Pharmacokinetics: Drug Elimination & Clearance
Pharmacokinetics of Elimination
This module covers the final stages of a drug's journey through the body: Elimination and Excretion. You will learn not only how the body gets rid of drugs, but the mathematical principles (kinetics) that govern this removal. Mastering these concepts is crucial for determining how much of a drug to give (dosing) and how often to give it (dosing intervals) to maintain safe, steady, and therapeutic levels in a patient.
The Fundamentals: Elimination vs. Excretion
While often used interchangeably in casual conversation, in pharmacology, these two terms have different meanings:
Elimination
Elimination is the broad, overarching term. It concerns all the processes involved in the removal of active drugs from the body (and/or plasma) and their kinetic characteristics. If a drug is no longer active in the body, it has been eliminated. The major modes of drug elimination are:
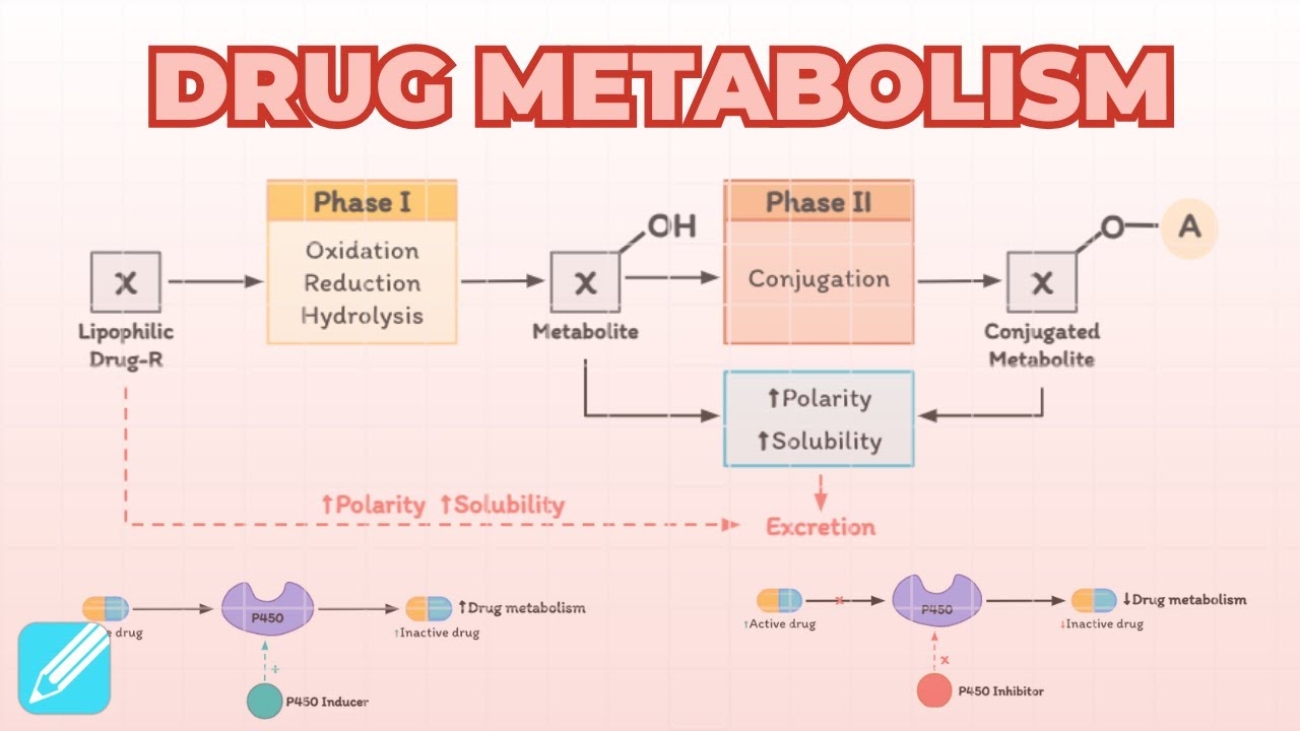
- Biotransformation (Metabolism): The liver chemically alters the active drug into inactive metabolites. Even though the physical atoms of the drug are still in the body, the active drug has been eliminated.
- Excretion: The physical removal of the drug from the body.
Excretion
Excretion is a specific sub-process of elimination. It is the process by which drugs or their metabolites are irreversibly transferred from the internal environment to the external environment (i.e., passed out of the systemically absorbed body).
Drugs and their metabolites can be excreted via several routes:
- Urine: The primary route (Renal Excretion).
- Feces: Biliary excretion via the bile duct into the intestines.
- Exhaled Air: Important for volatile anesthetics and alcohol.
- Saliva and Sweat: Minor routes.
- Breast Milk: Clinically crucial because excreted drugs can be unintentionally passed to a nursing infant.
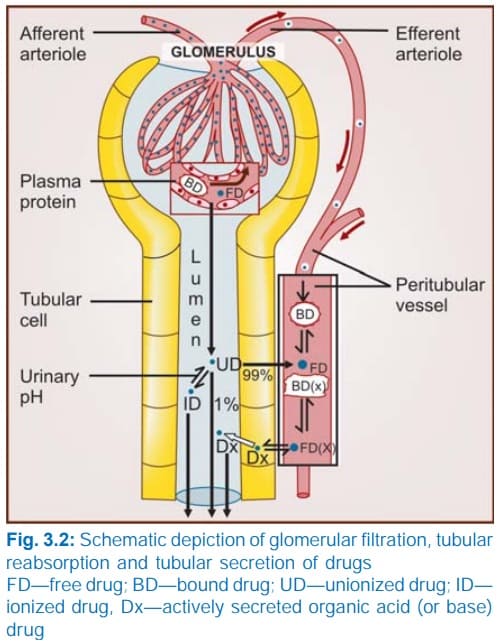
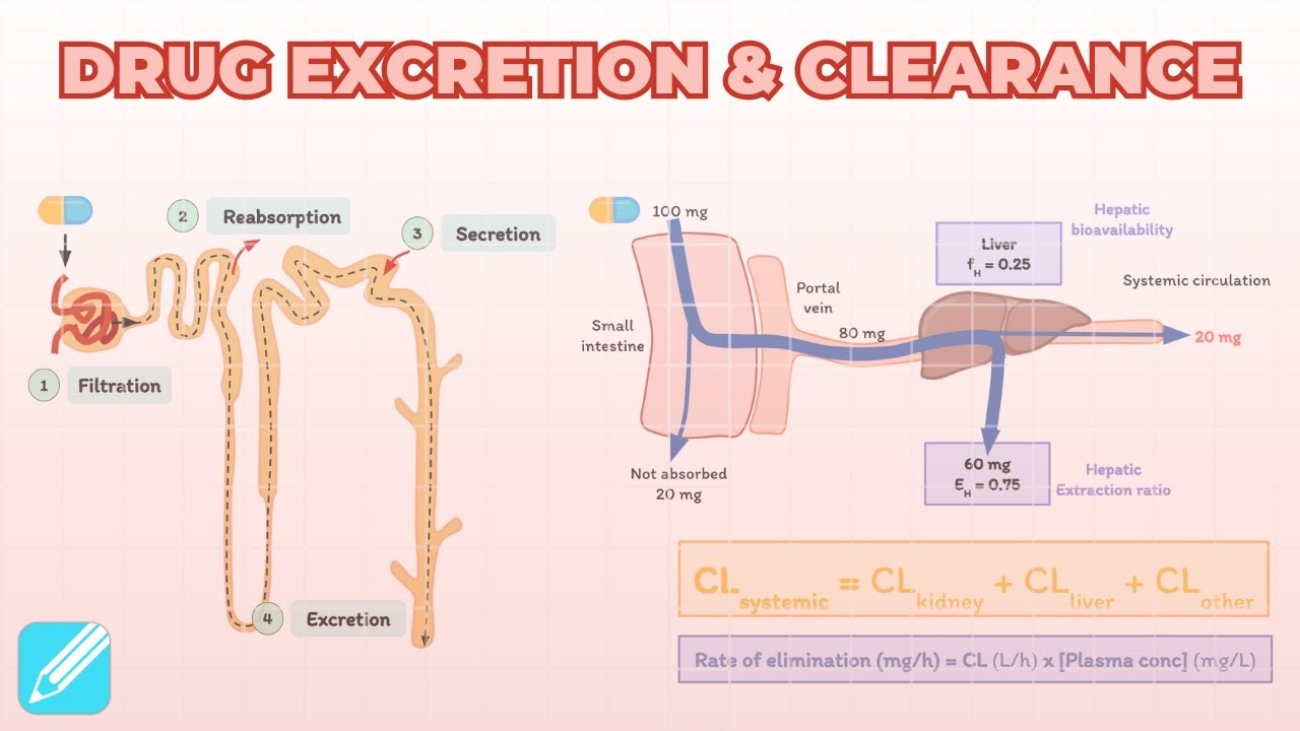
Renal Excretion: The Kidney's Role
The kidneys are the principal organs of excretion. For a drug to be efficiently excreted in the urine (renal excretion), it ideally needs to possess certain physical characteristics:
- Water-soluble (Hydrophilic): So it dissolves in urine.
- Small in molecular size: So it can be filtered.
- Slowly metabolized: If it is rapidly metabolized by the liver, the kidney only excretes the metabolites, not the parent drug.
- Non-volatile: Volatile gases are excreted by the lungs, not the kidneys.
Net Renal Excretion = (Glomerular Filtration + Tubular Secretion) - Tubular Reabsorption
To understand the equation above, we must break down the three distinct processes that occur inside the nephron (the functional unit of the kidney):
A. Glomerular Filtration
Blood enters the kidney's glomerulus under high pressure. Glomerular filtration is a non-selective, unidirectional process. It acts like a simple sieve.
- What gets filtered? Water, small molecules, and unbound (free) drugs.
- What does NOT get filtered? Large proteins (like albumin) and any drug bound to those plasma proteins. Protein-bound drugs are simply too large to pass through the glomerular filter.
- Normal Rate: The normal Glomerular Filtration Rate (GFR) is approximately 120 ml/min.
B. Tubular Reabsorption
As the filtered fluid travels down the renal tubules to become urine, the body realizes it has accidentally filtered out things it wants to keep. It reabsorbs them back into the blood. For drugs, this occurs mostly by passive diffusion.
- Lipid Soluble Drugs: If a drug is highly lipid-soluble, it will easily diffuse across the tubule walls back into the blood. In fact, 99% of the glomerular filtrate (mostly water) is reabsorbed, and lipid-soluble drugs follow this water back into the body.
- Non-Lipid Soluble & Ionized Drugs: These cannot cross the tubule membranes. They remain trapped in the urine and are excreted.
The Role of Urinary pH (Ion Trapping)
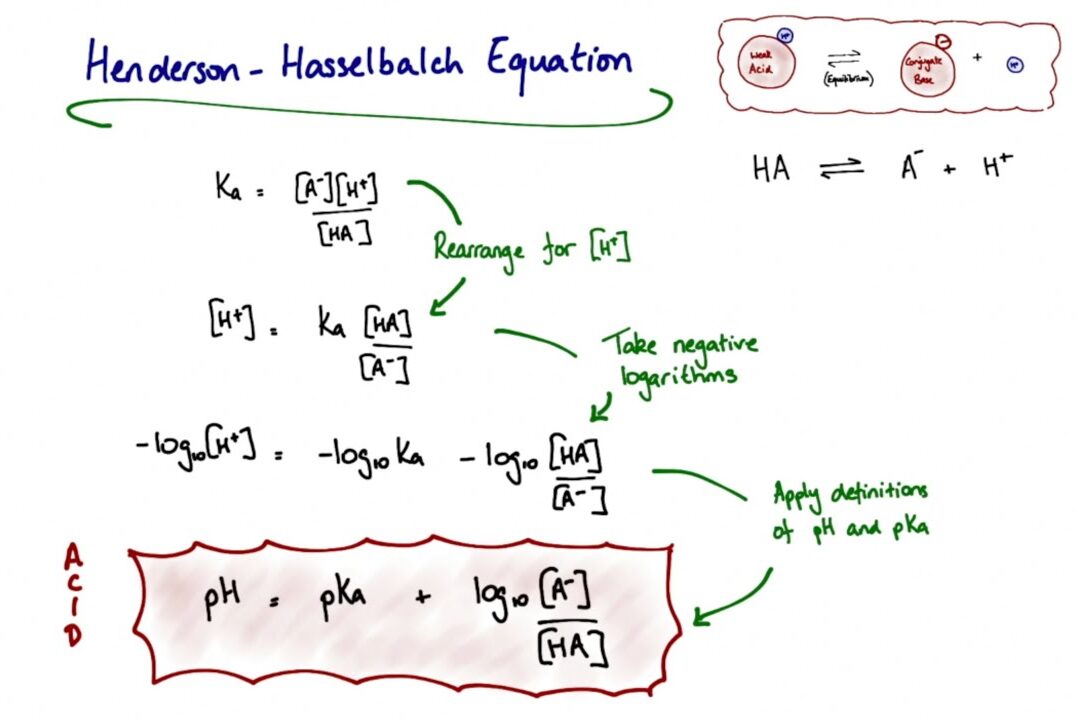
The pH of human urine can vary significantly (from 4.5 to 7.5). Because most drugs are weak acids or weak bases, the pH of the urine determines whether the drug becomes ionized (charged) or unionized (uncharged).
Rule of thumb: Drugs become highly ionized in opposite-pH environments.
- Weak Bases: Ionize more in an acidic medium. If urine is acidic, basic drugs become ionized, cannot be reabsorbed, and are excreted.
- Weak Acids: Ionize more in a basic (alkaline) medium. If urine is alkaline, acidic drugs become ionized, are trapped in the tubule, and are excreted.
Clinical Application: In an aspirin (weak acid) overdose, doctors administer sodium bicarbonate to alkalize the urine. This ionizes the aspirin in the kidney tubules, preventing its reabsorption and rapidly flushing it out of the body.
C. Tubular Secretion
This is the active transfer of organic acids and bases directly from the blood into the renal tubule, bypassing the glomerulus entirely.
- It is a carrier-mediated process that requires cellular energy because it pumps compounds against their concentration gradient.
- OATP (Organic Anion Transporting Polypeptide): Transports acidic drugs (anions). Examples include Penicillin, probenecid, uric acid, salicylates (aspirin), and furosemide.
- OCT (Organic Cation Transporter): Transports basic drugs (cations). Examples include Amiloride, quinine, procainamide, choline, and cimetidine.
- Competitive Inhibition: Because these transporters are limited in number, two drugs can compete for the same pump. For example, Probenecid competes with Penicillin for the OATP pump. Giving them together blocks Penicillin from being secreted, keeping it in the blood longer (historically used to prolong the effects of scarce penicillin).
Elimination Kinetics: The Half-Life (t1/2)
To mathematically model how fast a drug leaves the body, pharmacologists rely heavily on the concept of half-life.
Definition: The Elimination Half-Life (t1/2) is the time required to eliminate 50% of a given amount of drug from the body, or specifically, the time it takes for the plasma concentration of a drug to fall to exactly half of its initial concentration.
- Plasma half-life: Time for plasma levels to drop by 50%.
- Whole body half-life: Time to eliminate 50% of the total drug content from the entire body.
Why is Half-Life Important?
- It tells us the rate of decline of drug concentrations (though it does not necessarily dictate the duration of the biological effect).
- Most drugs are dosed according to their half-life. A drug with a 4-hour half-life might be taken every 6 hours, whereas a drug with a 24-hour half-life is taken once daily.
- It determines the time it takes to reach a Steady State (which we will cover below).
- Drug accumulation in the body is directly related to the drug's half-life and the dosing intervals.
Factors Affecting Half-Life:
Half-life is not always a static number; it changes based on physiological conditions. Major factors include:
- Age: Elderly patients often have slower metabolisms and reduced kidney function, significantly extending a drug's half-life.
- Renal Excretion: Kidney disease severely prolongs the half-life of renally excreted drugs.
- Liver Metabolism: Liver disease (cirrhosis) prolongs the half-life of hepatically metabolized drugs.
- Protein Binding: Highly bound drugs stay in the blood longer, extending their half-life.
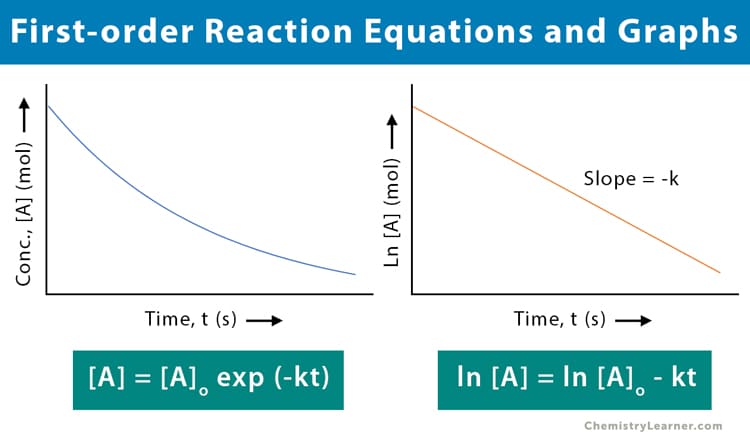
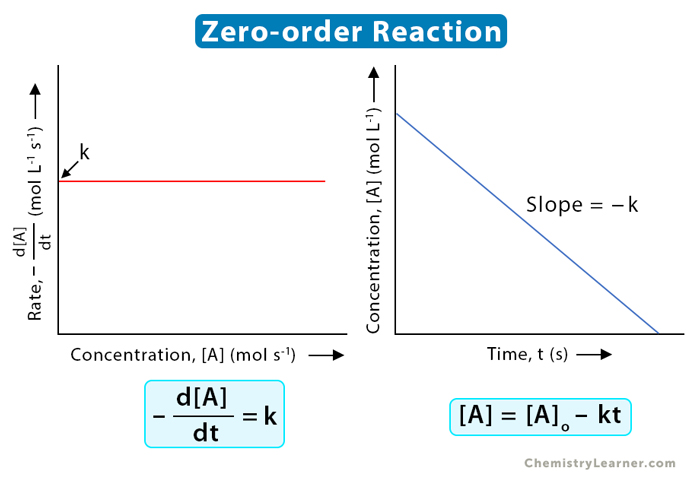
First-Order vs. Zero-Order Kinetics
How a drug's concentration declines over time falls into two distinct mathematical categories.
| Feature | First-Order Kinetics (Linear Kinetics) | Zero-Order Kinetics (Saturation Kinetics) |
|---|---|---|
| Core Principle | A constant FRACTION (percentage) of the drug is eliminated per unit of time (e.g., 50% every hour). | A constant AMOUNT of the drug is eliminated per unit of time (e.g., exactly 10 mg every hour). |
| Dependence on Concentration | Rate of elimination is directly proportional to drug concentration. (More drug in the body = faster elimination rate). | Rate of elimination is independent of plasma concentration. The elimination mechanisms (enzymes) are saturated and working at max capacity. |
| Half-Life (t1/2) | Constant. It always takes the same amount of time to cut the concentration in half. | No fixed half-life. It is highly variable and depends entirely on how much drug is currently in the body. |
| Graphical Plot | Plotting Concentration vs. Time yields an exponential (curved) graph. Plotting Log[Drug] vs. Time yields a straight, linear line. | Plotting Concentration vs. Time yields a straight, linear line descending directly downwards. |
| Clinical Examples | Applies to the vast majority of drugs within their normal therapeutic dosage range. | Applies to drugs that easily saturate liver enzymes: Ethanol (Alcohol), Phenytoin (seizure drug), and Aspirin/Salicylates (at high/toxic doses). |
Imagine emptying a swimming pool:
First-Order: You have a magic drain that always empties exactly half of whatever water is left in the pool every hour. Hour 1: 1000L to 500L (drained 500L). Hour 2: 500L to 250L (drained 250L). The amount drained changes, but the fraction (50%) is constant.
Zero-Order: You are using a bucket that can only hold 10 Liters, and you can only throw out one bucket per minute. It doesn't matter if the pool has 10,000 Liters or 50 Liters; your rate is maxed out at exactly 10 Liters per minute. The amount is constant.
The Concept of Clearance (Cl)
Clearance is a vital concept, yet frequently misunderstood. It does not refer to an amount of drug.
Definition: Clearance is the theoretical VOLUME of plasma from which a drug is completely removed (freed) in a unit of time. It provides an estimate of the functional capacity of the organs of elimination. It is expressed in volume/time (e.g., ml/min or Liters/hour).
Clearance (Cl) = Elimination Rate (mg/hr) / Plasma Drug Concentration (mg/L)
In First-Order kinetics, Clearance is a constant proportionality factor used to determine the rate of elimination.
Types of Clearance
- Total Body Clearance: The plasma volume cleared of the drug per unit time via all elimination mechanisms combined (liver metabolism + kidney excretion + sweat, etc.).
- Renal Clearance: Specifically, the volume of plasma cleared of the non-metabolized (unchanged) drug strictly via excretion by the kidneys per minute.
The Mathematical Relationship: Clearance, Volume of Distribution, and Half-Life
There is a holy trinity of pharmacokinetic variables that dictate a drug's behavior:
t1/2 = (0.693 × Vd) / Cl
How to interpret this:
- Elimination half-life is inversely proportional to clearance. If your kidneys are highly efficient and clear the drug rapidly (high Cl), the drug's half-life will be very short.
- Elimination half-life is directly proportional to Volume of Distribution (Vd). If a drug has a massive Vd, it means it is hiding deep inside fat or tissue cells, far away from the blood plasma. Because the kidneys and liver can only clear drugs that are in the blood, a high Vd protects the drug from elimination, resulting in a very long half-life.
Factors Affecting Renal Clearance
- Glomerular Filtration Rate (GFR): High GFR = higher clearance.
- Plasma Protein Binding: Only the free fraction of a drug can be filtered. Protein-bound drug is not cleared. Therefore,
Cl = Free Fraction × GFR. - Tubular Reabsorption: Reabsorption pulls drug back into the blood, decreasing clearance.
- Tubular Secretion: Secretion pumps extra drug into the urine, dramatically increasing clearance.
Interpreting Specific Renal Clearance Values
By measuring a drug's renal clearance against known standards, scientists can deduce exactly how the kidney is handling it:
| Renal Clearance Value | Mechanism in the Kidney | Classic Examples |
|---|---|---|
| 0 ml/min (Lowest) | Drug is filtered, but then 100% is actively reabsorbed back into the body. | Glucose (In a healthy person, you shouldn't pee out sugar). |
| < 130 ml/min | Drug is filtered, and partially reabsorbed passively. | Most highly lipophilic drugs. |
| Exactly 130 ml/min (Equal to GFR) | Drug is filtered ONLY. It is neither reabsorbed nor secreted. (This makes it the perfect marker to measure a patient's GFR). | Creatinine, Inulin. |
| > 130 ml/min | Drug is filtered AND actively secreted into the tubule by pumps. | Polar/ionic drugs (e.g., Penicillin). |
| ~ 650 ml/min (Highest) | Clearance is equal to the total Renal Plasma Flow Rate. Almost all drug arriving at the kidney is ripped from the blood and secreted. | PAH (Para-aminohippurate). |
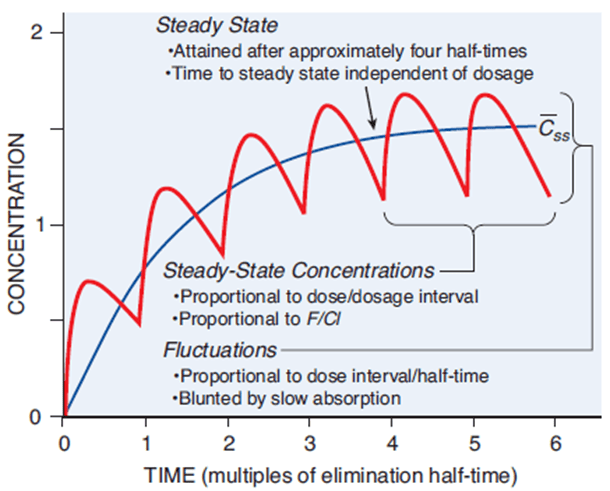
The Steady State (Css) and Drug Accumulation
Successful drug therapy for chronic illnesses usually requires keeping the drug concentration at a stable, continuous, effective level in the blood. This plateau is called the Steady State (Css).
Imagine a sink with the tap turned on (Rate of Administration/Rate In) and the drain left open (Clearance/Rate Out). When you first turn on the tap, water accumulates in the sink because the water entering is faster than the water draining. However, as the water level rises, the weight (pressure) of the water pushes it down the drain faster. Eventually, the rate of water entering exactly matches the rate of water leaving. The water level stops rising and stays perfectly flat. This is the Steady State.
Mathematical Definition: Steady State is reached when Rate In = Rate Out.
The Plateau Principle
How long does it take for a patient taking regular pills to reach this flat steady state? This is governed by the Plateau Principle:
- The time to reach steady state is dependent ONLY on the elimination half-life of the drug.
- It is completely independent of the dose size or how frequently the doses are administered. Taking double the dose doesn't get you to steady state faster; it just results in a higher final plateau.
- As a mathematical rule of thumb, it takes approximately 4 to 5 half-lives to reach a clinical steady state.
| Number of Half-Lives Elapsed | Percentage of Steady State Reached |
|---|---|
| 1 Half-Life | 50% |
| 2 Half-Lives | 75% (50 + 25) |
| 3 Half-Lives | 87.5% (75 + 12.5) |
| 4 to 5 Half-Lives | ~ 95% (Clinical Steady State) |
| > 7 Half-Lives | 100% (Mathematical Steady State) |
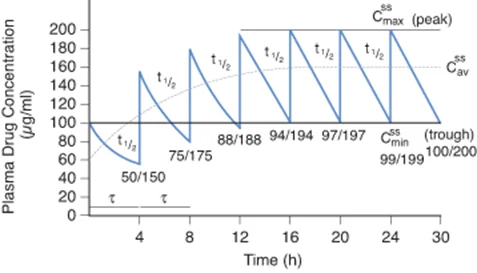
Plasma Level Fluctuations
The way a drug is administered determines how smooth that steady state is:
- Continuous IV Infusion: Provides a perfectly flat, smooth steady state line.
- Intermittent Dosing (e.g., Oral pills every 8 hours): Creates oscillations. The plasma level spikes after taking the pill (Peak/C-max) and drops right before the next pill (Trough/C-min). The average between these peaks and troughs is the steady-state concentration.
To minimize severe fluctuations (which could cause toxicity at the peak, or loss of effect at the trough), doctors prefer to divide the total daily dose into smaller, more frequent doses, or use sustained-release drug formulations. However, patient compliance drops if they have to take pills too frequently.
Clinical Significance: Dosing Equations
Using these pharmacokinetic principles, doctors can precisely calculate how to dose a patient.
1. Maintenance Dose (MD)
Once steady state is reached, you only need to administer enough drug to replace what the body cleared. Since Rate In = Rate Out, the Maintenance Dose rate must equal the Elimination Rate.
MD = (Clearance × Target Css × τ) / F
Where τ (tau) = dosing interval (e.g., every 8 hours), and F = Bioavailability fraction (For IV drugs, F = 1).
2. Infusion Rate (k0)
If giving a continuous IV drip, you want to set the machine's rate to exactly match clearance.
If the rate of infusion is doubled, the resulting steady-state plasma level will exactly double (linear kinetics).
k0 = Clearance × Target Css
3. Loading Dose (LD)
The Problem: For drugs with very long half-lives (e.g., Digitoxin, Methadone), waiting 4 to 5 half-lives to reach steady state could mean waiting weeks for the drug to start working effectively. In emergencies, this is unacceptable.
The Solution: Give a large, one-time Loading Dose to instantly fill the body's Volume of Distribution (Vd) up to the target steady-state concentration.
LD = (Vd × Target Css) / F
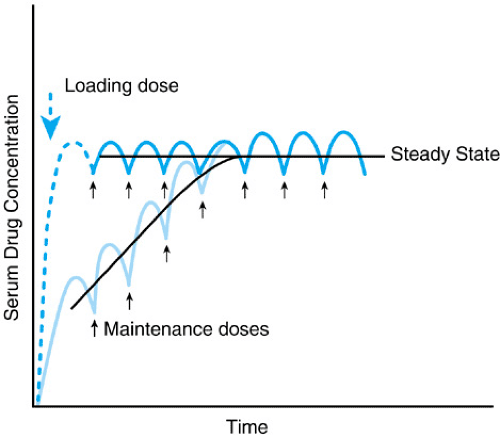
If a drug is given exactly every half-life, the mathematically calculated Loading Dose will be exactly twice the Maintenance Dose.
Change in Elimination Characteristics During Therapy
Steady state calculations assume the body's physiology remains constant. In the real world, patients change:
- Acceleration of Elimination (Enzyme Induction): Some drugs force the liver to produce more metabolic enzymes. This clears drugs faster. Consequently, the steady-state plasma level will steadily decline, and the drug effect may diminish or disappear completely unless the dose is increased.
- Changes in Urinary pH: Diet or concurrent medications can alter urine pH, increasing the excretion of certain drugs (as discussed in Ion Trapping).
- Inhibition or Impairment of Elimination: If a patient develops progressive renal insufficiency (kidney failure) or liver disease, the body's clearance plummets. If the doctor does not adjust the dose, the steady-state level will aggressively rise, potentially entering the toxic concentration range.
Detailed Pharmacokinetics: Clearance & Dosing Mathematics
An exhaustive, elaborated continuation focusing strictly on Clearance, Mathematical Relationships, and Clinical Dosing (Excluding basic Steady State definitions).
1. The Fundamental Concept of Clearance (Cl)
To safely dose a patient, a physician must know exactly how efficiently the patient's body removes the drug. This is quantified by Clearance (Cl).
Definition & Simplification
Clearance of a drug is the theoretical VOLUME of plasma from which a drug is completely removed (freed) in a unit of time.
Simplification: Do not think of clearance as an "amount" of drug (like 10 mg/hour). Think of it strictly as a volume of blood being purified. If a drug's clearance is 50 ml/min, it means the kidneys/liver act like a filter that completely scrubs all drug molecules out of 50 milliliters of blood every single minute.
- It is expressed in units of volume per time, typically ml/min or Liters/hour.
- For any drug following first-order kinetics, clearance is a constant for any given plasma drug concentration.
- It serves as the ultimate estimate of the function of the organs of elimination (kidneys, liver, etc.) and the rate of removal of the drug from the body.
Clearance (Cl) = Rate of Elimination (mg/hr) / Plasma Drug Concentration (mg/L)
Mathematically, Clearance is the proportionality factor used to determine the exact rate of elimination. If you know the clearance and the concentration in the blood, you can calculate exactly how many milligrams are leaving the body per hour.
Conditions for Clearance
- First-Order Kinetics: Cl remains absolutely constant.
- Relation to GFR: Cl is exactly equal to the Glomerular Filtration Rate (GFR) only when there is no tubular reabsorption, no active tubular secretion, and no plasma protein binding.
- Protein Binding Limitation: Protein-bound drug is not cleared by glomerular filtration because the proteins are too large to pass through the kidney's filter.
Therefore, the true clearance of a filtered drug is dictated by its free fraction:
Cl = Free Fraction × GFR
Types of Clearance & Affecting Factors
A. Total Body Clearance
This is the big picture. It is the total plasma volume cleared of the drug per unit of time via the elimination of the drug from all biotransformation (liver) and excretion (kidneys, lungs, bile) mechanisms combined in the entire body.
B. Renal Clearance
This is organ-specific. It is described strictly as the rate of the excretion of a drug specifically from the kidneys. In other words, it is the volume of plasma cleared from the non-metabolized (unchanged) drug via excretion by the kidneys per minute.
Four Important Factors Affecting Renal Clearance
Because renal clearance is a physical process happening in the kidney tubules, it is directly influenced by four biological factors:
- Plasma protein binding of the drug: High binding drastically reduces clearance because the drug cannot be filtered at the glomerulus.
- Tubular reabsorption ratio of the drug: High reabsorption (drug moving from urine back into blood) reduces net clearance.
- Tubular secretion ratio of the drug: High active secretion (pumping drug directly from blood into urine) drastically increases clearance.
- Glomerular filtration ratio (GFR) of the drug: A higher GFR (healthy kidneys) means more plasma is filtered, increasing clearance.
The Mathematical Relationship: Clearance, Elimination & Half-Life
The speed at which a drug leaves the body is a delicate balance between how efficiently the body clears it (Cl) and how deeply the drug is hiding in the body's tissues (Volume of Distribution, Vd).
- Core Principle: The faster the clearance, the better (more rapid) the elimination.
- Clearance (Cl) is directly proportional to the elimination rate constant (k). Written as:
Cl ∝ k.
Deriving the Ultimate Half-Life Equation
Let's trace the logic step-by-step from the lecture slides to see how we calculate half-life (T1/2):
- We know the unit for Clearance (Cl) is Volume / Time.
- Therefore, we can express it as:
Cl = Vd × (1/t)(where Vd is Volume of Distribution). - The elimination rate constant (k) represents 1/t. Therefore:
Cl = Vd × k. - Rearranging to solve for k, we get:
k = Cl / Vd. - The formula for half-life in first-order kinetics is:
T1/2 = 0.693 / k(often rounded to 0.7 for simplicity). - By substituting our k value into the half-life formula, we get the master equation:
T1/2 = (0.693 × Vd) / Clearance (Cl)
Important Points to Remember About This Half-Life Equation:
- Inverse Proportion to Clearance: Elimination half-life is inversely (negatively) proportional to clearance. If a patient has highly efficient kidneys (large Cl), the denominator is large, making the half-life (T1/2) very short.
- Direct Proportion to Volume of Distribution (Vd): Elimination half-life is directly proportional to the volume of distribution. Why? The higher the duration of stay of that drug in the body, the more it becomes distributed to peripheral tissues (hence, a higher volume of distribution). Because the drug is hiding in the fat and tissues, it is not in the blood, meaning the kidneys cannot clear it. Therefore, a massive Vd results in a massive (long) half-life.
- Metabolism: The plasma concentration of the drug is directly proportional to the rate of metabolism.
- Clinical Importance: Half-life and drug clearance are practically used to predict how long it takes for a periodic dosing regimen to achieve steady-state concentrations. Half-life establishes how often the drug must be administered (the dosing interval) to prevent dangerous drug accumulation, especially for drugs with a very long t1/2.
Calculating Renal Elimination & The Role of Inulin
The total rate of renal elimination can be summarized as:
Rate of Elimination = Glomerular Filtration Rate (GFR) + Active Secretion - Reabsorption (active or passive)
Remember, filtration is a non-saturable linear function. Both ionized and non-ionized forms of drugs are filtered freely, but protein-bound drug molecules are absolutely not.
The Marker for GFR: Inulin
To measure a patient's exact GFR, doctors use a substance called Inulin (not to be confused with insulin). Inulin clearance is the perfect estimate for GFR because it possesses unique properties: it is 100% filtered, and it is strictly NOT reabsorbed AND NOT secreted. Whatever amount is filtered is exactly the amount that ends up in the urine.
A normal, healthy GFR measured by inulin clearance is close to 120 ml/min.
Renal Clearance (CLR) = (V × CU) / (t × CP)
- V = Collected urine volume (amount of urine the patient produced).
- t = Duration to collect the urine (time).
- CP = Plasma concentration of the drug.
- CU = Urine concentration of the drug.
Relationship Between Renal Clearance Values & Mechanism
By calculating the Renal Clearance of an unknown drug and comparing it to the standard GFR (120-130 ml/min), pharmacologists can instantly deduce exactly how the kidney is handling that specific drug.
| Renal Clearance Value (ml/min) | Renal Clearance Ratio (Drug Cl / GFR) | Mechanism of Renal Clearance inside the Kidney | Classic Examples |
|---|---|---|---|
| 0 (Least Value) | 0 | Drug is filtered at the glomerulus, but then 100% is reabsorbed completely back into the bloodstream. | Glucose. (Healthy kidneys reabsorb all sugar; none should appear in urine). |
| < 130 | Above 0, Below 1 | Drug is filtered, and then partially reabsorbed. | Lipophilic drugs. (Fat-soluble drugs passively diffuse back into the blood). |
| Exactly 130 (Equal to GFR) | 1 | Drug is filtered only. Zero reabsorption, zero active secretion. | Creatinine, Inulin. |
| > 130 | > 1 | Drug is filtered, AND it is actively secreted into the urine via transport pumps. | Polar, ionic drugs. (e.g., Penicillin is actively pumped out). |
| ~ 650 (Highest Value) | 5 | Clearance is equal to the total Renal Plasma Flow Rate. 100% of the drug that arrives at the kidney is immediately ripped from the blood and dumped into the urine. | Iodopyracet, PAH (Para-aminohippurate). |
Changes in Elimination Characteristics During Therapy
When a patient takes a drug regularly, the goal is to accumulate the drug to a desired, steady plasma level. However, a clinician must remember that conditions for biotransformation (liver) and excretion (kidney) do not necessarily remain constant over time.
- Acceleration of Elimination: Elimination may suddenly be hastened due to enzyme induction (the liver produces more metabolic enzymes due to repeated exposure to the drug) or due to a change in urinary pH (which causes ion trapping in the urine).
- Consequence: The steady-state plasma level declines to a new, lower value corresponding to the faster rate of elimination. The drug effect may dangerously diminish or disappear entirely.
- Inhibition / Impairment of Elimination: Conversely, elimination can be impaired (e.g., a patient developing progressive renal insufficiency/kidney failure, or taking a second drug that inhibits liver enzymes).
- Consequence: Because the drug cannot leave the body, the mean plasma level of renally eliminated drugs rapidly rises and may enter a toxic concentration range, leading to overdose symptoms despite taking a "normal" dose.
Clinical Dosing: Rate of Infusion (ko) and Loading Dose (LD)
Rate of Infusion (ko)
When giving a drug via an IV drip, the rate of infusion directly determines the final plasma level at steady state. Because this operates on linear (first-order) kinetics:
- If the rate of infusion is doubled, then the plasma level of the drug at steady state is exactly doubled.
- A similar relationship exists for oral administration: doubling the oral dose will double the average plasma levels of a drug.
- Plotting dose against plasma concentration yields a perfect straight line.
The Loading Dose (LD)
We know it takes 4 to 5 half-lives to achieve a steady state. For drugs that are eliminated very slowly (e.g., phenprocoumon, digitoxin, methadone), the optimal, effective plasma level would only be attained after a very long period (sometimes weeks).
To solve this, doctors use a Loading Dose. This is an initial, abnormally high dose given to rapidly bypass the waiting period and instantly achieve effective blood levels.
- Loading doses are often a one-time only administration.
- They are mathematically estimated to put into the body the exact total amount of drug that should be there once a steady state is naturally reached.
- Example Rule: If doses are to be administered at an interval exactly equal to the half-life of the drug, then the loading dose is exactly twice the amount of the dose used for maintenance (assuming normal clearance and identical bioavailability).
LD = (Volume of Distribution (Vd) × Target Plasma Concentration (Css)) / Bioavailability (F)
Notice that the Loading Dose equation relies on the Volume of Distribution (Vd) to know how much fluid needs to be "filled up" with the drug.
Maintenance Dose (MD) & Master Equation Summary
Once the Loading Dose has forced the patient's blood up to the target concentration, the doctor switches to a Maintenance Dose. The goal of the maintenance dose is simply to replace exactly what the body is eliminating.
Deriving the Maintenance Dose:
- At steady state, the system is perfectly balanced: Rate In = Rate Out.
- The "Rate Out" is determined by Clearance. Therefore, Rate Out =
Css × Cl. - The "Rate In" is your dosing. If you give a Maintenance Dose (MD) every dosing interval (τ), the Rate In =
MD / τ. - Setting them equal:
MD / τ = Css × Cl. - Solving for MD gives the final formula:
MD = Css × Cl × τ.
MD = (Clearance (Cl) × Target Plasma Concentration (Css) × Dosing Interval (τ)) / Bioavailability (F)
Notice that the Maintenance Dose relies strictly on Clearance (Cl), because you only need to replace what the body clears.
Comprehensive Master List of Pharmacokinetic Equations
The following relationships are critical for clinical calculations:
Legend of Variables
| C0 = Concentration at time zero | Cl = Clearance |
| Cp = Concentration in plasma | Css = Steady state concentration |
| D = Dose | F = Bioavailability (Fraction reaching systemic circulation) |
| ko = Infusion rate | LD = Loading dose |
| MD = Maintenance dose | τ (tau) = Dosing interval (e.g., every 8 hours) |
| Vd = Volume of distribution | t1/2 = Half-life |
1. Single-Dose Equations
- Volume of Distribution:
Vd = D / C0 - Half-Life:
t1/2 = (0.7 × Vd) / Cl
2. Multiple Doses or Infusion Rate Equations
- Infusion Rate (ko):
ko = Cl × Css - Loading Dose (LD):
LD = Vd × Css - Maintenance Dose (MD):
MD = Cl × Css × τ
*Note: For oral dosing, always divide the final LD or MD calculation by the bioavailability fraction (F) to account for drug lost to first-pass metabolism or poor absorption.
Drug Elimination & Clearance Quiz
Pharmacology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Drug Elimination & Clearance Quiz
Pharmacology
Preparing questions...
Exam Completed!
See your performance breakdown below.