Infective Endocarditis (IE)
This master guide covers the complete pathophysiology, microbiology, clinical presentation, and management of Infective Endocarditis. As requested, the core lecture material is completely intact, heavily expanded with deeper clinical correlations, vivid analogies, and practical exam scenarios to ensure absolute mastery of the topic.
1. What is Infective Endocarditis? (The Basics)
Definition: Infective endocarditis (IE) is an infection of the endocardial surface of the heart. The endocardium is the smooth, innermost lining of the heart chambers and the heart valves. The term "infective" implies the physical, active presence of microorganisms (bacteria or fungi) invading this lining, resulting in a physical mass called a vegetation or lesion.
Deep Dive: What exactly is a vegetation? It is a bulky, deadly clump made of three things: host platelets, host fibrin (clotting material), and massive colonies of living microorganisms. Because it is physically attached to the heart, it moves with every heartbeat.
Where does it happen?
- Heart Valves: This is the most common site. The valves (Mitral, Aortic, Tricuspid, Pulmonary) are constantly opening and closing, dealing with high-pressure blood flow. If they get damaged, they are prime targets for infection.
- Septal Defects: If a patient has a hole in their heart (like a Ventricular Septal Defect - VSD), the blood aggressively shoots through that hole. This high-velocity "jet stream" damages the surrounding tissue on the opposite side of the hole, creating a rough, scarred patch where circulating bacteria can easily latch on.
- Mural Endocardium: This refers to the flat muscular walls of the heart chambers. Though less common than valves, infections can happen here if the wall is damaged by turbulent blood flow or foreign devices (like pacemaker wires rubbing against the tissue).
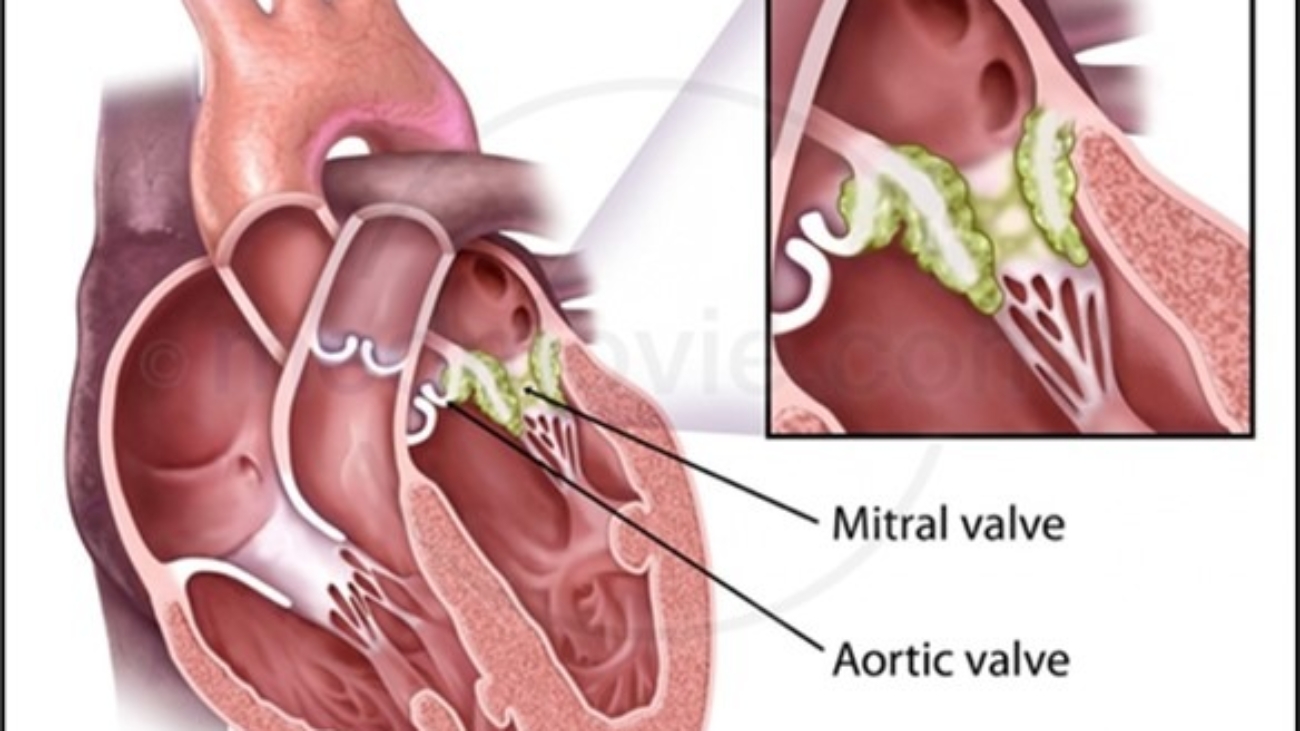
2. Anatomy Recap: The Valves of the Heart
To understand IE, you must deeply understand your valves and the path of blood flow. The heart has four main doors (valves) that keep blood flowing in one direction.
Pumps deoxygenated blood to the Lungs.
- Tricuspid Valve: Between the right atrium and right ventricle.
Clinical Pearl: This valve is most commonly infected in Intravenous (IV) Drug Users. Why? Because when dirty needles inject bacteria into a vein, the venous blood travels directly into the right side of the heart first. The tricuspid valve is the first door the bacteria hit. - Pulmonary Valve: Between the right ventricle and the pulmonary trunk. (Rarely infected).
Pumps oxygenated blood to the entire Body.
- Mitral Valve: Between the left atrium and left ventricle.
- Aortic Valve: Between the left ventricle and the aorta.
Note: Left-sided valves are under much higher pressure (working against systemic blood pressure) and experience more wear and tear. Therefore, they are the most common sites for IE in the general, non-IV-drug-using population.
3. Classification of IE: Acute vs. Subacute/Chronic
Historically, IE was divided based on how fast it killed the patient if left untreated. This is a crucial distinction for exams as it dictates both the causative bug and the clinical presentation.
A. Acute Infective Endocarditis
Analogy: Think of this as a rapid, violent home invasion. It happens incredibly fast and causes massive destruction.
- Progression: Follows a fulminant (severe and sudden) course. Death can occur in several days to less than 6 weeks without treatment.
- Clinical Picture: The patient looks incredibly sick. They will have a high-spiking fever, systemic toxicity (sepsis, dangerously low blood pressure, rapid heart rate), and severe leukocytosis (massively elevated white blood cell count).
- Valve Status: Because the bugs are so aggressive, Acute IE can attack and completely destroy perfectly normal, healthy heart valves.
- Causative Agents (The Aggressive Bugs):
- Staphylococcus aureus (The #1 cause of acute IE. Known for being highly destructive, producing enzymes that liquefy tissue).
- Streptococcus pyogenes (Group A Strep).
- Streptococcus pneumoniae.
- Neisseria gonorrhoeae (Rare today due to antibiotics, but a classic board-exam historical cause).
Clinical Scenario - Acute IE
A 28-year-old male with a history of heroin use is brought to the ER by his friends. He has a fever of 104°F (40°C), shaking chills, and looks extremely toxic and confused. When you listen to his heart, you hear a loud, brand-new systolic murmur that wasn't there last week. His right-sided tricuspid valve is being rapidly chewed apart by Staphylococcus aureus introduced via a dirty needle.
B. Subacute and Chronic Infective Endocarditis
Analogy: Think of this as termites slowly eating away at a house over months. It is insidious, sneaky, and gradual.
- Progression: Subacute leads to death in 6 weeks to 3 months. Chronic takes longer than 3 months.
- Valve Status: These bugs are weak. They usually CANNOT infect a healthy valve. They almost always occur in the setting of prior valvular disease (e.g., a patient with a history of rheumatic fever, a congenital bicuspid aortic valve, or mitral valve prolapse).
- Clinical Picture: Slow, indolent (lazy/painless) course. Symptoms are vague: low-grade fever (maybe 99.5°F - 100.5°F), heavy night sweats, unexplained weight loss, generalized fatigue, and "vague systemic complaints" (the patient just feels "off" for weeks).
- Causative Agents (The Sneaky Bugs):
- Viridans streptococci (These naturally live in your mouth and throat. They are less aggressive but will happily settle on a previously damaged valve because they produce dextrans that bind perfectly to scarred tissue).
Clinical Scenario - Subacute IE
A 65-year-old woman with a known childhood history of rheumatic fever (which permanently scarred her mitral valve) visits her dentist for a deep tooth extraction. Two months later, she goes to her primary care doctor complaining of severe fatigue, losing 10 pounds without trying, and waking up drenched in night sweats. Blood cultures reveal Viridans streptococci. The bacteria entered her bloodstream during the dental work and slowly grew on her old, scarred heart valve over the last 8 weeks.
4. Pathogenesis (How exactly does the vegetation form?)
This is a step-by-step process of how bacteria build a fortress on a heart valve. Examiners love testing these steps because it explains why treatment is so difficult.
- Endothelial Damage (The Scratch): The smooth valve surface must first be altered (trauma, turbulent blood flow, or metabolic changes). Bacteria slip right off smooth, healthy tissue. They need a rough patch to stick to.
- Formation of NBTE (The Sticky Band-Aid): The body senses the damaged valve and tries to heal it. Platelets and fibrin (clotting proteins) rush to the site and deposit there. This forms a sterile (no bacteria yet) clot called Nonbacterial Thrombotic Endocarditis (NBTE). It is essentially a sticky web.
- Bacteremia (The Invasion): Bacteria enter the bloodstream. This can happen from mucous membrane trauma (brushing teeth aggressively, dental work, chewing), skin infections, IV drug use, or surgery.
- Adherence and Colonization (The Landing): The circulating bacteria find the sticky NBTE web, latch onto it (often using special adherence proteins), and begin to multiply (colonize).
- The Mature Vegetation (The Fortress): Once attached, the bacteria trigger the body to lay down more fibrin and platelets over them. The surface rapidly covers the bacteria in a protective sheath. Now, the bacteria are encased inside a clot. This environment is perfect for them—they are protected from the bloodstream's roaming white blood cells (neutrophils) and can divide continuously, creating a bulky mass called a mature vegetation.
5. Etiologic Agents (The Microbes that cause IE)
Here is a detailed breakdown of the bugs you need to memorize, where they come from, and what they do:
| Organism | Source / Characteristics | Clinical Association |
|---|---|---|
| Viridans streptococci | Normal mouth/oral flora. Low virulence. Produces dextrans. | Classically causes subacute IE after dental procedures on already damaged valves. |
| Enterococci spp. | Normal gut and urinary tract flora. Highly resistant to many antibiotics. | Often causes IE after GI/GU procedures (like a colonoscopy, or urinary catheter insertion in older men with enlarged prostates). |
| Staphylococcus aureus | Highly virulent skin bug. Produces coagulase and tissue-destroying toxins. | Causes acute, destructive IE. Strongly associated with IV drug users, skin infections, and surgical wounds. Frequently attacks the Tricuspid valve. |
| Coagulase-negative staphylococcus (CoNS) | e.g., Staphylococcus epidermidis. Normal skin flora. This bug LOVES plastic and metal. | The #1 cause of prosthetic valve endocarditis (artificial valves) and pacemaker wire infections. |
| Gram-negative aerobic bacilli | Various (e.g., Pseudomonas). | Less common overall, but seen in healthcare settings or IV drug users. |
| Fungi | Candida spp., Aspergillus, Cryptococcus, Histoplasma. | Very difficult to treat. Usually seen in severely immunocompromised patients, patients on long-term IV antibiotics, or patients with central venous catheters. Fungal vegetations are famously massive and tend to break off easily (embolize). |
The HACEK Group
A special group of slow-growing, Gram-negative bacteria that live in the mouth/throat. They are notoriously difficult to grow in traditional lab cultures (often referred to as "culture-negative endocarditis" because the lab tests keep coming back empty while the patient dies). They include:
- Haemophilus parainfluenzae / aphrophilus
- Actinobacillus actinomycetemcomitans
- Cardiobacterium hominis
- Eikenella corrodens (Heavily associated with human bite wounds or "clenched-fist" bar fight injuries!)
- Kingella kingae
6. Clinical Manifestations (What happens to the patient's body?)
IE doesn't just damage the heart; it is a systemic disease that affects the whole body in four main ways:
- Local Intracardiac Complications:
- The bacteria eat away at the valve, causing holes or snapping the strings holding the valve (chordae tendineae). This leads to massive valve leakage (regurgitation) and sudden heart failure (fluid backing up into the lungs or body).
- The infection can bore into the heart muscle, creating an abscess. This abscess can cut off the heart's electrical wiring, causing deadly arrhythmias (heart blocks).
- Bland or Septic Embolization:
- The vegetation is fragile. Pieces of it (emboli) can break off and travel through the blood to block arteries anywhere in the body.
- If it blocks a vessel, it's a "bland" embolus (causes a stroke in the brain, or a dead spot/infarct in the spleen or kidney).
- Because the piece is packed with bacteria, it's a "septic" embolus, meaning wherever it lands, it starts a massive new infection (like a brain abscess, or if it breaks off the right side of the heart, it causes multiple lung abscesses).
- Constant Bacteremia:
- The vegetation constantly sheds bacteria into the blood. This leads to continuous fevers and can seed other organs, causing metastatic infections (e.g., infection settling in the spine - osteomyelitis).
- Immunopathologic Factors (Circulating Immune Complexes):
- The immune system produces antibodies against the bacteria. These antibodies bind to the bacteria fragments, creating large "immune complexes."
- These complexes get stuck in tiny capillaries around the body, causing painful inflammation in the skin, eyes, and kidneys.
Classic Physical Signs of IE:
- Osler's Nodes: Painful, raised red bumps on the pads of fingers and toes (caused by immune complex deposition). Memory trick: "O" for Osler, "O" for Ouch!
- Janeway Lesions: Painless, flat red spots on the palms and soles. (These are actually tiny micro-abscesses caused by bits of septic emboli breaking off the heart and landing in the hands/feet. Because they are dead tissue/abscesses, the nerve endings are destroyed, making them painless).
- Roth's Spots: White spots surrounded by hemorrhage seen when looking into the retina of the eye (immune complexes).
- Glomerulonephritis: Immune complexes get stuck in the kidney filters, causing blood in the urine (hematuria) and kidney failure.
- Rheumatoid Factor: A blood test that can turn positive simply due to chronic, prolonged immune system stimulation.
7. Diagnosis: The Duke Criteria
Because diagnosing IE is tricky (fever and fatigue can mean a thousand different diseases) and treating it requires massive doses of IV antibiotics for weeks, doctors use a strict scoring system called the Duke Criteria to prove a patient has it.
To diagnose IE definitively, you need: 2 Major criteria, OR 1 Major + 3 Minor, OR 5 Minor criteria.
MAJOR CRITERIA (The heavy hitters - Proof of Bug and Proof of Damage)
- Positive Blood Cultures (Proof the bug is in the blood):
- Must be a typical IE organism (Viridans, S. aureus, HACEK, Enterococci) from two separate blood draws.
- OR, persistently positive blood cultures drawn more than 12 hours apart (proving the bacteria are constantly swimming in the blood, not just a one-time accidental contamination from a dirty needle during the blood draw).
- OR, a single positive culture for Coxiella burnetii (The cause of Q fever. It gets a special exception because it is highly specific for a rare, intracellular type of IE and almost impossible to culture normally).
- Evidence of Endocardial Involvement (Seeing the damage):
- A positive Echocardiogram (ultrasound of the heart) showing a physical mass (vegetation) swinging on a valve, a hole/abscess, or a prosthetic valve coming loose (dehiscence).
- OR, the sudden appearance of a brand new heart murmur (specifically a regurgitation/leaking murmur, because the valve no longer closes properly).
MINOR CRITERIA (The supporting clues)
- Predisposition: Patient has a known bad heart valve or uses IV drugs.
- Fever: ≥ 38.0°C (100.4°F).
- Vascular Phenomena: Emboli to organs, septic lung infarcts, mycotic aneurysms (infected weakened blood vessels that can burst), brain bleeds, Janeway lesions.
- Immunologic Phenomena: Glomerulonephritis, Osler's nodes, Roth's spots, positive Rheumatoid factor.
- Microbiologic Evidence: Positive blood cultures that don't quite meet the strict "Major" rules above.
8. Treatment (Why is it so difficult?)
Treating IE is one of the hardest tasks in infectious disease medicine.
Complete eradication takes weeks (usually 4 to 6 weeks) of high-dose, continuous Intravenous (IV) antibiotics. Patients often go home with a PICC line (a long IV in their arm) to finish therapy. Relapse is not unusual.
Why is it so hard to kill?
- The Fortress: The bacteria are trapped inside the fibrin meshwork (the vegetation).
- Avascular Nature: Heart valves themselves do not have a dedicated blood supply. The vegetation has absolutely no blood vessels inside of it. Therefore, the body's white blood cells (phagocytes) cannot "swim" inside to eat the bacteria.
- Antibiotic Penetration: Because there's no blood supply, antibiotics have a terrible time penetrating the center of the mass. This is why bacteriostatic drugs (which just stop bacteria from growing) fail. You MUST use bactericidal drugs (drugs that outright explode the bacteria).
- Insane Bacterial Density: The bacteria inside this protected fort reach tremendous population densities, often 109 to 1010 CFU/g (Colony Forming Units per gram of tissue). They run out of nutrients and go into a dormant, slow-growing state. Most antibiotics (like Penicillin) only work well on rapidly dividing bacteria, making dormant bugs highly resistant.
Surgery: Often, antibiotics aren't enough. If the valve is destroyed and the patient goes into heart failure, if the vegetation is massive (e.g., >10mm) and about to break off, or if it's a fungal infection, medical therapy fails. The chest must be opened, the infected valve cut out, and a new artificial valve sewn in.
9. Infections of Prosthetic Valves and Cardiovascular Devices
Since the 1950s, medical technology has given us artificial valves, pacemakers, and implantable defibrillators (ICDs). While life-saving, introducing plastic and metal into the body creates a massive risk for infection.
How do these devices get infected? (Three Ways)
- At the time of surgery (Microbial Contamination): Bacteria from the patient's skin or the operating room air land on the device right as the surgeon implants it. (Nosocomial/hospital-acquired).
- Hematogenous Dissemination: The device is implanted perfectly. Years later, the patient gets a severe blood infection (bacteremia) from another source (like a kidney infection or dental abscess), and the bacteria travel through the blood and stick to the artificial valve.
- Contiguous Spread: An infection happening in tissue right next door to the device physically spreads and touches the device.
The Biofilm Superpower
Artificial surfaces lack the body's natural defenses. Bacteria love this.
- Adherence: Bacteria, especially Staphylococci, attach to host proteins (like fibronectin) that rapidly coat the implanted device after surgery.
- MSCRAMMs: S. aureus uses specialized sticky proteins on its surface called MSCRAMMs (Microbial Surface Components Recognizing Adhesive Matrix Molecules). Think of these as microscopic Velcro hooks that lock onto the artificial valve. This process is controlled by specific bacterial genes (the global gene regulators like agr and sar).
- The Biofilm: Once attached, the staphylococci secrete a thick, slimy, glue-like substance called an extracellular polymeric substance (EPS) matrix. This creates a Biofilm.
- Why Biofilms are dangerous: An assemblage of microbes living inside a biofilm is practically invincible. They are irreversibly attached to the foreign body. Antibiotics bounce right off the slime layer, and immune cells cannot penetrate it.
Causative Factors & Clinical Manifestation for Devices
- Coagulase-Negative Staphylococci (CoNS - e.g., S. epidermidis) & Corynebacterium spp.: Normal skin flora. They commonly infect devices during placement. They are highly associated with hospital settings (nosocomial) and are notorious for multidrug resistance (like MRSA).
- Symptoms: These bugs are slow-growing. A patient might present months after surgery with a low-grade fever, weight loss, malaise, and muscle aches (myalgias).
- S. aureus, β-hemolytic streptococci, Pseudomonas aeruginosa, and Candida: These are highly aggressive.
- Symptoms: Acute presentation with profound sepsis. High fever, shock, rapid deterioration. Pseudomonas is especially dangerous and resistant to many drugs.
Diagnosis and Treatment for Device Infections
- Diagnosis: Relies heavily on continuous Blood Cultures to identify the specific organism, alongside Echocardiography (Transesophageal echo is preferred to see behind the metal artifacts of the artificial valve).
- Treatment Protocol:
- Pathogen-specific antimicrobial therapy: Hitting the bug with the exact IV antibiotic it is susceptible to (often Vancomycin + Rifampin for resistant Staph in biofilms).
- Device Removal: Because of the invincible biofilm, you usually cannot cure the patient until the surgeon goes back in, rips out the infected pacemaker wires or artificial valve, and clears out the surrounding dead tissue.
Quick Quiz
Infective Endocarditis Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Infective Endocarditis Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.