Neoplasia & Oncology
Neoplasia & Oncology
Neoplasia is one of the most heavily tested topics in medical exams. To master this, you must shift your thinking from normal physiology to pathological, rogue biology. By the end of this exhaustive guide, you will master:
- The precise nomenclature (naming rules) of tumors, including the famous "exceptions" that appear on every exam.
- The distinct morphological and behavioral differences between Benign vs. Malignant tumors.
- The intricate genetic mechanisms (Oncogenes vs. Tumor Suppressor Genes) and viral etiologies of cancer.
- The systemic effects of cancer, especially the high-yield Paraneoplastic Syndromes.
- The crucial difference between Grading and Staging.
1. Definitions & Anatomy of a Tumor
Before we classify tumors, we must define exactly what we are dealing with. The terminology is precise and highly testable.
- Cancer: A genetic disorder caused by DNA mutations. It is not a single disease, but a collection of disorders driven by corrupted genetic code.
- Neoplasia: Literally translates to "new growth." A neoplasm is an abnormal mass of tissue whose growth exceeds and is uncoordinated with that of normal tissues, and persists even after the stimuli that evoked the change is removed.
- Tumor: Literally means "Swelling." While originally a sign of inflammation, in modern medicine, "tumor" is used interchangeably with "neoplasm." Tumors can be Benign (innocent, localized) or Malignant (cancerous, spreading).
- Oncology: The clinical and scientific study of tumors (from the Greek oncos, meaning tumor).
The Two Basic Components of ALL Tumors
Whether a tumor is benign or malignant, it is constructed of two main parts. Think of a tumor like a rogue city:
- The Parenchyma: These are the transformed neoplastic cells. These are the actual mutated "bad guys." The parenchyma determines the biological behavior of the tumor and is what we use to name the tumor.
- The Stroma: This is the supporting, host-derived, non-neoplastic tissue. It is made up of connective tissue, blood vessels, and host-derived inflammatory cells.
2. Nomenclature: How We Name Tumors
Tumor nomenclature is based entirely on the parenchyma (the cell of origin). This is a heavily tested area where suffixes give away the diagnosis.
A. Benign Tumors
General Rule: Benign tumors are designated by attaching the suffix "-oma" to the cell type from which the tumor arises.
- Fibroblast + oma = Fibroma (Benign tumor of fibrous tissue).
- Chondrocyte (cartilage) + oma = Chondroma.
- Lipocyte (fat) + oma = Lipoma (e.g., benign lipoma of the small intestine or under the skin).
- Osteocyte (bone) + oma = Osteoma.
- Smooth Muscle + oma = Leiomyoma (e.g., uterine fibroids are actually benign leiomyomas!).
Special Benign Epithelial Tumors:
- Papillomas: Benign epithelial neoplasms growing on any surface that produce microscopic or macroscopic finger-like fronds (e.g., Squamous cell papilloma).
- Polyp: A mass that projects above a mucosal surface (like in the gut/colon) to form a macroscopically visible structure. (Note: A polyp is a descriptive macroscopic term. It can technically be benign or malignant, but most are benign adenomatous polyps).
- Cystadenomas: Hollow, cystic masses that typically arise in the ovary.
- Adenoma: A benign epithelial tumor that either arises from glands or forms a glandular pattern.
B. Malignant Tumors (CANCERS)
Malignant tumors are named based on their embryological origin (Mesenchymal vs. Epithelial).
Mesenchymal / Connective Tissue Origin
Malignant tumors arising in solid mesenchymal tissues (bone, cartilage, fat, muscle, blood vessels).
- Fibrosarcoma
- Liposarcoma
- Chondrosarcoma
- Osteogenic sarcoma (Osteosarcoma)
- Angiosarcoma (blood vessels)
- Leiomyosarcoma (smooth muscle)
- Rhabdomyosarcoma (skeletal/striated muscle)
Liquid Mesenchymal: Malignancies arising from blood-forming cells are called Leukemias or Lymphomas.
Epithelial Origin
Malignant neoplasms of epithelial cell origin (regardless of which of the 3 germ layers the epithelium came from). Carcinomas are the most common cancers in adults.
- Adenocarcinoma: Carcinomas that grow in a glandular pattern (e.g., Colon adenocarcinoma, Prostate adenocarcinoma).
- Squamous cell carcinoma: Carcinomas that produce squamous cells (often arising in the skin, cervix, or lung). Microscopically, these often show "pink keratin pearls" or "intercellular bridges".
CRITICAL EXAM TRAPS: The Malignant "-omas"
The suffix "-oma" usually means benign. However, examiners LOVE to test the famous exceptions that sound benign but are absolutely, lethally MALIGNANT. Memorize these:
- Melanoma: Malignant tumor of melanocytes (skin).
- Lymphoma: Malignant tumor of lymphoid tissue.
- Mesothelioma: Malignant tumor of the mesothelium (pleura of lung, strongly linked to asbestos exposure).
- Seminoma: Malignant tumor of testicular germ cells.
C. Mixed Tumors & Teratomas
- Mixed Tumors: Arise from a single clone of cells capable of differentiating into more than one cell type (e.g., Pleomorphic adenoma of the salivary gland, containing both epithelial tissue and cartilage-like stroma).
- Teratoma: A special type of mixed tumor containing recognizable mature or immature cells/tissues derived from more than one germ cell layer (endoderm, mesoderm, ectoderm), and sometimes all three!
- Origin: They originate from totipotential germ cells (cells with the capacity to turn into ANY tissue in the body). These normally reside in the ovary and testis, or abnormally in midline embryonic rests.
- Pathology: Because germ cells can differentiate into anything, a teratoma might contain hair, bone, epithelium, muscle, fat, and teeth all thrown together in a disorganized "helter-skelter" fashion! (e.g., Ovarian cystic teratoma / dermoid cyst).
D. The "Fake Tumors" (Non-Neoplastic Lesions)
These two are often tested to confuse you. They sound like tumors, but they are congenital anomalies or disorganized normal tissue.
A mass of disorganized tissue indigenous (native) to that particular site. It is the right tissue, just messy.
Example: A disorganized mass of normal lung cartilage and respiratory epithelium in the lung, or bile ducts inside the liver.
Note: Newer evidence shows some have clonal mutations and are now considered benign neoplasms, but classically they are disorganized native tissue.
A congenital anomaly consisting of a heterotopic (out of place) nest of normal cells. It is perfectly normal tissue, completely lost.
Example: A perfectly normal, tiny piece of pancreatic tissue found living inside the wall of the stomach. It functions normally, it is just in the wrong zip code.
Mnemonic to remember the difference:
Hamartoma = Here (Right tissue, wrong organization).
Choristoma = Completely out of place (Normal tissue, wrong location).
3. Characteristics of Benign vs. Malignant Neoplasms
There are four fundamental features used by pathologists to distinguish a benign tumor from a malignant cancer. Metastasis is the absolute most reliable discriminator.
1. Differentiation and Anaplasia
Differentiation refers to the extent to which neoplastic cells resemble their normal parenchymal cells of origin, both morphologically (how they look) and functionally (what they do).
- Benign Tumors: Usually well-differentiated. A benign lipoma looks exactly like normal fat cells under a microscope. Mitoses (cell divisions) are rare and look normal.
- Malignant Tumors: Range from well-differentiated to entirely undifferentiated.
Anaplasia literally means "backward formation" and refers to a complete lack of differentiation. Anaplastic cells look nothing like their tissue of origin. Anaplasia is a hallmark of malignancy.
Microscopic Features of Anaplasia (Highly Testable)
If a pathologist sees these features on a slide, they are looking at aggressive cancer:
- Pleomorphism: Extreme variation in the size and shape of the cells and their nuclei. (They don't look uniform like healthy cells).
- Nuclear Abnormalities:
- Extreme hyperchromatism (darkly staining, ink-black nuclei because of massive amounts of mutated, condensed DNA).
- Variation in nuclear size/shape. Prominent single or multiple nucleoli.
- Abnormal Nuclear-to-Cytoplasmic (N:C) ratio. (Normal is 1:4 or 1:6; cancer is often 1:1, meaning the massive, mutated nucleus takes up the entire cell!).
- Atypical Mitoses: You see cells dividing rapidly, but the mitotic spindles are bizarre, tripolar, or multipolar (looks like a Mercedes-Benz sign under the microscope), not normal bipolar spindles.
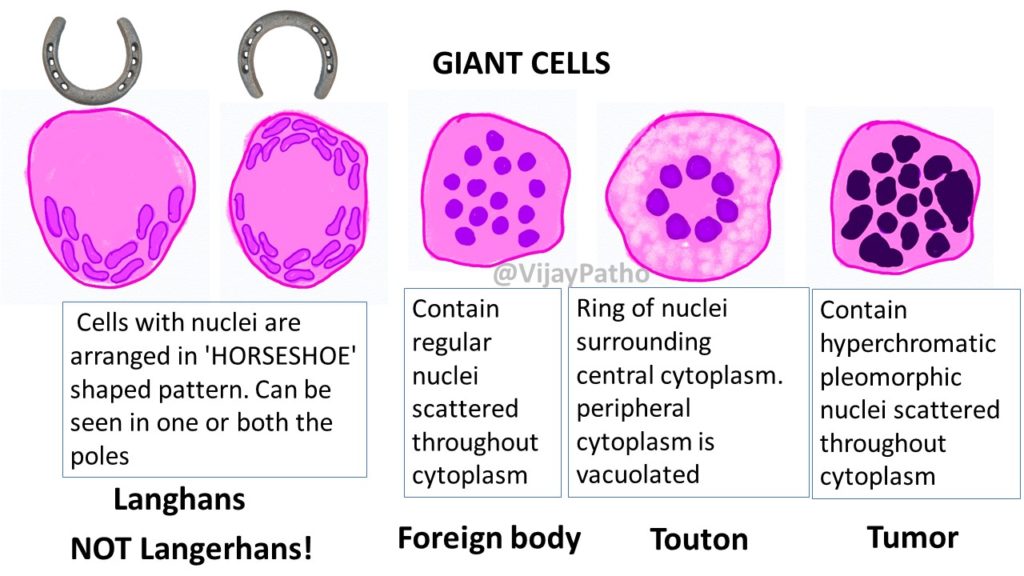
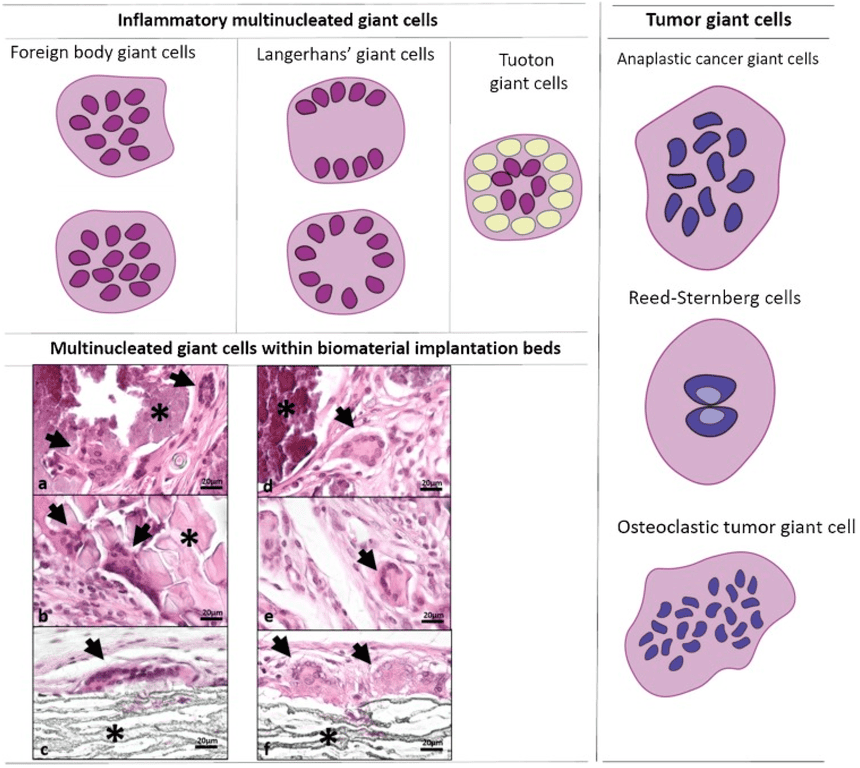
- Tumor Giant Cells: Massive cells with single huge polymorphic nuclei or multiple nuclei (not to be confused with foreign body giant cells).
2. Dysplasia & Carcinoma in Situ (The Pre-Cancer Spectrum)
Dysplasia means "disorderly proliferation." It is encountered primarily in epithelia (e.g., the cervix or respiratory tract).
- Dysplastic epithelium shows a loss in the uniformity of individual cells and a loss in their architectural orientation.
- It is a precursor to cancer, but it is not yet cancer because it has not broken through the basement membrane.
- Carcinoma in situ (CIS): When dysplastic changes are so severe that they involve the entire thickness of the epithelium, it is called CIS. It is the absolute final pre-invasive stage of cancer. Once it breaches the basement membrane into the stroma, it officially becomes invasive carcinoma.
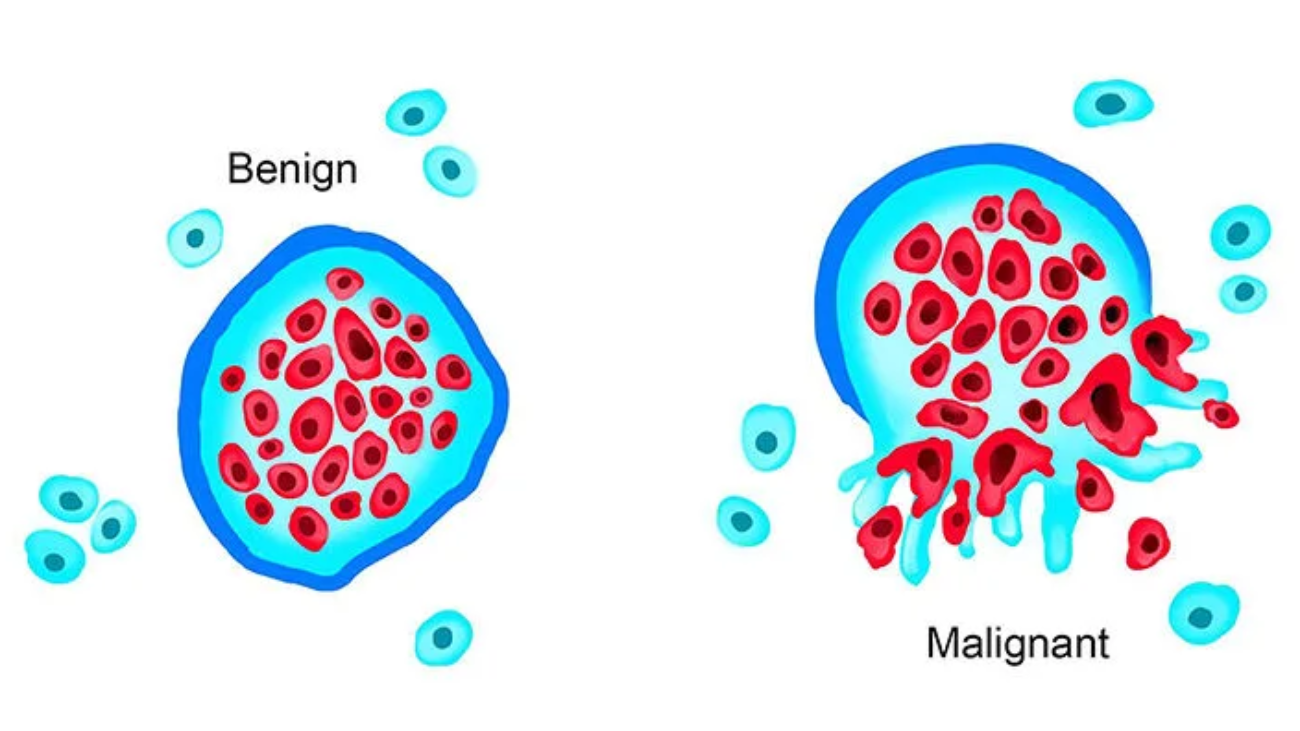
3. Local Invasion (Encapsulation vs. Infiltration)
- Benign Tumors: Grow as cohesive, expansile masses that remain localized. Because they grow slowly, they compress the surrounding normal tissue, causing the host fibroblasts to deposit a fibrous capsule. This capsule makes the tumor discrete, moveable, and easily excisable by a surgeon (surgical enucleation).
Exception Exam Trap: Not all benign tumors have capsules! Hemangiomas (benign blood vessel tumors) are not encapsulated and can be messy to remove. - Malignant Tumors (Cancer): Growth is accompanied by progressive infiltration, invasion, and destruction of surrounding tissues. They do not have well-defined capsules. They send out "crab-like" penetrating roots into normal tissue. (Note: Invasiveness is the feature that most reliably distinguishes local cancers from benign tumors).
4. Metastasis
Metastasis unequivocally marks a tumor as malignant. By definition, benign neoplasms DO NOT metastasize.
Metastasis is the spread of a tumor to sites that are physically discontinuous with the primary tumor. The invasiveness of cancers allows them to penetrate blood vessels, lymphatics, and body cavities to spread.
4. Dissemination Pathways (How Cancer Spreads)
Cancers spread via three main routes. Examiners love matching the cancer type to the route of spread:
Occurs when neoplasms invade a natural body cavity (like the peritoneum or pleura).
Classic Example: Ovarian cancer frequently penetrates the surface of the ovary and coats the entire peritoneal cavity with cancerous "seeds." This often leads to massive abdominal fluid accumulation (ascites).
The tumor invades lymphatic vessels and travels to regional lymph nodes.
This is the most typical pathway for CARCINOMAS (epithelial cancers like breast cancer).
Sentinel Lymph Node: The very first regional lymph node that receives lymph flow from a primary tumor. Surgeons inject blue dye or radiolabeled tracers into the tumor to find this exact node. If a biopsy of the sentinel node is negative for cancer, it means the cancer likely hasn't spread further down the chain, sparing the patient from massive, debilitating lymph node removal surgeries.
The tumor invades veins and travels through the bloodstream. (Arteries are harder to penetrate due to their thick muscular walls).
This is the favored pathway for SARCOMAS (connective tissue cancers).
Because all venous blood eventually drains through the liver (portal system) and the lungs (caval system), the LIVER and LUNGS are the most common secondary sites for metastatic tumors.
Exam Exception: Renal Cell Carcinoma and Hepatocellular Carcinoma are carcinomas, but they famously prefer to spread via the blood (hematogenous) by invading the renal vein and portal vein, respectively!
Note: There are numerous interconnections between the lymphatic and vascular systems, so all forms of cancer may eventually disseminate through either or both systems.
5. Rate of Growth
In general, rapid growth signifies malignancy, but many malignant tumors grow slowly, so growth rate alone is not a perfect discriminator.
Tumor growth rate is determined by three factors:
- Doubling time of the tumor cells.
- The Growth Fraction: The fraction of tumor cells that are actively in the replicative pool (actively dividing in the cell cycle).
- Cell Loss: The rate at which cells are shed, die by apoptosis, or are lost due to a lack of blood supply in the growing lesion.
Clinical Correlate: Why does Chemotherapy cause hair loss?
Traditional chemotherapy drugs do not "know" which cell is cancer. They simply target and kill any cell that is actively dividing (cells in the Growth Fraction). Cancers usually have a high growth fraction, so they take heavy damage. However, your hair follicles, GI tract lining, and bone marrow also have naturally high growth fractions to keep your body renewed. The chemotherapy destroys these healthy dividing cells too, resulting in alopecia (hair loss), severe nausea, and anemia/immunosuppression.
6. Etiology: Risk Factors and Pre-disposing Conditions
A. Environmental Risk Factors
- Diet: High fat, low fiber linked to colorectal cancer.
- Smoking: Heavily linked to lung squamous cell carcinoma, mouth, throat, and notably bladder cancers (carcinogens are excreted in urine).
- Alcohol consumption: Liver, mouth, esophagus cancers.
- Reproductive history: Nulliparity (no pregnancies) increases risk of breast/endometrial cancer due to a lifetime of prolonged, uninterrupted estrogen cycles.
- Infectious agents: Viruses (HPV, Hepatitis) and Bacteria (H. pylori).
- Age: Most cancers occur between ages 55-75 years. This is simply because it takes decades for a cell to accumulate enough random somatic mutations to become cancerous.
B. Acquired Predisposing Conditions (Pre-Malignant Lesions)
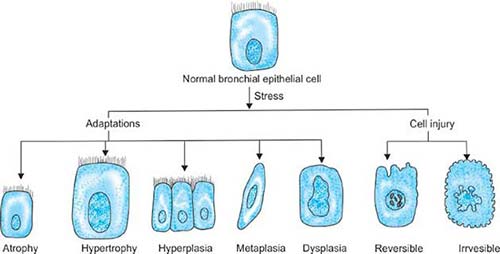
Certain chronic irritations cause tissues to change (metaplasia) and eventually become disorderly (dysplasia). These are high-risk states for cancer:
| Condition (The Precursor) | Associated Cancer Risk |
|---|---|
| Squamous metaplasia and dysplasia of bronchial mucosa (seen in habitual smokers). | Lung cancer (Squamous cell carcinoma). |
| Endometrial hyperplasia and dysplasia (seen in women with unopposed estrogenic stimulation, e.g., PCOS or obesity). | Endometrial carcinoma. |
| Leukoplakia (thick, un-scrapeable white patches) of oral cavity, vulva, and penis. | Squamous cell carcinoma. |
| Villous adenoma of the colon. | High risk for progression to Colorectal carcinoma. |
| Barrett's Esophagus (acid reflux changing lower esophagus to intestinal columnar epithelium). | Esophageal Adenocarcinoma. |
7. The Genetics of Cancer (Carcinogenesis)
Cancer is fundamentally a genetic disease. No single mutation is sufficient to transform a normal cell into a cancer cell. Carcinogenesis is a multistep process resulting from the accumulation of multiple genetic alterations. Genetic evolution shaped by Darwinian selection explains why cancers become more aggressive and resistant to therapy over time (the cells that survive chemo mutate and reproduce).
The Four Main Classes of Cancer Genes
1. Oncogenes
Mutated versions of normal growth genes (proto-oncogenes). When mutated, they are permanently turned "ON," inducing a transformed phenotype by promoting unchecked cell growth.
Analogy: A brick stuck on the gas pedal of a car.
2. Tumor Suppressor Genes (TSGs)
Genes that normally prevent uncontrolled growth. When these are mutated or lost, the cell loses its brakes, allowing the transformed phenotype to develop.
Analogy: The brakes of the car are completely cut.
3. Genes that regulate Apoptosis
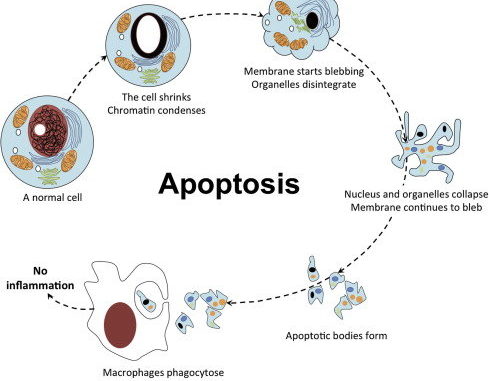
These genes normally program severely damaged cells to die (suicide). Mutations here enhance cell survival, making the cancer cell immortal.
4. Tumor/Host Interaction Genes
Genes that help the tumor evade the immune system or recruit blood vessels (angiogenesis to feed the growing tumor).
Inherited Predisposition to Cancer (The Genetic Syndromes)
This table is heavily tested. Memorize the gene associated with the disease!
| Inherited Syndrome | Mutated Gene(s) | Type / Mechanism |
|---|---|---|
| Autosomal Dominant Cancer Syndromes | ||
| Retinoblastoma (Eye cancer in children) | RB | Tumor Suppressor |
| Li-Fraumeni syndrome (Patient gets multiple cancers at young ages: sarcomas, breast, brain, leukemias) | TP53 | Tumor Suppressor (p53 is known as "The Guardian of the Genome") |
| Melanoma | CDKN2A | Tumor Suppressor |
| Familial Adenomatous Polyposis (FAP) / Colon cancer (100% chance of colon cancer by age 40) | APC | Tumor Suppressor |
| Neurofibromatosis 1 and 2 | NF1, NF2 | Tumor Suppressor |
| Breast and Ovarian tumors | BRCA1, BRCA2 | DNA Repair / Tumor Suppressor |
| Multiple Endocrine Neoplasia (MEN) 1 and 2 | MEN1, RET | Tumor Suppressor (MEN1) / Oncogene (RET) |
| Hereditary Nonpolyposis Colon Cancer (HNPCC / Lynch Syndrome) | MSH2, MLH1, MSH6 | DNA Mismatch Repair defect |
| Nevoid basal cell carcinoma syndrome (Gorlin syndrome) | PTCH1 | Tumor Suppressor |
| Autosomal Recessive Syndromes of Defective DNA Repair | ||
| Xeroderma pigmentosum (Extreme sensitivity to UV light / massive risk of skin cancers) | Diverse genes | Defective Nucleotide Excision Repair (Cannot fix UV damage) |
| Ataxia-telangiectasia | ATM | Defective DNA repair |
| Bloom syndrome | BLM | Defective DNA repair |
| Fanconi anemia | Diverse genes | Defective repair of DNA cross-links |
8. Etiology: Carcinogenic Agents
Carcinogens inflict the genetic damage that lies at the heart of carcinogenesis. There are 3 main classes:
- Chemicals: E.g., Tobacco smoke, asbestos, aflatoxin (from moldy grains, causes liver cancer).
- Radiant energy: UV radiation from the sun (causes pyrimidine dimers in DNA), Ionizing radiation (X-rays, nuclear disasters).
- Microbial products: Viruses and bacteria.
Viral and Microbial Oncogenesis (High Yield)
ONCOGENIC RNA VIRUSES:
- Human T-cell Leukemia Virus type 1 (HTLV-1): Causes adult T-cell leukemia/lymphoma. Endemic in Japan, Caribbean, South America, and Africa. Transmitted via sexual intercourse, blood, breast milk. Leukemia occurs in 3-5% of infected individuals.
- Mechanism: The viral genome encodes a protein called Tax, which stimulates T-cell proliferation, enhances cell survival, and interferes with cell cycle controls.
ONCOGENIC DNA VIRUSES:
Human Papillomavirus (HPV) - Classic Board Topic
- Low-risk (HPV 1, 2, 4, 7, 6, 11): Cause benign squamous papillomas (warts) and genital warts. Very low malignant potential.
- High-risk (HPV 16 & 18): Cause several cancers, particularly Squamous Cell Carcinoma of the cervix and anogenital region.
The Lethal Mechanism: The virus produces viral proteins E6 and E7.
-> E6 binds and destroys human tumor suppressor p53.
-> E7 binds and destroys human tumor suppressor RB.
By destroying both the "brakes" and the "guardian" of the cell, cancer flourishes.
- Epstein-Barr Virus (EBV): Strongly associated with Burkitt lymphoma (a B-cell lymphoma endemic in Africa, often presenting as a jaw mass), Hodgkin lymphoma, and Nasopharyngeal carcinoma.
- Hepatitis B (HBV) and Hepatitis C (HCV) viruses: Chronic infection leads to cirrhosis and is strongly associated with Hepatocellular carcinoma (Liver cancer).
- Kaposi Sarcoma Herpesvirus (Human Herpesvirus-8 [HHV-8]): Causes Kaposi Sarcoma, a vascular tumor heavily seen as dark skin lesions in immunocompromised HIV/AIDS patients.
- Merkel cell polyoma virus: Causes Merkel cell carcinoma (a rare, aggressive skin cancer).
ONCOGENIC BACTERIA:
- Helicobacter pylori (H. pylori): A stomach bacteria implicated in the genesis of both Gastric adenocarcinomas and Gastric lymphomas (MALTomas).
9. Clinical Aspects of Neoplasia
Both malignant and benign tumors cause problems for patients because of:
- Location and impingement: A tiny 1cm benign meningioma growing in the brain can kill a patient by physically compressing vital respiratory centers.
- Functional activity: Tumors of endocrine glands may overproduce hormones (e.g., a benign beta-cell adenoma of the pancreas producing massive insulin, causing fatal hypoglycemia).
- Bleeding and infections: When a tumor expands, it often outgrows its blood supply, necrotizes, and ulcerates through adjacent surfaces (like the bowel wall), causing massive bleeding or peritonitis. Symptoms from rupture or infarction.
- Cachexia: Severe wasting, weight loss, and muscle atrophy seen in terminal cancer patients, caused by inflammatory cytokines (like TNF-alpha) released by the tumor and host macrophages.
Paraneoplastic Syndromes
Symptom complexes that occur in patients with cancer that cannot be readily explained by local/distant spread of the tumor or by the elaboration of hormones indigenous to the tissue of origin. (Basically, the tumor mutates and starts acting like an endocrine gland it has no business being).
| Clinical Syndrome | Major Forms of Neoplasia (Classic exam associations) | Causal Mechanism / Hormone Secreted |
|---|---|---|
| Cushing syndrome (Weight gain, central obesity, moon face, striae) | Small cell carcinoma of lung | Ectopic production of ACTH |
| SIADH (Syndrome of Inappropriate ADH - severe water retention, hyponatremia/low sodium) | Small cell carcinoma of lung | Ectopic Anti-diuretic hormone (ADH) |
| Hypercalcemia (High blood calcium: "Stones, bones, groans, psychiatric overtones") | Squamous cell carcinoma of lung, Breast, Renal CA | PTHrP (Parathyroid hormone-related protein) mimics normal PTH. |
| Polycythemia (Too many red blood cells) | Renal cell carcinoma, Hepatocellular carcinoma | Ectopic Erythropoietin (EPO) |
| Myasthenia (Muscle weakness) | Bronchogenic carcinoma, Thymoma | Immunologic cross-reactivity (antibodies against tumor attack muscles) |
| Acanthosis nigricans (Dark, velvety skin folds on neck/axilla) | Gastric carcinoma, Lung carcinoma | Secretion of epidermal growth factor |
| Hypertrophic osteoarthropathy / Clubbing of fingers | Bronchogenic carcinoma | Unknown mechanism |
| Trousseau phenomenon (Migratory venous thrombosis/recurrent blood clots) | Pancreatic carcinoma, Bronchogenic CA | Tumor products (mucins) that activate clotting cascade |
10. Grading, Staging, and Laboratory Diagnosis
Grading vs. Staging (Know the Difference!)
This is a fundamental concept in oncology. Between the two, STAGING is always the most important prognostic indicator (it tells you how likely the patient is to survive).
- GRADING (Microscopic): Based on the pathologist looking under a microscope at the degree of differentiation of the tumor cells, the number of mitoses, and the architectural features.
-> Grade 1 = Well differentiated / low grade / less aggressive.
-> Grade 4 = Anaplastic / high grade / highly aggressive. - STAGING (Macroscopic/Clinical): Based on the physical footprint of the cancer in the patient's body. It looks at the size of the primary lesion, extent of spread to lymph nodes, and presence of blood-borne metastases.
Uses the TNM System:- T = Tumor: Primary Tumor size and depth of invasion (T1-T4).
- N = Nodes: Regional Lymph Node involvement (N0 = no nodes, N1-N3 = increasing node spread).
- M = Metastasis: Distant blood-borne metastases (M0 = no spread, M1 = spread to distant organs). Note: Any M1 makes it automatically Stage IV cancer, generally incurable.
Laboratory Diagnosis of Cancer
- Morphological Methods: Looking at tissue. Rule: The laboratory evaluation is only as good as the specimen submitted. The specimen must be adequate, representative, and properly preserved (e.g., in formalin).
- Sampling Methods:
- FNA (Fine Needle Aspiration): Sucking out single cells with a tiny needle (e.g., used for thyroid or breast nodules).
- Cytology (Pap Smear): Scraping cells from a surface (e.g., cervix) to look for dysplasia.
- Excision Biopsy: Cutting out the whole lesion.
- Frozen Sections: Rapid freezing and slicing of tissue while the patient is still anesthetized on the operating table. The pathologist tells the surgeon immediately if the margins are clear of cancer, deciding if the surgeon needs to cut out more tissue right then and there.
- Immunohistochemistry (IHC): Using tagged antibodies to identify specific protein markers on cancer cells (e.g., determining if a breast cancer is Estrogen Receptor positive, which dictates if hormonal therapy will work).
- Flow Cytometry: Using lasers to analyze cells suspended in fluid (crucial for diagnosing specific types of liquid cancers like leukemias and lymphomas).
- Tumor Markers: Biochemical indicators found in blood/urine (e.g., PSA for prostate, AFP for liver, CEA for colon). Clinical Note: They lack sensitivity/specificity for definitive initial diagnosis (benign conditions can raise them), but they are excellent for monitoring therapy response or detecting recurrence after surgery.
- Molecular Diagnosis: DNA sequencing and PCR to detect specific mutations (like BRCA or BCR-ABL) to guide modern targeted therapies.
Quick Quiz
Neoplasia & Oncology Quiz
Pathology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Neoplasia & Oncology Quiz
Pathology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.