
Under certain physiological conditions, particularly prolonged fasting, starvation, or uncontrolled diabetes, the liver produces significant amounts of ketone bodies from Acetyl-CoA. These ketone bodies serve as an alternative fuel source for extrahepatic (outside the liver) tissues, especially the brain, which cannot directly use fatty acids for energy.
Conditions That Lead to Ketogenesis
Ketogenesis is stimulated when:
- Low Glucose Availability: This is the primary driver. When glucose is scarce, the body turns to fat as its main energy source.
- High Rate of Fatty Acid Oxidation: Increased breakdown of fatty acids in the liver leads to an abundance of Acetyl-CoA.
- Low Oxaloacetate (OAA) Levels in the Liver: OAA is a crucial intermediate in the Citric Acid Cycle (TCA cycle) that combines with Acetyl-CoA to form citrate.
- During fasting, OAA is diverted to gluconeogenesis (glucose synthesis) in the liver to maintain blood glucose levels.
- This depletion of OAA means that Acetyl-CoA cannot efficiently enter the TCA cycle.
- High Glucagon/Insulin Ratio: Glucagon promotes fatty acid mobilization and gluconeogenesis, further contributing to the conditions favoring ketogenesis.
- Clinical States: Starvation/Fasting, Uncontrolled Diabetes Mellitus (Type 1), Low Carbohydrate, High-Fat Diets (Ketogenic Diets).
In essence, ketogenesis is a response to an oversupply of Acetyl-CoA (from fat breakdown) and an undersupply of OAA (due to gluconeogenesis) in the liver.
Synthesis of Ketone Bodies (Ketogenesis) in the Liver
Ketogenesis occurs exclusively in the mitochondrial matrix of liver cells.
The Three Ketone Bodies:
- Acetoacetate: The primary ketone body produced.
- β-Hydroxybutyrate: Formed by the reduction of acetoacetate.
- Acetone: A volatile byproduct of acetoacetate breakdown, produced in smaller quantities and excreted via breath.
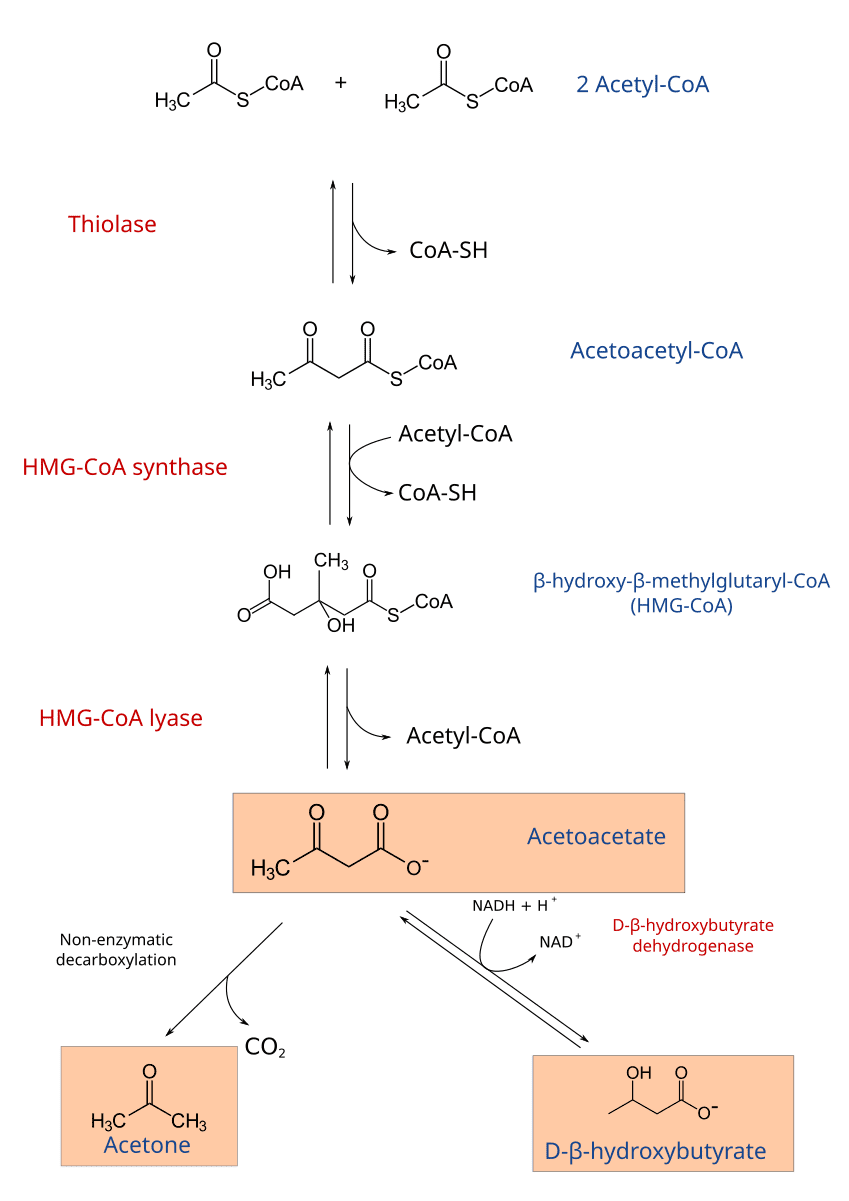
Steps of Ketogenesis:
-
1. Condensation of Two Acetyl-CoA Molecules:
- Enzyme: Thiolase (the reverse reaction of the last step of β-oxidation).
- Reaction:
2 Acetyl-CoA → Acetoacetyl-CoA + CoA-SH
-
2. Condensation with a Third Acetyl-CoA:
- Enzyme: HMG-CoA Synthase (Hydroxymethylglutaryl-CoA Synthase).
- Reaction:
Acetoacetyl-CoA + Acetyl-CoA + H₂O → β-hydroxy-β-methylglutaryl-CoA (HMG-CoA) + CoA-SH - Note: This is the rate-limiting step of ketogenesis.
-
3. Cleavage of HMG-CoA:
- Enzyme: HMG-CoA Lyase.
- Reaction:
HMG-CoA → Acetoacetate + Acetyl-CoA - This reaction produces the first ketone body, acetoacetate.
-
4. Interconversion and Breakdown of Acetoacetate:
- Acetoacetate can be reduced to β-hydroxybutyrate.
- Enzyme: β-hydroxybutyrate Dehydrogenase.
- Reaction:
Acetoacetate + NADH + H⁺ ⇌ β-Hydroxybutyrate + NAD⁺
- Acetoacetate can also spontaneously decarboxylate to Acetone (
Acetoacetate → Acetone + CO₂).
- Acetoacetate can be reduced to β-hydroxybutyrate.
Utilization (Ketolysis) of Ketone Bodies by Extrahepatic Tissues
Ketone bodies are water-soluble and can be transported via the bloodstream to peripheral tissues, which then convert them back into Acetyl-CoA for energy. The liver cannot utilize ketone bodies because it lacks a key enzyme for ketolysis.
Tissues that use Ketone Bodies: Brain, heart, skeletal muscle, renal cortex.
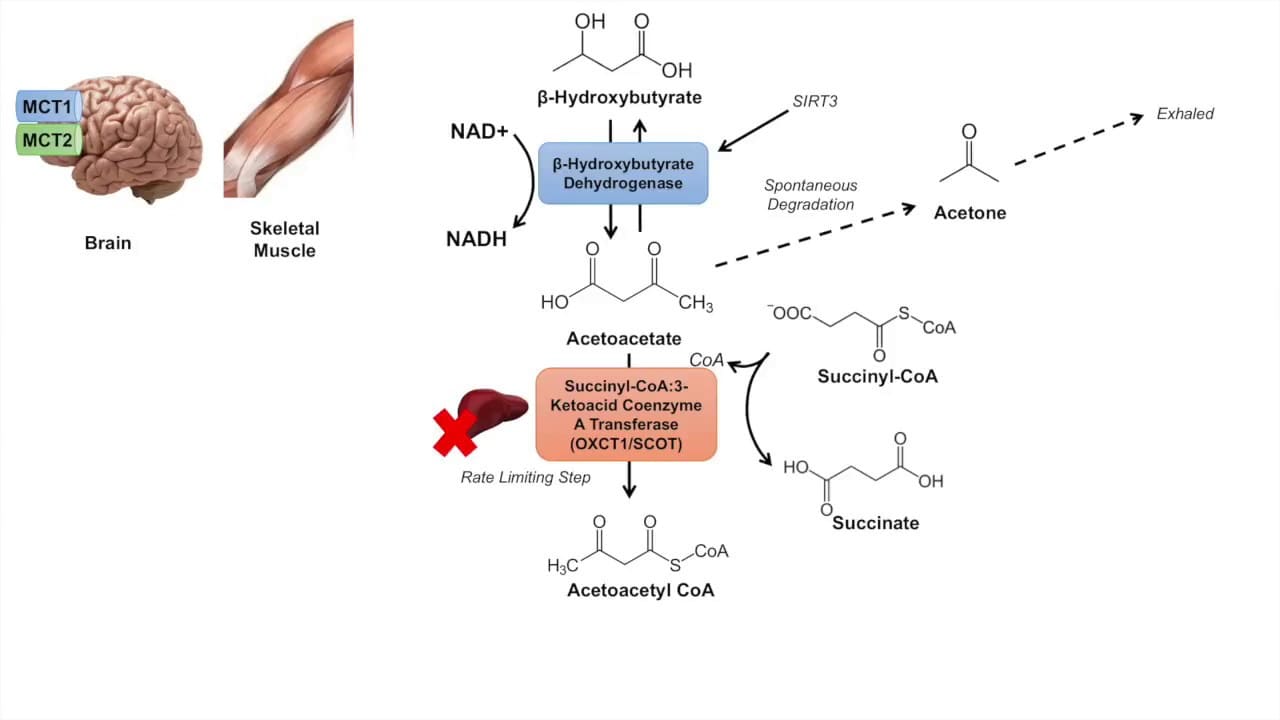
Steps of Ketolysis (Example: in the Brain/Muscle):
-
1. Conversion of β-Hydroxybutyrate to Acetoacetate:
- Enzyme: β-hydroxybutyrate Dehydrogenase.
- Reaction:
β-Hydroxybutyrate + NAD⁺ → Acetoacetate + NADH + H⁺
-
2. Activation of Acetoacetate:
- Enzyme: β-ketoacyl-CoA Transferase (also called Thiophorase).
- Reaction:
Acetoacetate + Succinyl-CoA → Acetoacetyl-CoA + Succinate - Crucial: This enzyme is absent in the liver, which is why the liver produces but cannot utilize ketone bodies.
-
3. Cleavage of Acetoacetyl-CoA:
- Enzyme: Thiolase.
- Reaction:
Acetoacetyl-CoA + CoA-SH → 2 Acetyl-CoA
The 2 molecules of Acetyl-CoA produced can then enter the Citric Acid Cycle to generate ATP.
Clinical Significance of Ketogenesis
The production and utilization of ketone bodies are normally well-regulated. However, imbalances can lead to serious clinical conditions.
- Physiological Ketosis: A normal and beneficial state that occurs during prolonged fasting, starvation, or a strict ketogenic diet. Ketone bodies provide a crucial fuel source, especially for the brain, preserving muscle protein.
- Pathological Ketosis (Ketoacidosis):
- Diabetic Ketoacidosis (DKA): This is a life-threatening complication of Type 1 Diabetes Mellitus.
- Cause: Absolute or severe relative insulin deficiency combined with elevated glucagon levels.
- Mechanism: Lack of insulin means cells cannot take up glucose, leading to severe hyperglycemia. Simultaneously, high glucagon promotes massive lipolysis and unchecked ketogenesis in the liver.
- Consequences: The rapid and excessive production of acidic ketone bodies overwhelms the body's buffering capacity, leading to a significant drop in blood pH (acidosis), dehydration, electrolyte imbalances, and potentially coma and death if untreated.
- Acetone: The increased production of acetoacetate leads to increased spontaneous decarboxylation to acetone, giving the breath of DKA patients a characteristic "fruity" odor.
- Alcoholic Ketoacidosis: Can occur in chronic alcoholics, often exacerbated by poor nutrition. Alcohol metabolism produces excess NADH, which shifts OAA to malate and inhibits gluconeogenesis, leading to a similar state of excessive ketogenesis and acidosis.
- Diabetic Ketoacidosis (DKA): This is a life-threatening complication of Type 1 Diabetes Mellitus.
Fatty Acid Synthesis (Lipogenesis)
When the body has an abundance of energy, especially from a diet rich in carbohydrates, it converts excess glucose into fatty acids for long-term storage as triacylglycerols. This process is called lipogenesis.
Overview and Key Tissues
- Definition: The metabolic pathway that synthesizes fatty acids from acetyl-CoA.
- Primary Precursor: Acetyl-CoA, which is largely derived from carbohydrate metabolism (pyruvate oxidation).
- Location: Primarily in the cytosol of cells.
- Major Sites:
Liver: The most active site of fatty acid synthesis.Adipose Tissue: Also synthesizes fatty acids.Lactating Mammary Glands: Synthesize fatty acids for milk production.
- Main Product: Palmitate (16:0), a saturated 16-carbon fatty acid.
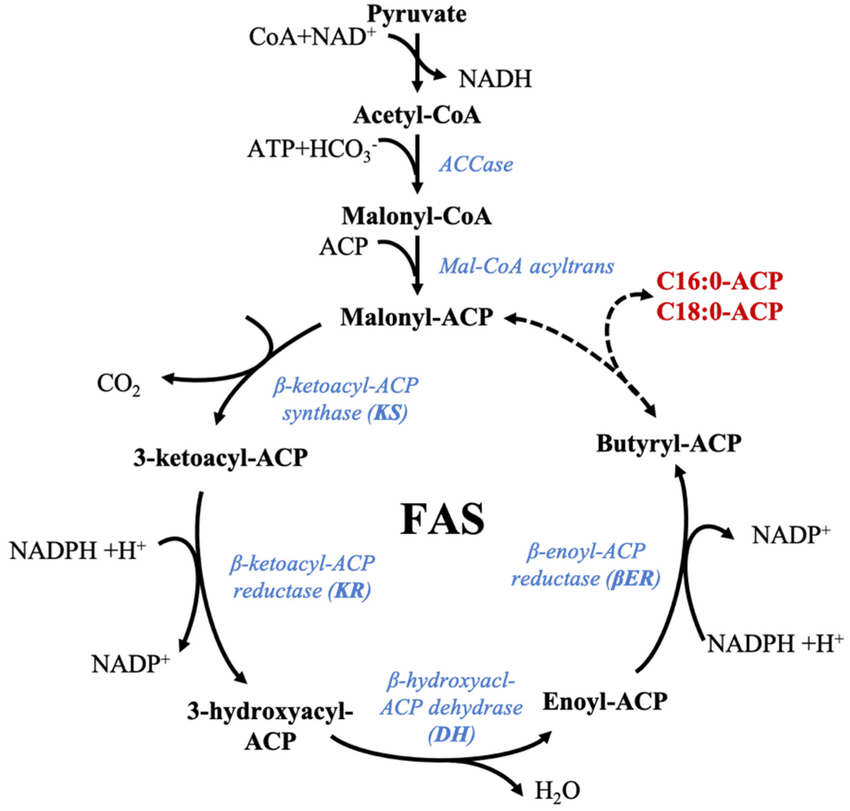
Key Steps and Enzymes in Fatty Acid Synthesis
Fatty acid synthesis is essentially a reversal of β-oxidation, but it uses different enzymes, occurs in a different cellular compartment, and employs a different electron donor.
A. Transport of Acetyl-CoA from Mitochondria to Cytosol:
- Problem: Acetyl-CoA is produced in the mitochondrial matrix, but synthesis occurs in the cytosol. The inner mitochondrial membrane is impermeable to Acetyl-CoA.
- Solution: The Citrate Shuttle
- Condensation: Acetyl-CoA combines with oxaloacetate (OAA) in the mitochondrial matrix to form citrate (catalyzed by Citrate Synthase).
- Transport: Citrate is transported across the inner mitochondrial membrane into the cytosol.
- Cleavage: In the cytosol, citrate is cleaved back into Acetyl-CoA and OAA by ATP Citrate Lyase. This step requires ATP.
Citrate + ATP + CoA-SH → Acetyl-CoA + OAA + ADP + Pi - Recycling OAA: The cytosolic OAA is converted to malate and then pyruvate (producing NADPH in the process via malic enzyme) before returning to the mitochondria.
B. Carboxylation of Acetyl-CoA to Malonyl-CoA:
- Enzyme: Acetyl-CoA Carboxylase (ACC).
- Reaction:
Acetyl-CoA + HCO₃⁻ + ATP → Malonyl-CoA + ADP + Pi - Significance: This is the rate-limiting and committed step of fatty acid synthesis.
- Requirements: Biotin and ATP.
C. The Fatty Acid Synthase Complex:
Synthesis is carried out by a multi-enzyme complex called Fatty Acid Synthase (FAS). It contains seven different enzymatic activities and an acyl carrier protein (ACP).
- Electron Donor: NADPH (not NADH or FADH₂).
Steps of the FAS Cycle (Repeated 7 Times):
Each cycle adds a two-carbon unit from Malonyl-CoA and involves four steps:
- Condensation: The growing fatty acyl chain condenses with malonyl-ACP, releasing CO₂. (Enzyme: β-ketoacyl-ACP Synthase).
- Reduction (by NADPH): The β-keto group is reduced to a β-hydroxy group. (Enzyme: β-ketoacyl-ACP Reductase).
- Dehydration: Water is removed, creating a double bond. (Enzyme: β-hydroxyacyl-ACP Dehydratase).
- Reduction (by NADPH): The double bond is reduced, resulting in a saturated acyl-ACP chain that is two carbons longer. (Enzyme: Enoyl-ACP Reductase).
After 7 cycles, the 16-carbon palmitoyl-ACP is formed and then released as free palmitate by a Thioesterase.
Summary of Palmitate Synthesis:
Overall Reaction: 8 Acetyl-CoA + 7 ATP + 14 NADPH → Palmitate + 8 CoA + 7 ADP + 7 Pi + 14 NADP⁺ + 6 H₂O
Regulation of Fatty Acid Synthesis
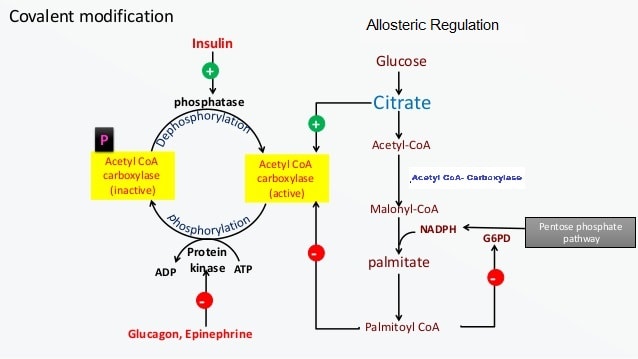
A. Short-Term Regulation (of ACC):
- Allosteric Activators: Citrate. High levels of citrate indicate excess energy and activate ACC.
- Allosteric Inhibitors: Long-Chain Fatty Acyl-CoAs. High levels of the end-product inhibit ACC.
- Covalent Modification:
- Dephosphorylation (Activation): Insulin activates a phosphatase that dephosphorylates and activates ACC.
- Phosphorylation (Inhibition): Glucagon and Epinephrine activate PKA, which phosphorylates and inactivates ACC. AMP-activated protein kinase (AMPK) also inactivates ACC when cellular energy is low.
B. Long-Term Regulation (Gene Expression):
- Dietary Factors: High-carbohydrate, low-fat diets increase the synthesis of ACC and FAS enzymes. Fasting or high-fat diets decrease their synthesis.
- Hormonal Factors: Insulin increases the synthesis of enzymes for fatty acid synthesis.
Elongation and Desaturation of Fatty Acids
Once palmitate (16:0) is synthesized, it can be further modified:
- Elongation: Occurs primarily in the endoplasmic reticulum (ER). Adds two carbons at a time, using Malonyl-CoA and NADPH, to produce stearate (18:0) and other longer fatty acids.
- Desaturation: Occurs in the ER. Introduces double bonds into saturated fatty acids.
- Enzymes: Fatty Acyl-CoA Desaturases, which require O₂, NADH (or NADPH), and cytochrome b5.
- Limitations: Mammals can introduce double bonds at Δ9, Δ6, and Δ5 positions but cannot introduce double bonds beyond Δ9. This is why linoleic acid (Δ9,12) and α-linolenic acid (Δ9,12,15) are essential fatty acids.
Regulation and Interplay with Other Pathways
The metabolism of fatty acids is not an isolated process; it is intricately woven into the overall metabolic fabric of the cell and the organism. Regulation ensures that energy is stored when abundant and mobilized when needed, all while maintaining metabolic homeostasis.
Hormonal Regulation
Hormones are the primary messengers that coordinate fatty acid metabolism across different tissues in response to the body's energy status.
A. Insulin (Hormone of the "Fed" State):
- Effect on Lipogenesis (Fatty Acid Synthesis):
Promotes.- Increases glucose uptake into adipocytes and liver.
- Activates Pyruvate Dehydrogenase, increasing Acetyl-CoA supply.
- Activates Acetyl-CoA Carboxylase (ACC) by dephosphorylation (reducing its Km for citrate).
- Induces gene expression of ACC and Fatty Acid Synthase (FAS).
- Increases the activity of Lipoprotein Lipase (LPL) in adipose tissue, facilitating uptake of dietary TAGs.
- Effect on Lipolysis (Fatty Acid Breakdown):
Inhibits.- Decreases cAMP levels, leading to dephosphorylation and inactivation of Hormone-Sensitive Lipase (HSL).
- Overall: Insulin promotes energy storage in the form of glycogen and triacylglycerols.
B. Glucagon (Hormone of the "Fasted" State):
- Effect on Lipogenesis: Inhibits. Inactivates ACC by phosphorylation (via PKA).
- Effect on Lipolysis: Promotes. Increases cAMP levels, leading to phosphorylation and activation of HSL.
- Overall: Glucagon promotes the mobilization of stored energy, including fatty acids.
C. Epinephrine and Norepinephrine (Catecholamines - "Fight or Flight" Hormones):
- Effect on Lipolysis: Potent stimulators. Bind to adrenergic receptors on adipocytes, leading to increased cAMP and activation of HSL via PKA.
- Overall: Rapidly mobilizes fatty acids for immediate energy needs during stress.
D. Thyroid Hormones:
Generally increase metabolic rate, which can indirectly affect fatty acid metabolism by increasing both synthesis and breakdown, depending on the overall energy balance.
Allosteric and Covalent Regulation
Beyond hormones, specific molecules within metabolic pathways can directly activate or inhibit key enzymes.
A. Regulation of Acetyl-CoA Carboxylase (ACC) - Key for Synthesis:
- Allosteric Activator:
Citrate(high levels indicate abundant energy and Acetyl-CoA). - Allosteric Inhibitor: Long-chain fatty acyl-CoAs (product inhibition).
- Covalent Modification: Phosphorylation (by PKA, AMPK) inactivates; dephosphorylation (by insulin-activated phosphatase) activates.
B. Regulation of Carnitine Palmitoyltransferase I (CPT-I) - Key for Oxidation:
- Allosteric Inhibitor:
Malonyl-CoA. - This is a crucial point of reciprocal regulation: When fatty acid synthesis is active (high
Malonyl-CoA), fatty acid oxidation is inhibited at the entry point to the mitochondria. This prevents a "futile cycle".
C. Regulation of Hormone-Sensitive Lipase (HSL) - Key for Mobilization:
- Covalent Modification: Phosphorylation (by PKA) activates; dephosphorylation (by insulin-activated phosphatase) inactivates.
Transcriptional (Gene Expression) Regulation
Long-term adaptation to dietary and hormonal changes involves altering the amount of enzymes present.
- Insulin: Upregulates the synthesis of enzymes for lipogenesis (ACC, FAS, ATP citrate lyase).
- Fasting/Starvation: Downregulates the synthesis of lipogenic enzymes and upregulates enzymes for fatty acid oxidation.
- PPARs (Peroxisome Proliferator-Activated Receptors): These are nuclear receptors that act as transcription factors. For example, PPARα is activated by fatty acids and promotes the expression of genes involved in fatty acid oxidation.
Interplay with Other Metabolic Pathways
A. Fatty Acid-Carbohydrate Interplay (The Glucose-Fatty Acid Cycle / Randle Cycle):
- In the Fed State: High glucose leads to insulin release, promoting glucose utilization and lipogenesis.
- In the Fasted State: Low glucose leads to glucagon release, promoting lipolysis. The increased fatty acids and their oxidation products (
Acetyl-CoA,NADH,citrate) inhibit glucose utilization in peripheral tissues, sparing glucose for the brain.- High
Acetyl-CoAinhibits Pyruvate Dehydrogenase. - High
citrateinhibits PFK-1 (Phosphofructokinase-1). - High
NADH/NAD⁺ratio also inhibits various steps in carbohydrate metabolism.
- High
- Overall: There's a reciprocal relationship: high fatty acid oxidation inhibits glucose oxidation, and vice versa.
B. Fatty Acid-Protein Interplay:
- Some amino acids can be converted to
Acetyl-CoAfor fatty acid synthesis. - During starvation, protein breakdown becomes a more significant source of energy and gluconeogenic precursors.
C. Fatty Acid-Ketone Body Interplay:
When fatty acid oxidation is high and OAA is diverted to gluconeogenesis, excess Acetyl-CoA is converted into ketone bodies in the liver, serving as an alternative fuel for extrahepatic tissues, particularly the brain.
D. Connection to Cholesterol Synthesis:
Acetyl-CoA is the sole precursor for cholesterol synthesis. HMG-CoA (an intermediate in ketogenesis) is also an intermediate in cholesterol synthesis.
Biochemistry: Fatty Acid Metabolism
Test your knowledge with these 40 questions.
Fatty Acid Metabolism Quiz
Question 1/40
Quiz Complete!
Here are your results, .
Your Score
38/40
95%
Pentose Phosphate Pathway
Pentose Phosphate Pathway: PPP
Pentose Phosphate Pathway (PPP)
The Pentose Phosphate Pathway (PPP), also known as the Hexose Monophosphate Shunt (HMP Shunt), is an alternative metabolic route for glucose metabolism that runs parallel to glycolysis. The HMP pathway is also known as the Warburg-Dickens pathway. About 10% of glucose entering this pathway per day. The liver & RBCs metabolise about 30% of glucose by this pathway.
Unlike glycolysis, its primary purpose is not to generate ATP. Instead, its main functions are:
- Production of NADPH: Essential for reductive biosynthetic reactions and for protecting cells from oxidative stress.
- Production of Ribose-5-Phosphate: A vital precursor for the synthesis of nucleotides (DNA, RNA) and coenzymes.
Think of the PPP as a "shunt" because it diverts glucose-6-phosphate away from glycolysis to serve these distinct purposes, and can then feed intermediates back into glycolysis. It primarily occurs in the cytosol of cells.
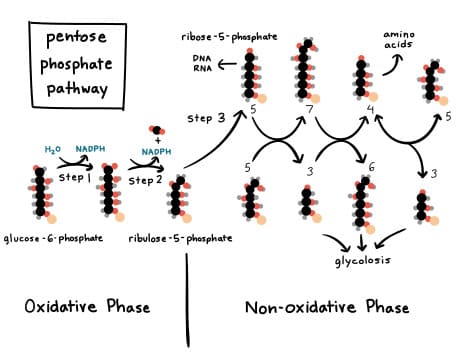
Two Major Phases of the PPP
The Pentose Phosphate Pathway is divided into two distinct phases:
a) The Oxidative (Irreversible) Phase:
- Function: This phase is responsible for the generation of NADPH and the production of ribulose-5-phosphate (which is then converted to ribose-5-phosphate).
- Nature: It is largely irreversible.
- Key Reactions: Involves oxidative decarboxylation reactions where glucose-6-phosphate is oxidized, releasing CO₂, and reducing NADP⁺ to NADPH.
b) The Non-Oxidative (Reversible) Phase:
- Function: This phase interconverts various sugar phosphates, primarily transforming pentose phosphates into glycolytic intermediates (fructose-6-phosphate and glyceraldehyde-3-phosphate). This allows carbon skeletons to be recycled back into glycolysis or used for gluconeogenesis.
- Nature: This phase is entirely reversible.
- Key Enzymes: Involves transketolase and transaldolase enzymes, which facilitate the transfer of two-carbon and three-carbon units, respectively.
Products of the PPP
The PPP is critically important because it provides two essential molecules:
a) NADPH (Nicotinamide Adenine Dinucleotide Phosphate, reduced form)
- Structure: Similar to NADH, but with an additional phosphate group.
- Function: Unlike NADH (used in catabolism for ATP), NADPH is predominantly used in anabolic (biosynthetic) processes and as a reductant in antioxidant defense.
- Reductive Biosynthesis: Providing reducing power for the synthesis of fatty acids, cholesterol, and steroid hormones. Tissues actively involved in these syntheses (e.g., liver, adipose tissue, adrenal cortex) have a highly active PPP.
- Antioxidant Defense: Protecting cells from damage by reactive oxygen species (ROS) by maintaining the reduced state of glutathione.
b) Ribose-5-Phosphate
- Structure: A five-carbon sugar phosphate.
- Function: This molecule is the direct precursor for the synthesis of:
- Nucleotides: The building blocks of DNA and RNA.
- Coenzymes: Such as ATP, NADH, FADH₂, and Coenzyme A.
- Demand: Cells that are rapidly dividing (e.g., bone marrow, skin, cancer cells) will have a high demand for ribose-5-phosphate.
Location of the pathway
- The enzymes are located in the cytosol.
- The tissues such as liver, adipose tissue, adrenal gland, erythrocytes, testes & lactating mammary gland, are highly active in the HMP shunt.
- Most of these tissues are involved in the biosynthesis of fatty acids and steroids, which are dependent on the supply of NADPH.
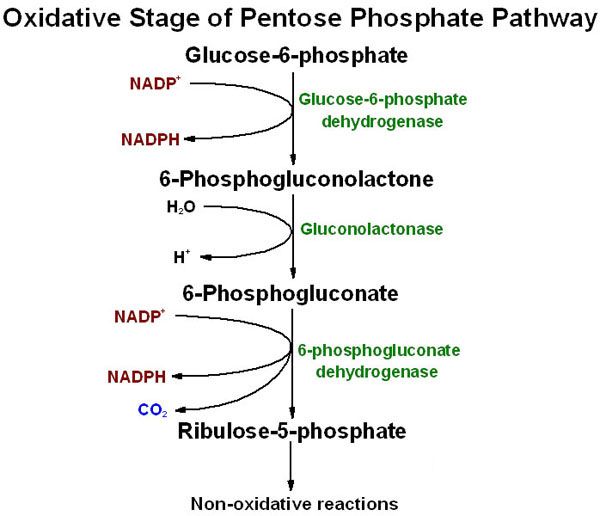
The Oxidative (Irreversible) Phase
This phase consists of three main reactions, starting with glucose-6-phosphate and culminating in the production of NADPH and ribulose-5-phosphate.
Key Concepts of the Oxidative Phase:
- Irreversible: The reactions in this phase are essentially unidirectional under physiological conditions.
- NADPH Production: This is the primary site of NADPH generation. Each molecule of glucose-6-phosphate entering this phase yields two molecules of NADPH.
- Substrate: Glucose-6-phosphate, which is also an intermediate in glycolysis.
- Location: Occurs in the cytosol.
The Three Reactions of the Oxidative Phase:
The oxidative phase involves the following sequential reactions:
1. Glucose-6-Phosphate Dehydrogenation (The Rate-Limiting Step)
- Enzyme: Glucose-6-Phosphate Dehydrogenase (G6PD)
- Reaction: Glucose-6-phosphate is oxidized, and NADP⁺ is reduced to NADPH. A lactone (cyclic ester) intermediate, 6-phosphogluconolactone, is formed.
- Equation:
Glucose-6-phosphate + NADP⁺ → 6-Phosphogluconolactone + NADPH + H⁺ - Significance: This is the rate-limiting and committed step of the entire Pentose Phosphate Pathway. The activity of G6PD is highly regulated.
2. Hydrolysis of 6-Phosphogluconolactone
- Enzyme: 6-Phosphogluconolactonase
- Reaction: The lactone ring is hydrolyzed to an open-chain carboxylic acid, 6-phosphogluconate.
- Equation:
6-Phosphogluconolactone + H₂O → 6-Phosphogluconate - Significance: This step prepares the molecule for the second oxidative reaction.
3. Oxidative Decarboxylation of 6-Phosphogluconate
- Enzyme: 6-Phosphogluconate Dehydrogenase
- Reaction: 6-phosphogluconate undergoes oxidative decarboxylation, meaning it is oxidized (another molecule of NADP⁺ is reduced to NADPH) and a molecule of CO₂ is released. The product is Ribulose-5-phosphate.
- Equation:
6-Phosphogluconate + NADP⁺ → Ribulose-5-phosphate + NADPH + H⁺ + CO₂ - Significance: This reaction generates the second molecule of NADPH and the first pentose phosphate, which serves as the entry point into the non-oxidative phase.
Summary of the Oxidative Phase:
The net reaction for the oxidative phase is:
→
Ribulose-5-phosphate + 2 NADPH + 2 H⁺ + CO₂
Key Takeaways from the Oxidative Phase:
- Two molecules of NADPH are produced per molecule of glucose-6-phosphate.
- One molecule of CO₂ is released.
- Ribulose-5-phosphate (a pentose sugar) is the final product.
- G6PD is the critical, rate-limiting enzyme.
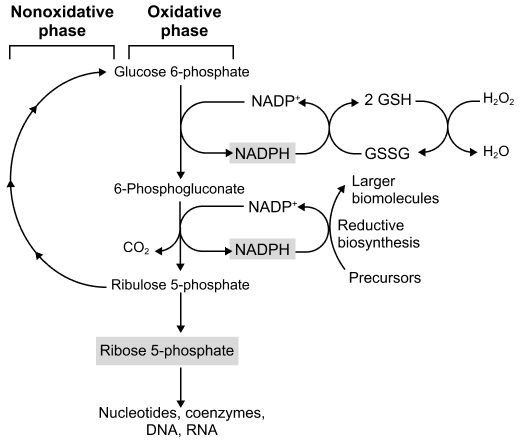
The Non-Oxidative (Reversible) Phase
The non-oxidative phase is a series of reversible reactions that interconvert various sugar phosphates. Its primary functions are:
- Conversion of Ribulose-5-Phosphate: To other pentose phosphates, including ribose-5-phosphate (essential for nucleotide synthesis).
- Recycling of Carbon Skeletons: To produce glycolytic intermediates (fructose-6-phosphate and glyceraldehyde-3-phosphate) from excess pentose phosphates, linking the PPP back to glycolysis.
- Flexibility: The reversibility allows the cell to adjust the production of ribose-5-phosphate and NADPH according to its needs.
Key Enzymes and Reactions of the Non-Oxidative Phase
This phase involves three main enzymes: an isomerase, an epimerase, and two transketolases/transaldolases.
1. Interconversion of Pentose Phosphates
The ribulose-5-phosphate generated in the oxidative phase needs to be converted into other pentose sugars.
a) Ribulose-5-Phosphate Isomerase
- Enzyme: Ribose-5-phosphate Isomerase
- Reaction: Converts the ketose sugar ribulose-5-phosphate into the aldose sugar ribose-5-phosphate. This is crucial as ribose-5-phosphate is the direct precursor for nucleotide synthesis.
- Equation:
Ribulose-5-phosphate ⇌ Ribose-5-phosphate
b) Ribulose-5-Phosphate Epimerase
- Enzyme: Xylulose-5-phosphate Epimerase
- Reaction: Converts ribulose-5-phosphate into another ketose sugar, xylulose-5-phosphate, which is important for subsequent transketolase reactions.
- Equation:
Ribulose-5-phosphate ⇌ Xylulose-5-phosphate
2. Transketolase and Transaldolase Reactions (The "Shunt" Part)
These two enzymes are responsible for moving two-carbon and three-carbon units between sugar phosphates to produce glycolytic intermediates.
a) Transketolase
- Enzyme: Transketolase
- Cofactor: Requires Thiamine Pyrophosphate (TPP).
- Function: Transfers a two-carbon (ketol) unit.
- First Reaction: Transfers 2 carbons from Xylulose-5-phosphate (5C) to Ribose-5-phosphate (5C), producing Glyceraldehyde-3-phosphate (3C) and Sedoheptulose-7-phosphate (7C).
b) Transaldolase
- Enzyme: Transaldolase
- Function: Transfers a three-carbon unit.
- Reaction: Transfers 3 carbons from Sedoheptulose-7-phosphate (7C) to Glyceraldehyde-3-phosphate (3C), producing Erythrose-4-phosphate (4C) and Fructose-6-phosphate (6C).
c) Second Transketolase Reaction
- Enzyme: Transketolase (again, requires TPP)
- Reaction: Transfers 2 carbons from another Xylulose-5-phosphate (5C) to Erythrose-4-phosphate (4C), producing another Glyceraldehyde-3-phosphate (3C) and another Fructose-6-phosphate (6C).
Overall Summary of the Non-Oxidative Phase
If 3 molecules of glucose-6-phosphate enter the oxidative phase, they produce 3 molecules of ribulose-5-phosphate and 6 NADPH. These 3 molecules of ribulose-5-phosphate are then processed through the non-oxidative phase:
These glycolytic intermediates can then enter glycolysis, be used for gluconeogenesis, or be recycled to continue the PPP.
Flexibility of the PPP
The reversibility of the non-oxidative phase is key, allowing the pathway to operate in different modes:
- If the cell needs more NADPH than ribose-5-phosphate: The oxidative phase is active, and pentose phosphates are recycled back to glucose-6-phosphate to maintain the flow.
- If the cell needs more ribose-5-phosphate than NADPH: The oxidative phase can be bypassed, and glycolytic intermediates can enter the non-oxidative phase in reverse to produce ribose-5-phosphate.
- If the cell needs both NADPH and ATP: The oxidative phase produces NADPH, and the non-oxidative phase converts pentose phosphates into F6P and G3P, which then enter glycolysis for ATP production.
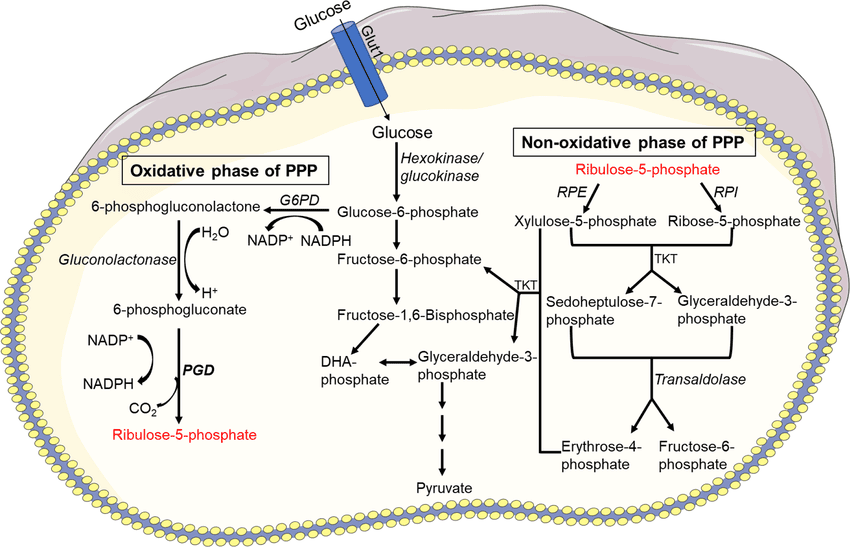
Primary Tissues/Cells of Activity
The activity of the PPP varies significantly among different tissues, directly reflecting their metabolic demands for its key products: NADPH and ribose-5-phosphate.
Tissues with High Demand for NADPH:
Liver (Hepatocytes)
The liver is a central metabolic hub with a high demand for NADPH for:
- Fatty acid synthesis
- Cholesterol and steroid synthesis
- Drug detoxification (cytochrome P450 system)
Adipose Tissue (Adipocytes)
Adipocytes are specialized for fat storage and have a very high demand for NADPH to support the massive amount of fatty acid synthesis that occurs here.
Red Blood Cells (Erythrocytes)
RBCs lack mitochondria and are constantly exposed to oxidative stress. The PPP is their only source of NADPH for antioxidant defense, used to maintain reduced glutathione (GSH) and protect the cell.
Steroidogenic Tissues
Tissues like the adrenal cortex, testes, and ovaries are primary sites of steroid hormone synthesis and have a high demand for NADPH for these hydroxylation reactions.
Mammary Gland (Lactating)
During lactation, the mammary gland synthesizes large amounts of fatty acids for milk production, requiring a high supply of NADPH.
Tissues with High Demand for Ribose-5-Phosphate:
Rapidly Dividing Cells
Tissues like bone marrow, skin, intestinal mucosa, and tumors are continuously proliferating and require constant DNA and RNA synthesis. They have a high demand for ribose-5-phosphate for nucleotide synthesis.
The non-oxidative phase can be reversed in these cells to primarily produce ribose-5-phosphate from glycolytic intermediates.
Regulation
The regulation of the Pentose Phosphate Pathway primarily occurs at its committed and rate-limiting step, catalyzed by Glucose-6-Phosphate Dehydrogenase (G6PD). The non-oxidative phase is primarily driven by substrate availability.
1. Regulation of Glucose-6-Phosphate Dehydrogenase (G6PD)
G6PD is the most important regulatory enzyme of the PPP. Its activity is controlled by:
a) Substrate Availability (Glucose-6-Phosphate)
Higher levels of G6P generally lead to increased G6PD activity.
b) Product Inhibition by NADPH (The Primary Regulator)
- NADPH is a potent competitive inhibitor of G6PD. This is the most crucial regulatory mechanism.
- When the cellular concentration of NADPH is high, it binds to G6PD and inhibits its activity, reducing further NADPH production.
- Conversely, when NADPH is low (and NADP⁺ is high), inhibition is relieved, and G6PD activity increases.
- Therefore, the ratio of NADPH/NADP⁺ is the primary determinant of the flux through the oxidative phase.
c) Transcriptional Regulation (Gene Expression)
The synthesis of G6PD can be regulated at the gene expression level. For example, a high-carbohydrate diet and insulin can lead to an increase in the synthesis of G6PD, increasing the capacity to produce NADPH for fatty acid synthesis.
2. Regulation of the Non-Oxidative Phase
The reversible reactions are primarily regulated by the availability of substrates and the cell's demand for products.
- If the cell needs ribose-5-phosphate, the equilibrium shifts towards its production.
- If the cell needs to recycle carbons back into glycolysis, the equilibrium shifts towards F6P and G3P.
3. Interplay with Glycolysis
The PPP and glycolysis compete for the common substrate, glucose-6-phosphate.
- High demand for NADPH and/or ribose-5-phosphate directs G6P into the PPP.
- High demand for ATP favors glycolysis.
Physiological Roles of NADPH
NADPH, produced almost exclusively by the PPP, plays essential roles in maintaining cellular homeostasis and facilitating various metabolic processes.
Reductive Biosynthesis
NADPH provides the electrons (reducing power) necessary for many synthetic (anabolic) reactions. Key examples include:
- Fatty Acid Synthesis: A major consumer of NADPH in the liver, adipose tissue, and lactating mammary gland.
- Cholesterol and Steroid Hormone Synthesis: Involves several NADPH-dependent reduction steps in the liver, adrenal cortex, and gonads.
- Deoxyribonucleotide Synthesis: The conversion of ribonucleotides to deoxyribonucleotides for DNA synthesis ultimately relies on NADPH.
Antioxidant Defense
NADPH is crucial for protecting cells from damage by Reactive Oxygen Species (ROS). It maintains the cellular defense system through its role in the glutathione system.
- Glutathione Reductase: This enzyme uses NADPH to reduce oxidized glutathione (GSSG) back to its protective, reduced form (GSH).
GSSG + NADPH + H⁺ → 2 GSH + NADP⁺ - Glutathione Peroxidase: Reduced glutathione (GSH) is then used to detoxify hydrogen peroxide (H₂O₂) by converting it into water.
2 GSH + H₂O₂ → GSSG + 2 H₂O
Phagocytosis (Respiratory Burst)
In phagocytic immune cells (e.g., neutrophils), NADPH plays a critical role in the "respiratory burst."
NADPH Oxidase: This enzyme uses NADPH to produce superoxide radicals (O₂•⁻), which are then converted into other potent oxidants (like hydrogen peroxide) to kill engulfed bacteria and pathogens.
O₂ + NADPH → O₂•⁻ + NADP⁺ + H⁺Significance of the Hexose Monophosphate (HMP) Shunt / Pentose Phosphate Pathway (PPP)
The HMP Shunt holds paramount significance due to its unique role in generating two crucial products: pentoses and NADPH. Unlike glycolysis, its value lies in providing essential building blocks and reducing power for various anabolic and protective processes.
I. Importance of Pentoses
The HMP shunt converts hexoses into pentose sugars, with ribose-5-phosphate being the most important. These are indispensable for:
- Nucleic Acid Synthesis: Ribose-5-phosphate is a direct precursor for the ribose in RNA and, after reduction, the deoxyribose in DNA.
- Nucleotide Coenzyme Synthesis: Ribose is necessary for synthesizing vital coenzymes such as ATP, NAD⁺, FAD, and Coenzyme A.
II. Importance of NADPH
NADPH is a versatile reducing agent, distinct from NADH, and serves as a critical source of electrons for a wide array of anabolic and protective cellular functions.
1. Reductive Biosynthesis
NADPH provides reducing power for building complex molecules like fatty acids, cholesterol, steroid hormones, and amino acids.
2. Antioxidant Defense
NADPH is critical for regenerating reduced glutathione (GSH), which is used by glutathione peroxidase to neutralize harmful free radicals and peroxides, protecting cells from oxidative damage.
3. Erythrocyte Membrane Integrity
In red blood cells, the concerted action of NADPH and the glutathione system is vital for preserving the integrity of the cell membrane, protecting it from oxidative damage and preventing premature lysis (hemolytic anemia).
4. Prevention of Met-Hemoglobinemia
NADPH-dependent reductase systems are essential for keeping the iron within hemoglobin in its reduced (ferrous, Fe²⁺) state. This prevents the formation of met-hemoglobin (Fe³⁺), which cannot carry oxygen.
5. Detoxification of Drugs
The liver's microsomal cytochrome P450 monooxygenase system depends on NADPH to detoxify drugs and foreign substances by increasing their solubility for excretion.
6. Preservation of Lens Transparency
The eye's lens has a high concentration of NADPH, which is vital for protecting lens proteins from oxidative damage, thereby guarding against conditions like cataracts.
7. Macrophage Bactericidal Activity
In phagocytic cells, NADPH oxidase uses NADPH to generate large quantities of superoxide radicals in a process called the "respiratory burst." These reactive oxygen species are potent antimicrobial agents used to kill ingested bacteria.
Biochemistry: Pentose Phosphate Pathway
Test your knowledge with these 35 questions.
PPP Exam
Question 1/35
Exam Complete!
Here are your results, .
Your Score
33/35
94%