Endocrine System
Regulation of Hormone Secretion
The endocrine system meticulously controls hormone secretion to ensure physiological homeostasis and appropriate responses to environmental cues. This regulation involves complex feedback loops, neural reflexes, and intrinsic biological rhythms.
I. Characteristics of Hormone Secretion and Action
Before delving into the control mechanisms, it's important to understand some general characteristics of hormone dynamics:
Variable Latency and Duration of Action
- Rapid-Acting Hormones: Some hormones, typically those involved in immediate stress responses or neural signaling, are secreted within seconds after stimulation. Examples include epinephrine and norepinephrine from the adrenal medulla. Their full physiological actions can develop within another few seconds to minutes (e.g., increased heart rate, blood pressure, glucose mobilization).
- Slower-Acting Hormones: Others, like thyroxine (thyroid hormone) or growth hormone, may take hours, days, or even months to exert their full physiological effects. Thyroxine, for instance, influences metabolic rate over an extended period, and its full impact on growth and development is gradual. Growth hormone's effects on skeletal growth accumulate over years.
Extremely Low Serum Concentrations
- Hormones are incredibly potent, meaning they can exert significant effects even at very low concentrations. Their serum concentrations are typically measured in picograms to micrograms per 100 ml of blood.
- Consequently, they are secreted in equally small amounts, often in micrograms or milligrams per day. This underscores the efficiency and sensitivity of the endocrine signaling system, where a tiny amount of chemical messenger can orchestrate widespread physiological changes.
II. Mechanisms of Control of Hormonal Secretion
Hormone secretion is not a constant process but is dynamically regulated by several intricate mechanisms to maintain precise control.
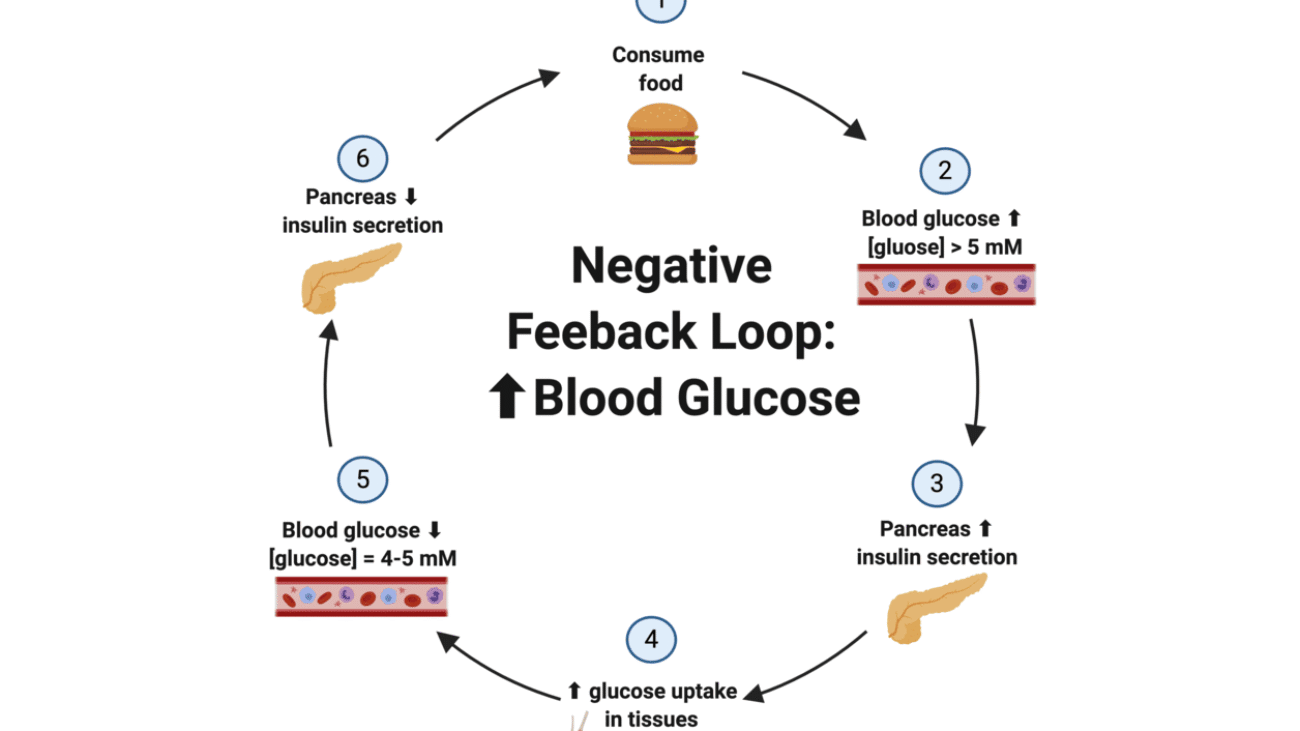
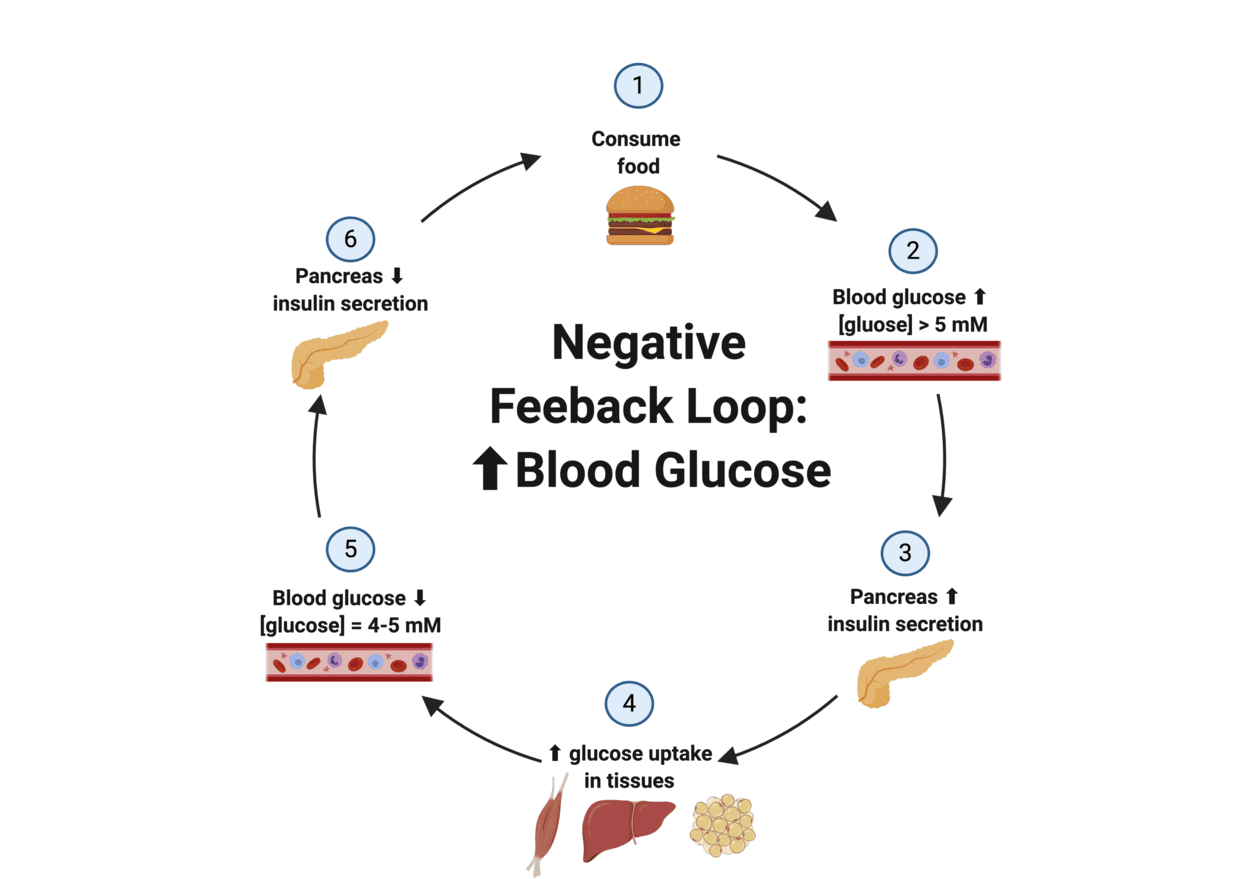
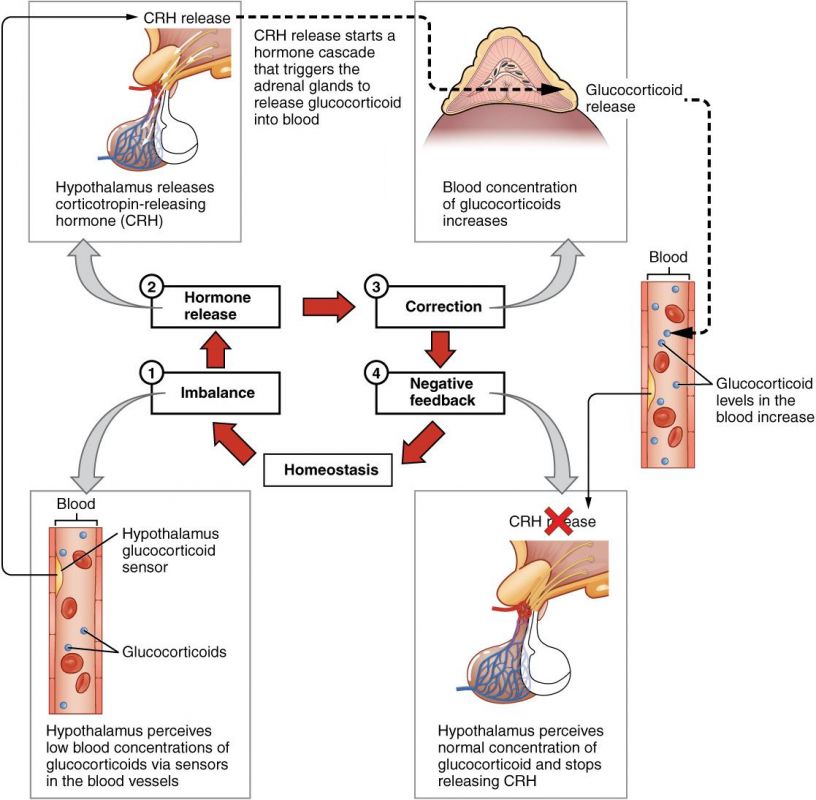
1. Negative-Feedback Control
This is the most common and fundamental regulatory mechanism in the endocrine system, ensuring stability and preventing overactivity.
- Definition: Negative feedback exists when the output of a system counteracts a change in input, thereby maintaining a controlled variable within a narrow, desired range (around a "set point").
- Mechanism:
- A stimulus (e.g., deviation from a set point) triggers an endocrine gland to release a hormone.
- This hormone then acts on target cells to produce a specific physiological response.
- Crucially, the physiological response itself, or the hormone's direct effect on the gland, then inhibits the further release of the initial hormone. This "turns off" the stimulatory signal once the desired level or effect is achieved.
Example: Thyroid Hormone Regulation (HPT Axis)
- Initial Stimulus: If the plasma concentration of free circulating thyroid hormone (T₃ and T₄) falls below a given set point.
- Hypothalamic Response: The hypothalamus senses this drop and secretes Thyrotropin-Releasing Hormone (TRH).
- Anterior Pituitary Response: TRH stimulates the anterior pituitary to secrete Thyroid-Stimulating Hormone (TSH).
- Thyroid Gland Response: TSH stimulates the thyroid gland to increase its secretion of thyroid hormones (T₃ and T₄).
- Negative Feedback Loop: As the levels of T₃ and T₄ in the blood rise back to the set point (or above), they exert negative feedback at two levels:
- On the Anterior Pituitary: T₃ and T₄ directly inhibit the secretion of TSH.
- On the Hypothalamus: T₃ and T₄ also inhibit the secretion of TRH.
- Outcome: This inhibition ensures that once thyroid gland secretion has been "turned on" by TSH, it will not continue unabated. Instead, it will be "turned off" when the appropriate level of free circulating thyroid hormone has been achieved, thus preventing excessive thyroid hormone levels.
Significance:
- Prevents Overactivity: Negative feedback is critical for preventing the overproduction or underproduction of hormones, maintaining stable internal conditions (homeostasis).
- Maintains Set Point: It ensures that hormone levels and their physiological effects remain within a relatively narrow, healthy range.
2. Positive-Feedback Mechanisms
While less common for maintaining ongoing homeostasis, positive feedback plays a vital role in specific, self-amplifying processes.
- Definition: Positive feedback mechanisms control events that are often "out of control" or self-amplifying, and do not require continuous adjustment to a set point; instead, they push the system further in the same direction.
- Nature: Rarely used to maintain homeostasis because they tend to drive physiological variables away from a stable state.
- Mechanism:
- A stimulus triggers an endocrine response.
- This response then amplifies the original stimulus, leading to an even greater response.
- This cycle continues until a specific event or external factor breaks the loop.
Example: Childbirth
- Initial Stimulus: Pressure of the baby's head against the cervix during labor.
- Neural Signal: Nerve impulses from the cervix are sent to the hypothalamus.
- Hormone Release: The hypothalamus stimulates the posterior pituitary to release oxytocin.
- Target Response: Oxytocin stimulates and enhances the contractions of the uterine smooth muscle.
- Positive Feedback Loop: The stronger uterine contractions push the baby further down, increasing the pressure on the cervix. This increased cervical stretching sends more nerve impulses to the hypothalamus, leading to the release of more oxytocin, which in turn causes even stronger contractions.
- Outcome: This intensifies contractions until the baby is delivered from the birth canal. Once the baby is born, the cervical stretching stops, the oxytocin production significantly decreases, and the labor contractions cease, thus breaking the positive feedback loop.
3. Neuroendocrine Reflexes
These mechanisms highlight the intimate connection between the nervous and endocrine systems, often allowing for rapid, precise hormonal responses to external stimuli.
- Definition: Many endocrine control systems involve neuroendocrine reflexes, which include both neural and hormonal components. They typically involve nerve impulses triggering hormone release.
- Mechanism: Neural input to an endocrine gland directly stimulates or inhibits hormone secretion. This often produces a sudden increase in hormone secretion (like "turning up the thermostat setting") in response to a specific stimulus, which is frequently an external one.
Pure Neural Control Example: Adrenal Medulla Secretion
- Mechanism: In some instances, neural input to the endocrine gland is the only factor regulating the secretion of a hormone.
- Example: The secretion of epinephrine and norepinephrine by the adrenal medulla is solely controlled by the sympathetic nervous system. When faced with a perceived threat (fight-or-flight response), sympathetic preganglionic neurons directly stimulate chromaffin cells in the adrenal medulla to release these catecholamines into the bloodstream. There is no hormonal intermediate from a higher endocrine gland.
Combined Feedback and Neuroendocrine Reflexes Example: Cortisol Secretion
- Mechanism: Some endocrine systems incorporate both feedback control (for maintaining a constant basal level of the hormone) and neuroendocrine reflexes (for causing sudden bursts in secretion in response to increased need).
- Example: Cortisol, often called the "stress hormone," is primarily regulated by the HPA axis through negative feedback, which maintains basal cortisol levels. However, during a stress response (e.g., physical injury, psychological stress), sensory input rapidly activates the hypothalamus to release a surge of CRH. This neural-derived signal (via CRH) quickly overrides the negative feedback loop to dramatically increase ACTH and subsequently cortisol secretion. This allows for a rapid and substantial elevation of cortisol to help the body cope with the stressor.
- Significance: This dual control allows for both long-term homeostatic regulation and rapid, adaptive responses to acute challenges.
4. Diurnal (Circadian) Rhythms
Many hormones exhibit predictable fluctuations in their secretion rates over a 24-hour cycle, influenced by the body's internal clock.
- Definition: The secretion rates of many hormones rhythmically fluctuate up and down as a function of time. The most common endocrine rhythm is the diurnal ("day-night") or circadian ("around a day") rhythm.
- Mechanism: These rhythms are controlled by an internal biological clock, the suprachiasmatic nucleus (SCN) in the hypothalamus, which is entrained by external cues, most notably the light-dark cycle (light perceived by the eyes sends signals to the SCN).
- Example: Cortisol Secretion
- Pattern: Cortisol secretion rises during the night, reaches its peak secretion in the morning (typically just before a person wakes up), then gradually falls throughout the day to its lowest level at bedtime.
- Physiological Role: This pattern anticipates the metabolic demands of the active daytime period, ensuring that the body has sufficient glucose and is prepared for activity and potential stressors upon waking.
- Other Examples:
- Growth Hormone: Secreted in pulses, with the largest pulse often occurring shortly after the onset of deep sleep.
- Melatonin: Exhibits a prominent circadian rhythm, with secretion increasing in the evening, peaking during the night, and decreasing in the morning, influencing the sleep-wake cycle.
- Significance: Circadian rhythms allow the endocrine system to anticipate and prepare the body for predictable daily changes in activity, feeding, and sleep, optimizing physiological function over the 24-hour cycle. Disruptions to these rhythms (e.g., jet lag, shift work) can significantly impact health.
Causes of Endocrine Disorders
Endocrine disorders arise when the delicate balance of hormone production, release, and action is disturbed. These disorders fundamentally stem from three main categories: hormone excess (hypersecretion), hormone deficiency (hyposecretion), or decreased target-cell responsiveness to a hormone. Understanding these root causes is crucial for diagnosis and treatment.
I. Hormone Deficiency (Hyposecretion)
Hyposecretion refers to the inadequate production or release of a hormone by an endocrine gland. It can be classified based on the origin of the problem:
A. Primary Hyposecretion
- Definition: Occurs when the abnormality (pathology) directly lies within the hormone-producing gland itself. This gland is failing to secrete sufficient hormone despite appropriate stimulation from its tropic hormone (if applicable).
- Examples:
- Primary Hypothyroidism: The thyroid gland itself is diseased or damaged and cannot produce enough thyroid hormones (T3 and T4), leading to elevated TSH (from the pituitary trying to stimulate it).
- Type 1 Diabetes Mellitus: The pancreatic beta cells (the primary gland) are destroyed and cannot produce insulin.
- Primary Adrenal Insufficiency (Addison's Disease): The adrenal cortex is damaged and cannot produce sufficient cortisol and aldosterone.
B. Secondary Hyposecretion
- Definition: Occurs when the endocrine gland itself is intrinsically normal, but it is secreting too little hormone because of a deficiency of its tropic hormone. The problem originates in a "upstream" gland (e.g., hypothalamus or pituitary) that normally stimulates the target gland.
- Examples:
- Secondary Hypothyroidism: The anterior pituitary fails to produce enough TSH, which then leads to understimulation of a healthy thyroid gland, resulting in reduced thyroid hormone production.
- Secondary Adrenal Insufficiency: The anterior pituitary fails to produce enough ACTH, leading to understimulation of a healthy adrenal cortex.
C. Tertiary Hyposecretion: (Often considered, though not in your provided text, it's a useful distinction)
- Definition: Occurs when the problem is in the hypothalamus, leading to a deficiency of a releasing hormone, which then causes secondary hyposecretion from the pituitary, and subsequently, target gland hyposecretion.
- Example: Insufficient TRH production from the hypothalamus leads to decreased TSH from the pituitary, and ultimately decreased thyroid hormones from the thyroid gland.
D. Causes of Primary Hyposecretion (Detailed)
When the problem resides within the gland itself, the underlying reasons can be diverse:
- Genetic Defects:
- Explanation: Inherited genetic mutations can impair the gland's ability to synthesize a specific hormone, form functional receptors for tropic hormones, or develop correctly.
- Examples:
- Congenital Adrenal Hyperplasia (CAH): A group of genetic disorders affecting enzymes necessary for adrenal steroid synthesis, often leading to cortisol and aldosterone deficiency.
- Genetic defects in insulin production: Some rare forms of diabetes result from genetic errors in beta cell development or insulin synthesis.
- Genetic defects in thyroid hormone synthesis: Can lead to congenital hypothyroidism.
- Dietary Deficiencies:
- Explanation: Lack of essential nutrients required for hormone synthesis.
- Example: Lack of iodine is a classic cause of hypothyroidism. Iodine is an absolute requirement for the synthesis of thyroid hormones (T3 and T4). Chronic iodine deficiency leads to inadequate thyroid hormone production and can cause goiter (enlargement of the thyroid gland) as the pituitary gland tries to compensate by releasing more TSH.
- Chemical or Toxin Exposure:
- Explanation: Certain chemicals or environmental toxins can damage endocrine glands or interfere with hormone synthesis/release.
- Examples:
- Endocrine-Disrupting Chemicals (EDCs): A broad class of chemicals found in pesticides, plastics, and industrial byproducts that can mimic hormones, block their action, or alter their synthesis and metabolism, potentially leading to hyposecretion (or hypersecretion).
- Exposure to certain heavy metals can damage glandular tissue.
- Immunologic (Autoimmune Diseases):
- Explanation: The body's immune system mistakenly attacks and destroys its own hormone-producing cells, leading to a reduction in hormone secretion.
- Examples:
- Autoimmune Thyroiditis (Hashimoto's disease): Autoimmune antibodies attack and progressively destroy the thyroid tissue, leading to hypothyroidism.
- Type 1 Diabetes Mellitus: Autoimmune destruction of the pancreatic beta cells.
- Addison's Disease (Autoimmune Adrenalitis): Autoimmune destruction of the adrenal cortex.
- Other Disease Processes (Non-Autoimmune):
- Explanation: Various pathologies, including infections, infiltrative diseases, or cancerous growth, can destroy or impair endocrine gland tissue.
- Examples:
- Infections: Tuberculosis can destroy adrenal tissue, leading to adrenal insufficiency.
- Infiltrative diseases: Hemochromatosis (iron overload) or amyloidosis can deposit abnormal substances in endocrine glands, impairing their function.
- Cancer: A tumor (even benign) originating within or metastasizing to an endocrine gland can physically destroy healthy hormone-producing cells.
- Iatrogenic Causes:
- Explanation: Physician-induced conditions, often as a consequence of medical treatment.
- Examples:
- Surgical removal of an endocrine gland: For example, a total thyroidectomy (surgical removal of the thyroid gland due to cancer or severe hyperthyroidism) will result in permanent hypothyroidism, requiring lifelong hormone replacement.
- Radiation therapy: Radiation to an area containing an endocrine gland can damage the gland's cells.
- Medications: Certain drugs can suppress hormone synthesis or release (e.g., long-term corticosteroid use can suppress adrenal function, leading to secondary adrenal insufficiency if withdrawn abruptly).
- Idiopathic Causes:
- Explanation: When the cause of the hyposecretion is not known, despite thorough investigation. This is essentially a diagnosis of exclusion.
II. Hormone Excess (Hypersecretion)
Hypersecretion refers to the excessive production or release of a hormone. Like hyposecretion, it can be primary or secondary.
A. Primary Hypersecretion
- Definition: Occurs when the endocrine gland itself is overactive and secretes too much hormone, independent of (or even despite inhibition by) its normal regulatory signals.
- Examples:
- Primary Hyperthyroidism (e.g., Graves' Disease or a hypersecreting thyroid adenoma): The thyroid gland produces excessive thyroid hormones.
- Primary Hyperparathyroidism: A parathyroid adenoma secretes too much PTH.
B. Secondary Hypersecretion
- Definition: Occurs when an endocrine gland is stimulated to produce excessive hormone due to an oversupply of its tropic hormone from an upstream gland.
- Examples:
- Secondary Hyperthyroidism: A pituitary tumor (adenoma) secretes excessive TSH, which overstimulates a healthy thyroid gland.
- Secondary Hyperparathyroidism: Chronic kidney disease leads to low calcium, which then chronically stimulates the parathyroid glands to produce excessive PTH.
C. Tertiary Hypersecretion: (Again, useful to note)
- Definition: Occurs when the hypothalamus produces too much releasing hormone, leading to secondary hypersecretion from the pituitary, and then target gland hypersecretion.
- Example: Excessive CRH production from a hypothalamic tumor leading to increased ACTH and cortisol.
D. Causes of Hypersecretion (Detailed)
1. Tumors (Benign or Malignant):
- Explanation: Endocrine glands are prone to developing tumors (adenomas or carcinomas) that often become autonomous, meaning they ignore normal regulatory input and continuously secrete excess hormone irrespective of the body's needs or negative feedback signals.
- Examples:
- Pituitary Adenomas: Can cause hypersecretion of specific pituitary hormones (e.g., prolactinoma leading to hyperprolactinemia; somatotropinoma leading to excess GH and acromegaly/gigantism; corticotropinoma leading to excess ACTH and Cushing's disease).
- Adrenal Adenomas: Can cause excess cortisol (Cushing's syndrome) or aldosterone (Conn's syndrome).
- Thyroid Adenomas: Can cause primary hyperthyroidism.
- Ectopic Hormone Production: Some non-endocrine tumors (e.g., lung cancer) can secrete hormones (e.g., ACTH, ADH) that they don't normally produce, leading to paraneoplastic syndromes of hormone excess.
2. Immunologic Causes (Autoimmune Stimulation):
- Explanation: In certain autoimmune disorders, the immune system produces abnormal antibodies that mimic the action of a tropic hormone, constantly stimulating the target gland to overproduce its hormones.
- Example: Graves' Disease (the most common cause of hyperthyroidism): The body produces an autoantibody called Thyroid-Stimulating Immunoglobulin (TSI). TSI binds to the TSH receptors on the thyroid gland cells and mimics the action of TSH, but unlike TSH, its activity is not regulated by negative feedback. This leads to excessive stimulation of the thyroid gland, resulting in continuous overproduction and secretion of thyroid hormones.
III. Abnormal Target-Cell Responsiveness
Even if hormone secretion is normal, an endocrine disorder can arise if the target cells fail to respond appropriately to the hormone. This is often due to problems with receptors or post-receptor signaling pathways.
- A. Definition: This occurs when the hormone is present in adequate amounts, but the target cells do not respond effectively to its presence. The problem can be due to:
- Lack of Functional Receptors: The cells may not produce enough receptors, or the receptors produced may be structurally abnormal and unable to bind the hormone effectively.
- Defective Intracellular Signaling Pathways: Even if the hormone binds to its receptor, the subsequent steps within the cell that normally translate the hormone-receptor binding into a cellular response may be impaired.
- B. Examples:
- Type 2 Diabetes Mellitus (Insulin Resistance): In the early stages, the pancreas often produces normal or even elevated levels of insulin. However, the target cells (especially muscle, liver, and adipose tissue) become resistant to insulin's effects. This resistance means that despite adequate insulin, glucose uptake into cells is impaired, leading to high blood glucose. This is a classic example of decreased target-cell responsiveness.
- Testicular Feminization Syndrome (Androgen Insensitivity Syndrome, AIS):
- Explanation: In this condition, individuals are genetically male (XY chromosomes) and have testes that produce normal amounts of testosterone (a masculinizing hormone).
- Problem: However, due to a specific genetic defect, the target cells throughout the body (e.g., those involved in the development of male secondary sexual characteristics and external genitalia) either do not produce functional androgen receptors or produce receptors that are unresponsive to testosterone.
- Outcome: Although adequate testosterone is available, masculinization does not take place. The body develops female external characteristics and a female gender identity, just as if no testosterone were present, because the cells cannot "hear" the testosterone signal.
- Nephrogenic Diabetes Insipidus: The kidneys (target cells for ADH) fail to respond to antidiuretic hormone (ADH), even when ADH levels are normal or high, leading to excessive water excretion.
Transport, Clearance, and Mechanisms of Action of Hormones
For hormones to exert their effects, they must first be transported from their site of synthesis to target cells, then cleared from the body once their job is done. Their action at the cellular level is precise, mediated by specific receptors and intracellular signaling pathways.
I. Transport of Hormones
The method of hormone transport in the blood plasma depends primarily on their chemical nature (water-soluble vs. lipid-soluble).
A. Water-Soluble Hormones:
- Examples: Catecholamines (epinephrine, norepinephrine), peptide hormones (e.g., insulin, growth hormone, TSH, ACTH, ADH, oxytocin, gastrin, secretin).
- Transport Mechanism: These hormones are hydrophilic (water-loving) and can therefore be transported dissolved directly in the plasma. The plasma, being largely water, provides an ideal medium for their circulation.
- Implications:
- Rapid Action: They can quickly diffuse out of capillaries to reach their target cells.
- Short Half-Life: They are typically more susceptible to enzymatic degradation and renal excretion, leading to a shorter half-life in the bloodstream.
B. Lipid-Soluble Hormones (Bound Transport):
- Examples: Steroid hormones (e.g., cortisol, aldosterone, estrogen, testosterone, progesterone) and thyroid hormones (T₃ and T₄).
- Transport Mechanism: These hormones are lipophilic (lipid-loving) and hydrophobic (water-fearing). They are poorly soluble in plasma and would quickly diffuse out or be degraded if not protected. Therefore, they are predominantly transported bound to specific plasma proteins.
- Specific Binding Proteins: For instance, thyroid hormones bind to Thyroxine-Binding Globulin (TBG), Transthyretin, and Albumin. Cortisol binds to Corticosteroid-Binding Globulin (CBG) and Albumin. Sex hormones bind to Sex Hormone-Binding Globulin (SHBG) and Albumin.
- "Free" Hormone: Only a small fraction, typically less than 10% (often 0.03% to 0.1% for thyroid hormones, 1-10% for steroids), exists in the unbound or "free" form. It is this free fraction that is biologically active, capable of diffusing across capillary walls and interacting with target cell receptors. The bound portion acts as a circulating reservoir.
- Implications:
- Protection from Degradation: Binding to plasma proteins protects the hormones from rapid enzymatic degradation and filtration by the kidneys.
- Prolonged Half-Life: This significantly slows their clearance from plasma, giving them a much longer half-life (e.g., hours to days) compared to water-soluble hormones.
- Buffer System: The bound form acts as a buffer, ensuring a relatively stable concentration of free hormone available to tissues, even if secretion rates fluctuate.
- Delivery Mechanism: When free hormone levels drop (e.g., due to tissue uptake), some bound hormone dissociates from the binding proteins to maintain equilibrium, thus replenishing the free, active pool.
II. Clearance of Hormones from Plasma
Hormones, once released and having exerted their effects, must be inactivated and removed from the circulation to prevent prolonged action and allow for dynamic regulation. While the secretion of hormones is tightly controlled, their inactivation and excretion are generally not regulated in the same dynamic, feedback-loop manner.
Hormones are "cleared" from the plasma through several mechanisms:
- Metabolic Destruction by the Tissues:
- Explanation: Enzymes within target cells, liver, kidneys, and other tissues break down hormones into inactive metabolites. This is the primary route for many peptide and protein hormones, which are degraded by peptidases.
- Example: Insulin is rapidly metabolized by insulinase in the liver and kidneys. Catecholamines are broken down by Monoamine Oxidase (MAO) and Catechol-O-Methyltransferase (COMT).
- Binding with the Tissues (Cellular Uptake):
- Explanation: Hormones, particularly those that are lipid-soluble, are taken up by target cells where they bind to their receptors and are then internalized or degraded intracellularly. Even non-target cells can take up and metabolize hormones.
- Example: Thyroid hormones are taken up by cells throughout the body, where they bind to nuclear receptors and are eventually metabolized.
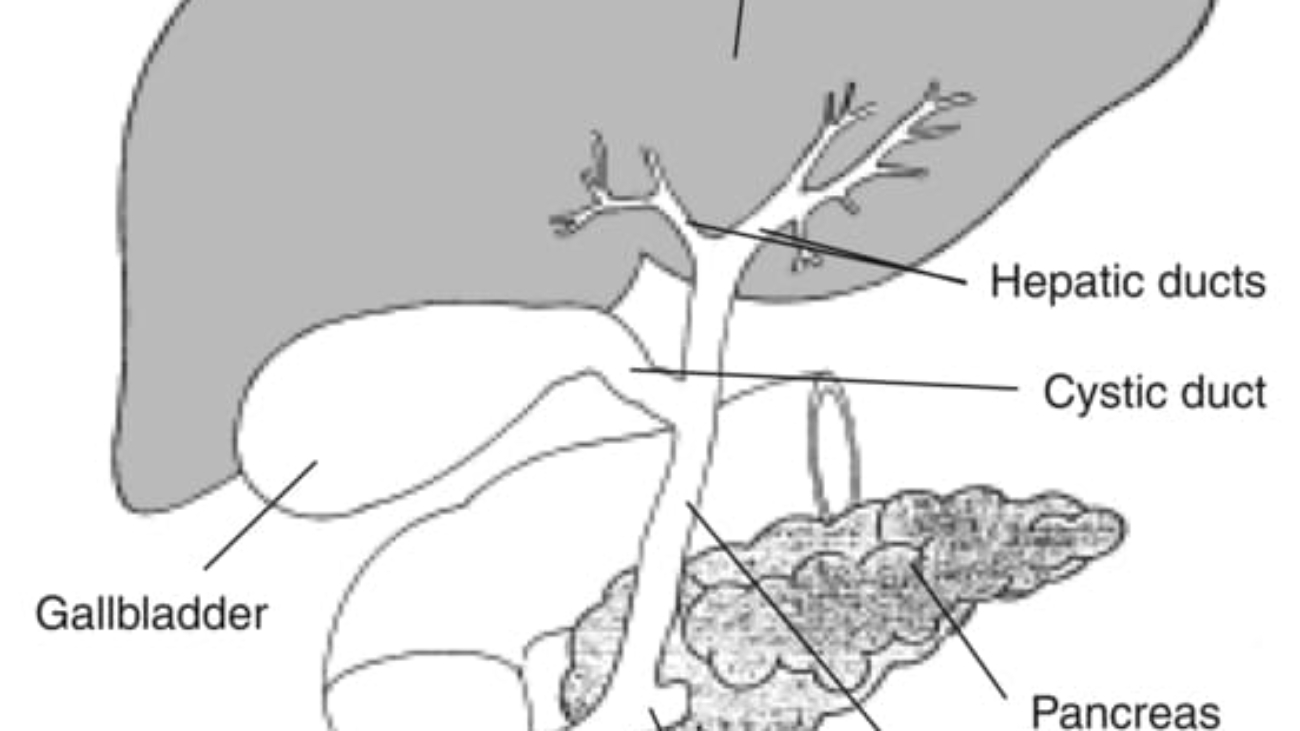
- Excretion by the Liver into the Bile:
- Explanation: Many lipid-soluble hormones (especially steroid hormones) and their metabolites are conjugated in the liver (e.g., with glucuronic acid or sulfate) to make them more water-soluble. These conjugated forms can then be excreted by the liver into the bile, which passes into the intestine and is eventually eliminated in the feces.
- Example: Estrogen and testosterone metabolites are often excreted via the bile.
- Excretion by the Kidneys into the Urine:
- Explanation: Water-soluble hormones (like most peptide hormones) and the water-soluble metabolites of lipid-soluble hormones are readily filtered by the glomeruli in the kidneys and excreted in the urine.
- Example: Many small peptide hormones and the conjugated forms of steroid and thyroid hormones are eliminated renally. Renal failure can significantly prolong the half-life of many hormones.
III. Mechanisms of Action of Hormones & Role of Receptors
Hormones do not act directly on target cells in a generalized way. Instead, their actions are highly specific and mediated by the interaction with specialized proteins called receptors.
A. Hormone Receptors:
- Definition: Hormone receptors are large protein molecules present in target cells. They are the molecular "locks" that specific hormones (the "keys") fit into.
- Specificity: Each receptor is highly specific for a particular hormone or a class of hormones. This means a hormone can act on a target cell only if that target cell possesses the specific receptor for that hormone. Non-target cells, lacking the receptor, will not respond to the hormone.
- Abundance: Target cells typically have a large number of receptors, ranging from 2,000 to 100,000 or more per cell. The number and sensitivity of these receptors can be dynamically regulated (up-regulation or down-regulation) in response to varying hormone concentrations or physiological states.
- Hormone-Receptor Complex: When a hormone binds to its receptor, it forms a hormone-receptor complex. This binding is reversible and initiates a cascade of intracellular events that ultimately lead to the characteristic cellular response.
B. Location of Hormone Receptors (Determined by Hormone Solubility):
The chemical nature of the hormone dictates where its receptor will be located within the target cell.
1. Cell Membrane (Surface Receptors)
- Hormones: Receptors for protein/peptide hormones (e.g., insulin, growth hormone, TSH, ADH) and adrenal medullary hormones (catecholamines) (epinephrine, norepinephrine). These hormones are water-soluble and cannot easily cross the lipid bilayer of the cell membrane.
- Mechanism: The hormone binds to its receptor on the outer surface of the cell membrane. This binding then triggers a conformational change in the receptor, activating intracellular signaling pathways.
2. Cytoplasm (Intracellular Receptors)
- Hormones: Receptors for steroid hormones (e.g., cortisol, aldosterone, estrogen, testosterone, progesterone). These hormones are lipid-soluble and can readily diffuse across the cell membrane into the cytoplasm.
- Mechanism: Once inside the cytoplasm, the steroid hormone binds to its specific receptor. The hormone-receptor complex then typically translocates into the nucleus.
3. Nucleus (Intracellular Receptors)
- Hormones: Receptors for thyroid hormones (T₃ and T₄). Although lipid-soluble and able to cross the cell membrane, thyroid hormone receptors are predominantly located within the nucleus, often bound to DNA even in the absence of hormone.
- Mechanism: Thyroid hormones enter the cell and then the nucleus, where they bind to their receptors.
C. Mechanisms of Action (How the Hormone-Receptor Complex Triggers a Response)
The binding of a hormone to its receptor initiates a series of events that alter cellular function. These mechanisms vary depending on the receptor location.
1. By Altering Permeability of Cell Membrane (Ligand-Gated Ion Channels):
- Explanation: Some hormones (or neurotransmitters) bind to receptors that are directly coupled to ion channels. When the hormone binds, it causes a conformational change that opens or closes the ion channel, altering the permeability of the cell membrane to specific ions (e.g., Na⁺, K⁺, Ca²⁺). This change in ion flow can alter the cell's electrical potential or trigger downstream signaling.
- Example: While more characteristic of neurotransmitter action at synapses (e.g., acetylcholine at the neuromuscular junction opening Na⁺ channels), some hormones can influence ion channels. For instance, insulin can indirectly affect ion transport, and some growth factors can modulate ion channel activity.
2. By Activating Intracellular Enzyme - Action Through Second Messenger Systems (for Membrane-Bound Receptors):
- Explanation: This is the most common mechanism for water-soluble hormones. Since they cannot enter the cell, they act as "first messengers." Their binding to a surface receptor activates an enzyme or a G-protein within the membrane. This activation then leads to the production of intracellular "second messengers" that amplify the signal and relay it to various effector proteins within the cell.
- Key Second Messenger Systems:
- cAMP (Cyclic Adenosine Monophosphate): Hormone binds to receptor → activates G-protein → activates adenylyl cyclase → converts ATP to cAMP → cAMP activates protein kinase A → phosphorylation of various proteins → cellular response. (e.g., Glucagon, ACTH, TSH, LH, FSH, ADH [V2 receptor], Epinephrine [β-adrenergic receptor]).
- cGMP (Cyclic Guanosine Monophosphate): Similar to cAMP, but uses guanylyl cyclase. (e.g., ANP, nitric oxide).
- IP₃ (Inositol Trisphosphate) and DAG (Diacylglycerol): Hormone binds to receptor → activates G-protein → activates phospholipase C → cleaves PIP₂ into IP₃ and DAG. IP₃ mobilizes intracellular Ca²⁺, and DAG activates protein kinase C. (e.g., TRH, GnRH, ADH [V1 receptor], Angiotensin II, Epinephrine [α₁-adrenergic receptor]).
- Ca²⁺-Calmodulin Complex: Increased intracellular Ca²⁺ (from IP₃ or other sources) binds to the protein calmodulin, forming a complex that then activates various enzymes to produce a cellular response. Ca²⁺ itself is often considered a second messenger.
- Signal Amplification: Second messenger systems allow for significant signal amplification: one hormone molecule can lead to the production of many second messenger molecules, which in turn activate many downstream targets, resulting in a large cellular response.
3. By Acting on Genes (for Intracellular Receptors):
- Explanation: This is the primary mechanism for lipid-soluble hormones (steroids and thyroid hormones) that bind to intracellular receptors (cytoplasmic or nuclear). The hormone-receptor complex directly interacts with the cell's genetic material.
- Mechanism:
- The hormone diffuses into the cell and binds to its receptor, often causing the receptor to change shape.
- The hormone-receptor complex then binds to specific DNA sequences called Hormone Response Elements (HREs) located in the promoter regions of target genes.
- This binding can either activate or repress the transcription of specific genes, leading to changes in the production of mRNA.
- The altered mRNA levels then lead to changes in the synthesis of specific proteins (enzymes, structural proteins, regulatory proteins), which ultimately mediate the long-term physiological effects of the hormone.
- Examples: All steroid hormones (cortisol, estrogen, testosterone, aldosterone) and thyroid hormones act primarily by altering gene expression, which explains their relatively slower but more sustained effects on target cells.
https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Hormone Regulation, Disorders & Transport
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Hormone Regulation, Disorders & Transport
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.