Fetal Membranes, Placenta, Cord and Circulation: Safety and Feeding
Fetal Membranes, Placenta, Cord and Circulation
The fetal membranes and the placenta are temporary, yet essential, organs that develop alongside the embryo and fetus. They provide a complete life-support system, handling protection, nourishment, gas exchange, waste removal, and hormonal regulation critical for successful intrauterine development. They are expelled from the body after birth.
Formation of Embryonic Cavities and Membranes
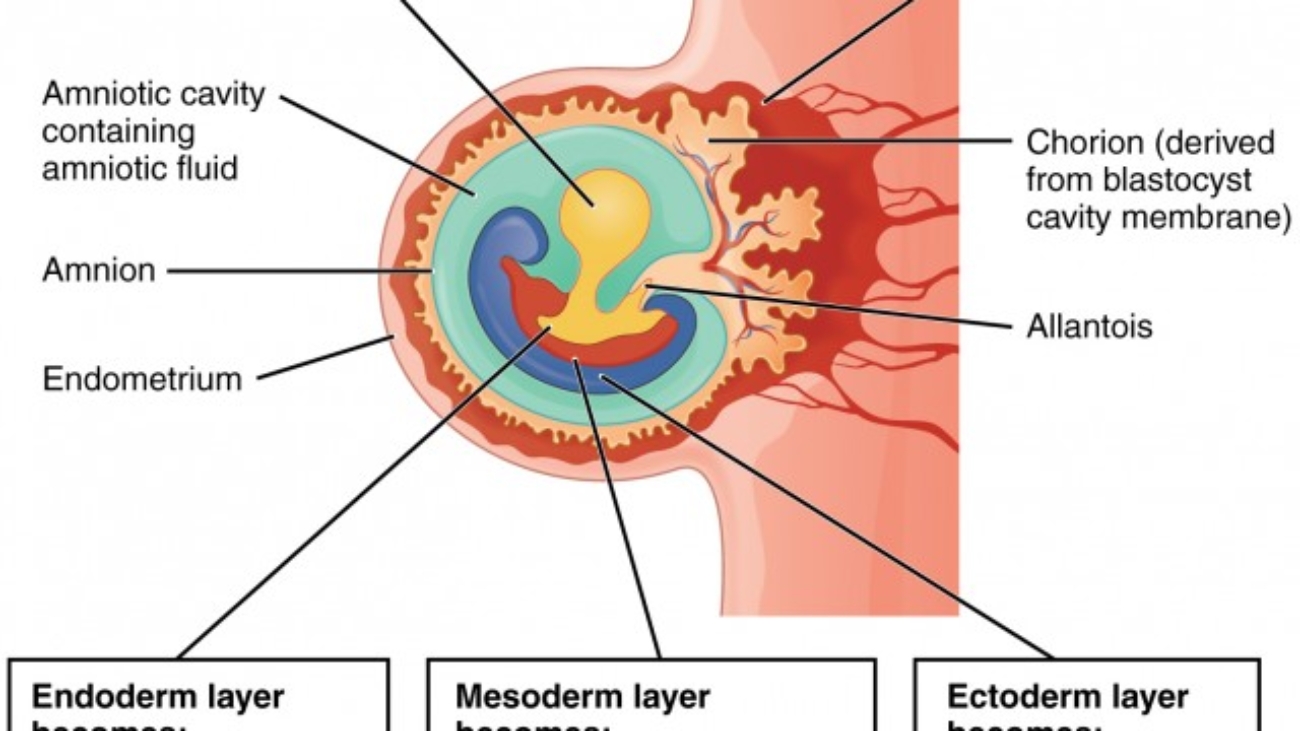
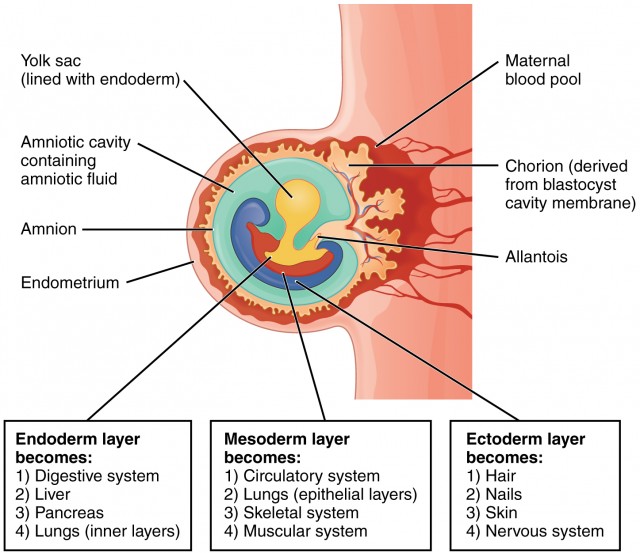
The period of early embryonic development (roughly Day 8 to Day 12-14 post-fertilization) is characterized by the rapid formation of several extraembryonic structures, which are vital for the embryo's survival and subsequent development. These include the amniotic cavity, primary and secondary yolk sacs, and the chorionic cavity, along with their associated membranes.
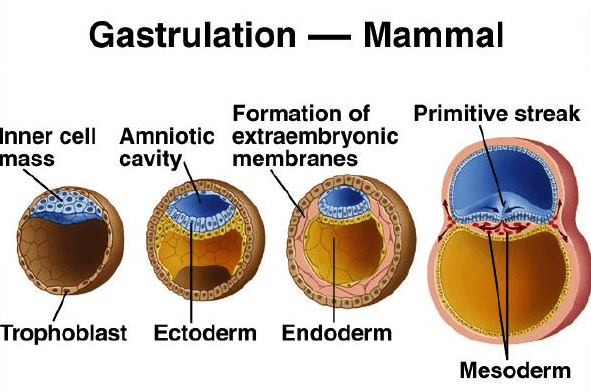
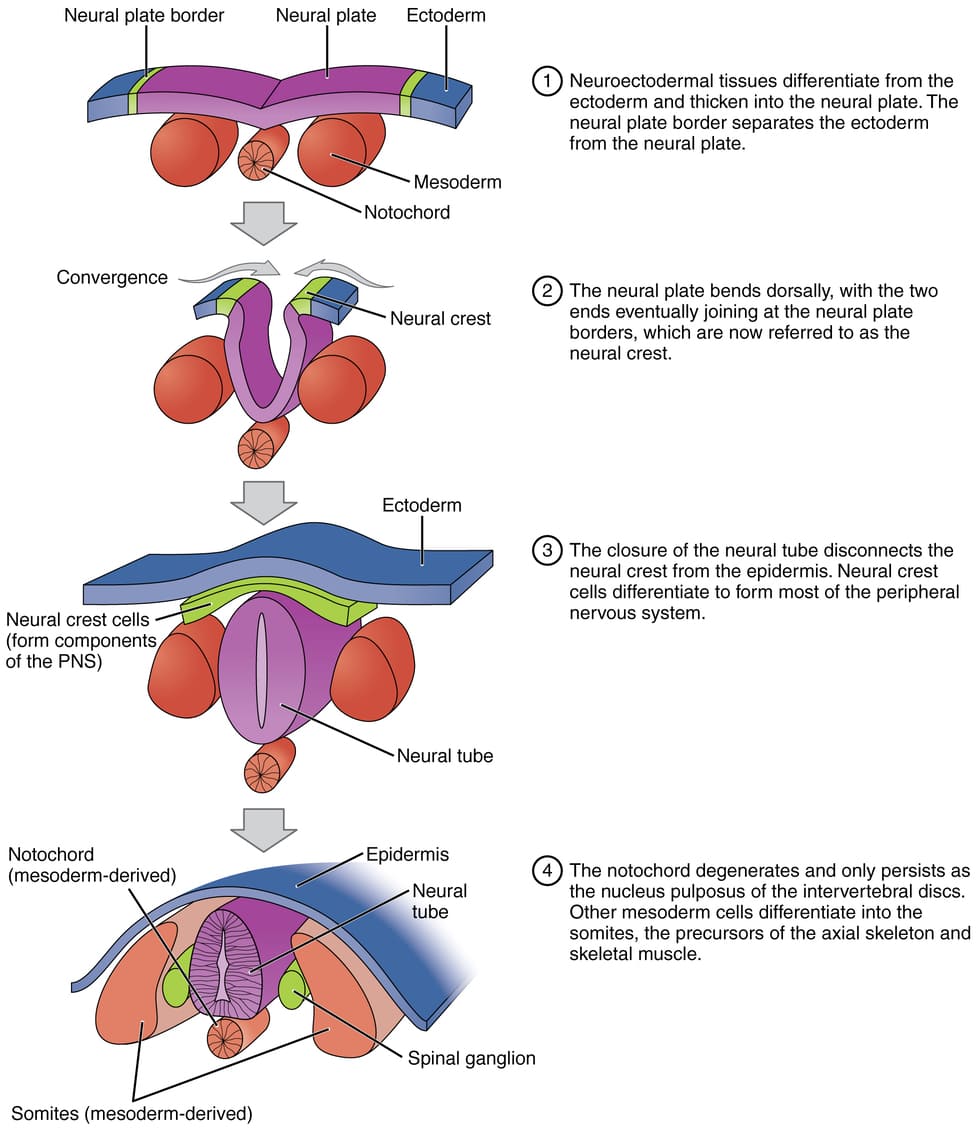
A. Formation of the Amniotic Cavity and Amnion
- Timeline: Begins around Day 8 post-fertilization.
Process:
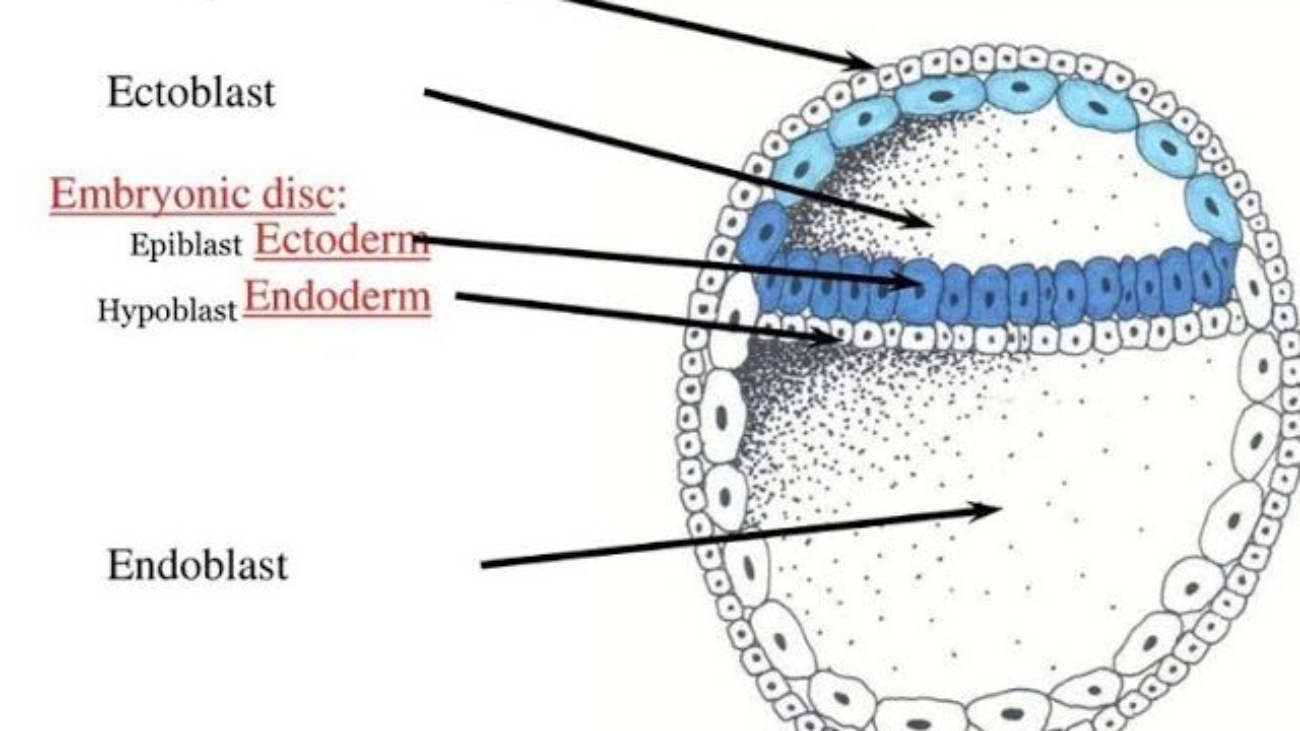
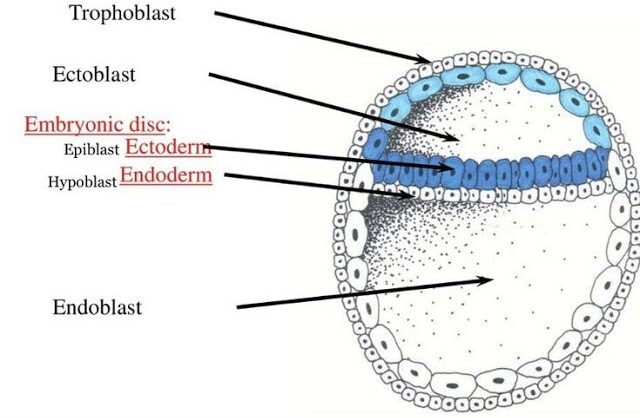
- Cavity Formation: As the blastocyst implants, a small space appears within the epiblast, which is the dorsal layer of the bilaminar germ disc (formed from the Inner Cell Mass).
- Enlargement: This space rapidly expands to become the amniotic cavity.
- Amnioblast Differentiation: Cells from the epiblast adjacent to the cytotrophoblast differentiate into thin, flattened cells called amnioblasts.
- Amniotic Membrane Formation: These amnioblasts, along with a layer of extraembryonic mesoderm, form the amnion, which eventually encloses the entire amniotic cavity.
- Roof and Floor: The roof is formed by the amnion/cytotrophoblast, while the floor is formed by the epiblast of the bilaminar germ disc.
Key Features & Function of the Amnion/Amniotic Fluid:
Amniotic Sac: The amnion forms the inner lining of the amniotic sac, which will eventually surround the entire embryo and then fetus.
Amniotic Fluid: The cavity fills with amniotic fluid. Initially derived from maternal blood, it is later maintained largely by fetal urine excretion and fetal swallowing. It serves crucial functions:
- Protection: Acts as a shock absorber against mechanical trauma.
- Temp Regulation: Maintains a constant, optimal intrauterine temperature.
- Symmetry & Movement: Allows symmetrical external growth and free movement for proper musculoskeletal development (preventing contractures).
- Prevents Adhesion: Stops the developing embryo from physically sticking to the amnion (which would cause amniotic band syndrome).
- Lung/GI Development: Fetal swallowing of the fluid actively aids the maturation of the GI tract; while fetal "breathing" movements pull fluid into the lungs, providing the mechanical stretch essential for pulmonary development.
Amniotic Fluid Volume Abnormalities
- Oligohydramnios (Too little fluid): Often caused by placental insufficiency or fetal renal agenesis (inability to produce urine). It can lead to Potter sequence (flattened face, clubbed feet, pulmonary hypoplasia) because the fetus is compressed without the fluid cushion.
- Polyhydramnios (Too much fluid): Often caused by fetal swallowing defects (e.g., esophageal atresia, anencephaly) or maternal diabetes. It can lead to premature rupture of membranes and preterm labor.
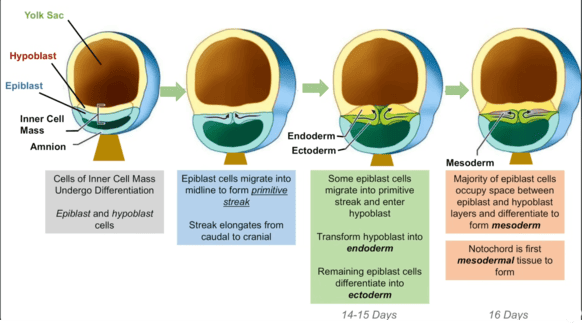
B. Formation of the Yolk Sac
- Timeline: Primary yolk sac begins around Day 9; Secondary yolk sac around Day 12-13.
1. Primary Yolk Sac (Exocoelomic Cavity) - Day 9
- Cells from the hypoblast (ventral layer) migrate and line the inner surface of the cytotrophoblast.
- These cells form a thin membrane called the exocoelomic membrane (Heuser's membrane).
- This membrane + hypoblast encloses the primary yolk sac.
- Position: The bilaminar disc lies between the Amniotic Cavity (dorsal) and the Primary Yolk Sac (ventral).
2. Extraembryonic Mesoderm - Day 10-11
A new layer of loose connective tissue appears and fills the space between the exocoelomic membrane/amnion externally and the cytotrophoblast internally.
3. Secondary Yolk Sac (Definitive) - Day 12-13
- The primary sac constricts due to chorionic cavity expansion.
- A smaller, definitive secondary yolk sac forms from a portion of the primary sac. The larger pinched-off part degenerates into exocoelomic cysts.
Key Features & Function of the Yolk Sac:
Early Nutrient Transfer
Plays a vital role in nutrient transfer to the embryo during weeks 2 and 3, before the uteroplacental circulation is fully functional.
Hematopoiesis
It is the primary site of early blood cell formation (Weeks 3-6). It produces primitive nucleated red blood cells expressing embryonic hemoglobin. After week 6, the fetal liver takes over this function.
Primordial Germ Cells
Precursors to sperm/eggs originate in the wall of the yolk sac around week 3. They migrate along the hindgut to reach the developing gonads by week 5.
Vestigial Structure
In humans, the yolk sac does not contain yolk. It is small, regresses rapidly by week 20, and is incorporated into the primitive gut tube and the umbilical cord.
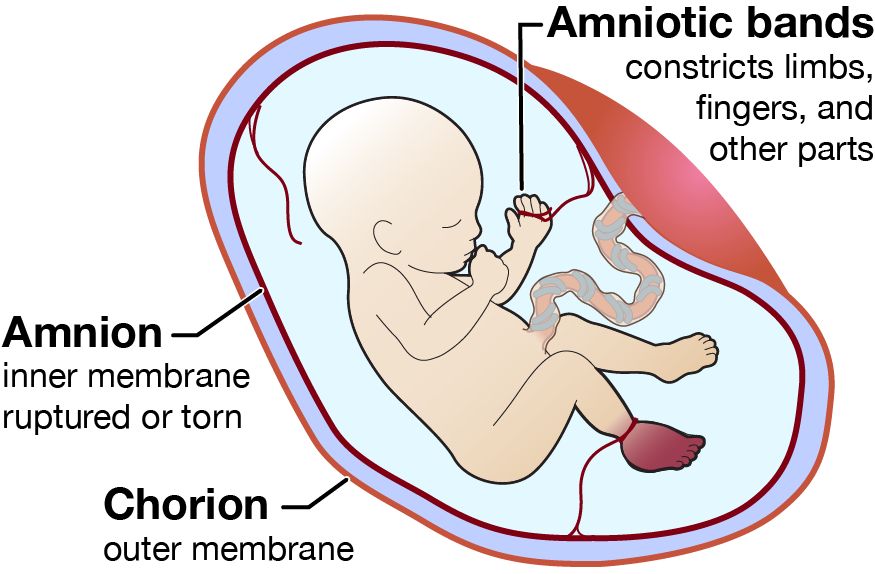
C. Formation of the Chorionic Cavity and Chorion
- Timeline: Begins around Day 11-12.
Process:
- Vacuole Formation: Numerous large spaces and vacuoles appear within the extraembryonic mesoderm.
- Coalescence: These fuse to form a large, isolated cavity called the chorionic cavity (extraembryonic coelom).
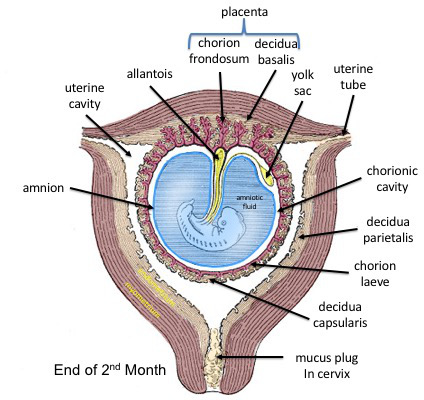
- Suspension of Embryo: The embryo (with its amnion and yolk sac) is suspended in this massive cavity by the connecting stalk (which is the future umbilical cord).
The Chorion (Outer Wall)
The chorion forms the wall of the chorionic cavity and is strictly formed by three layers (from outside to inside):
- Syncytiotrophoblast (outermost)
- Cytotrophoblast
- Somatic layer of extraembryonic mesoderm (innermost)
Functions:
- Chorionic Villi: Gives rise to the villi (the functional exchange units of the placenta).
- Protection: Forms an additional protective layer around the entire conceptus.
- Part of Placenta: The villous part (the chorion frondosum) forms the fetal component of the placenta, while the smooth part (chorion laeve) eventually fuses with the amnion.
- Central: Bilaminar germ disc centrally located.
- Dorsal: Amniotic cavity.
- Ventral: Secondary yolk sac.
- Surrounding all: Chorionic cavity (enclosed entirely by the Chorion).
- Bridge: Connecting stalk linking the bilaminar disc to the chorion.
D. The Allantois: Development and Significance
- Origin: Appears around Day 16-18 as a small, sausage-shaped diverticulum (outpouching) from the caudal wall of the yolk sac (specifically the hindgut), extending directly into the connecting stalk.
- Vascular Development: This is its most significant role in humans. Blood vessels develop in the mesoderm wall of the allantois to become the umbilical arteries and the umbilical vein. These vessels extend through the connecting stalk to strictly link the embryonic and placental circulation.
- Urinary Bladder Formation: The intraembryonic proximal part of the allantois incorporates into the developing cloaca to form the urinary bladder.
- Regression & Relationship to Umbilical Cord: In humans, the allantois itself is largely vestigial as a sac. As the amniotic cavity heavily expands and forms the definitive umbilical cord, the allantois regresses into a fibrous cord (the urachus) within it, while its associated vessels remain as the vital umbilical vessels. Postnatally, the urachus obliterates to form the median umbilical ligament.
Urachal Anomalies (Clinical Deep Dive)
If the allantois (urachus) fails to completely obliterate before birth, several defects can occur:
- Urachal Fistula: The entire duct remains open, connecting the bladder to the umbilicus. Urine will leak directly out of the baby's belly button.
- Urachal Cyst: Only a middle portion remains open, forming a fluid-filled cavity. It is usually asymptomatic unless it becomes infected, presenting as a painful umbilical mass.
- Urachal Sinus: The distal end near the umbilicus remains open, causing a small, discharging blind pouch.
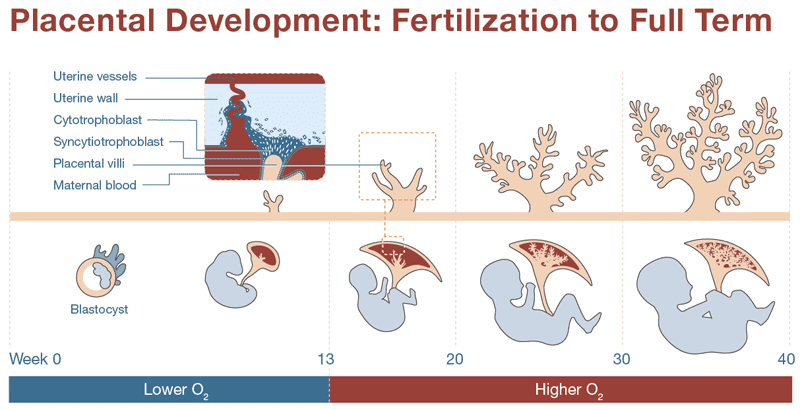
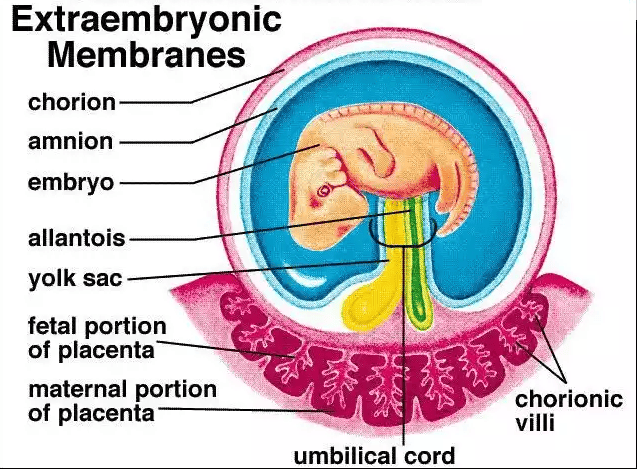
The Placenta
The placenta is a composite, highly specialized temporary organ formed by both fetal tissues (the chorionic villi/chorion frondosum) and maternal tissues (the decidua basalis of the modified uterine endometrium). It serves as the complete life-support system for the fetus and is also a critical endocrine organ driving the progression of pregnancy.
A. Development of the Placenta
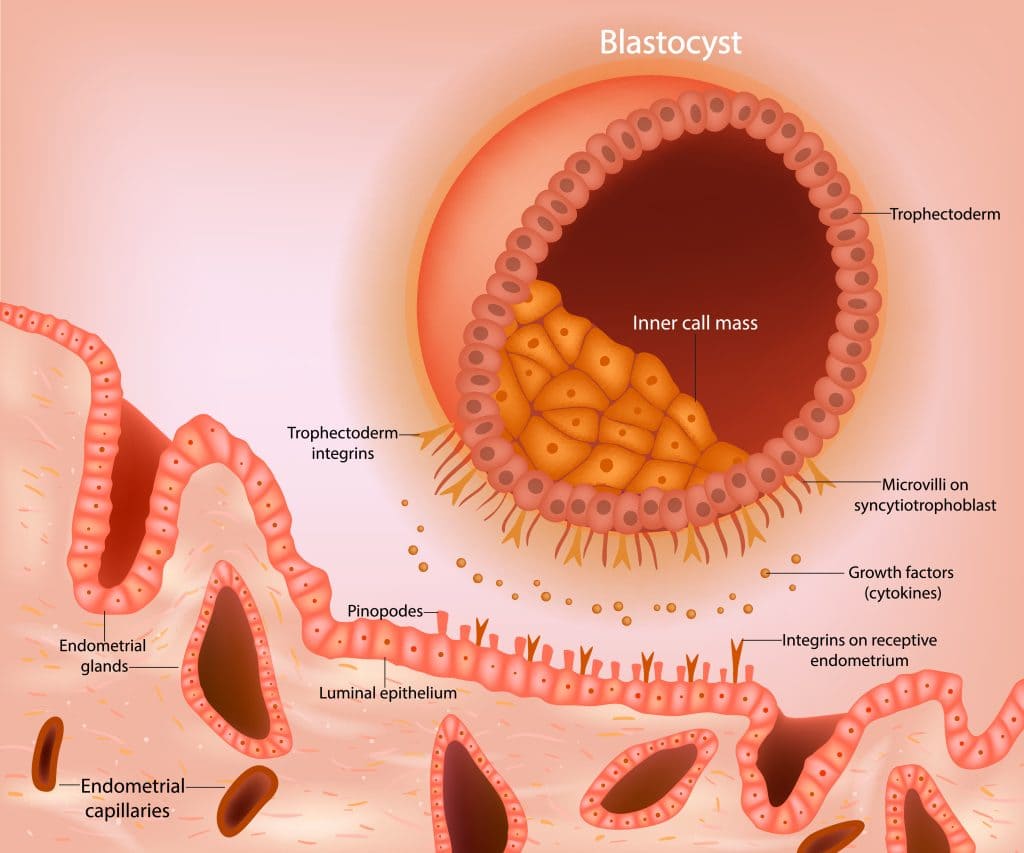
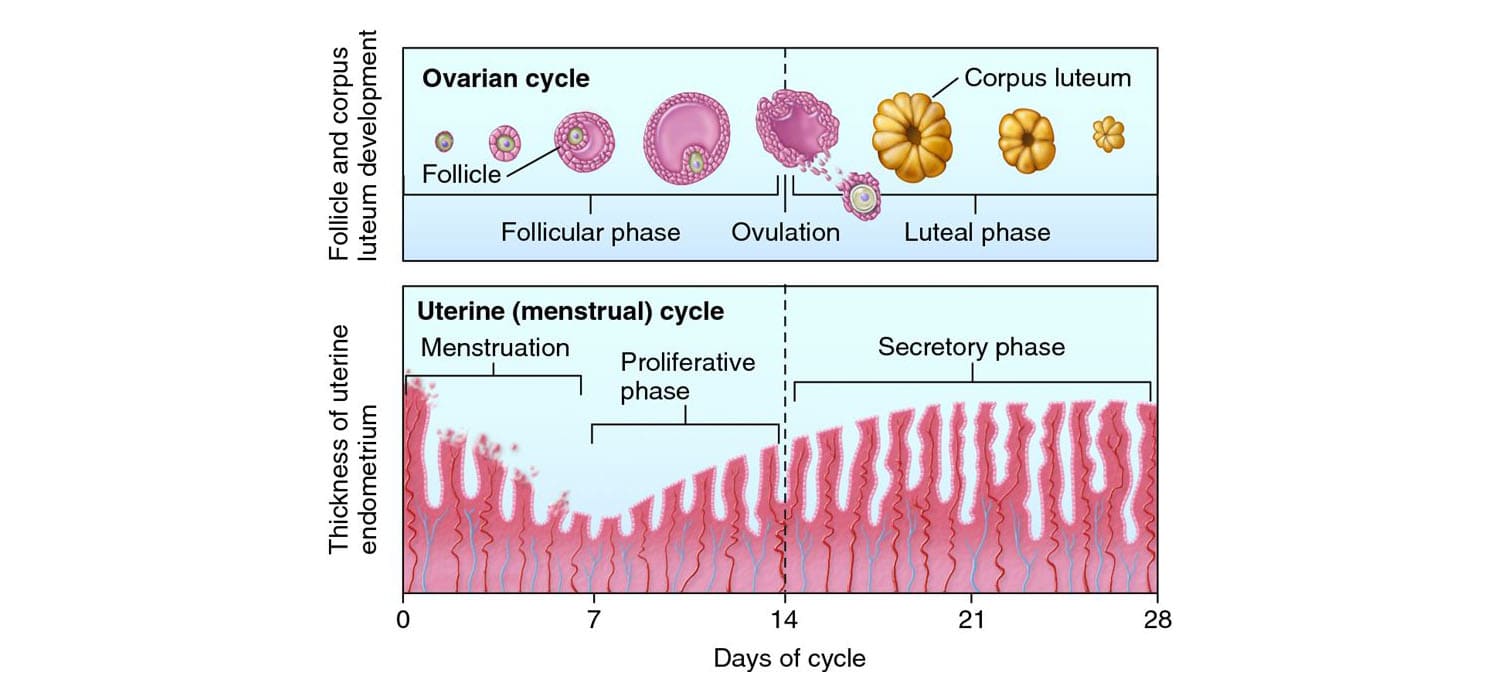
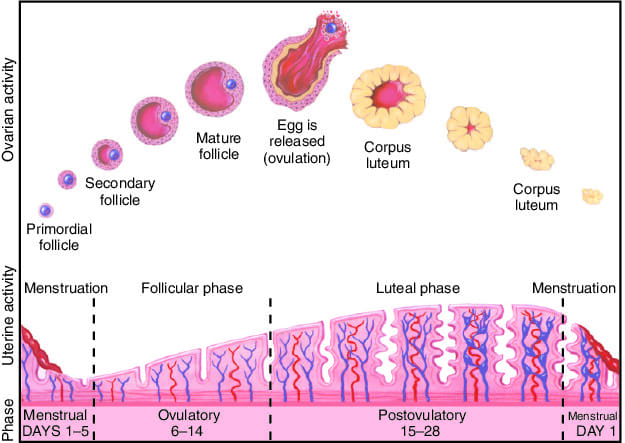
The placenta begins to form as soon as the blastocyst implants, with the trophoblast rapidly differentiating and aggressively invading the uterine wall. The fetal portion of the placenta develops through three distinct stages of villi maturation:
- Primary Villi: (Late week 2) Characterized by solid columns of cytotrophoblast cells penetrating outwards into the surrounding syncytiotrophoblast.
- Secondary Villi: (Early week 3) Extraembryonic mesenchyme (connective tissue) actively invades the core of the primary villi, giving them a structural backbone.
- Tertiary Villi: (Late week 3) Fetal blood vessels develop within the mesenchymal core of the secondary villi. This establishes the critical capillary networks of the feto-placental circulation. Simultaneously, maternal spiral arteries are remodeled by extravillous trophoblasts to pump high-volume, low-resistance maternal blood into the intervillous spaces to bathe these tertiary villi.
Pathophysiology of Preeclampsia
Normal placental development requires fetal trophoblast cells to deeply invade the maternal spiral arteries, destroying their muscular walls to make them wide, flaccid, low-resistance vessels. This ensures massive blood flow to the placenta. Preeclampsia occurs when this invasion is shallow or fails entirely. The maternal spiral arteries remain narrow and high-resistance, leading to severe placental ischemia. The distressed placenta releases toxic factors into the mother's blood, causing global maternal endothelial dysfunction, severe high blood pressure, and organ damage (kidneys, liver, brain).
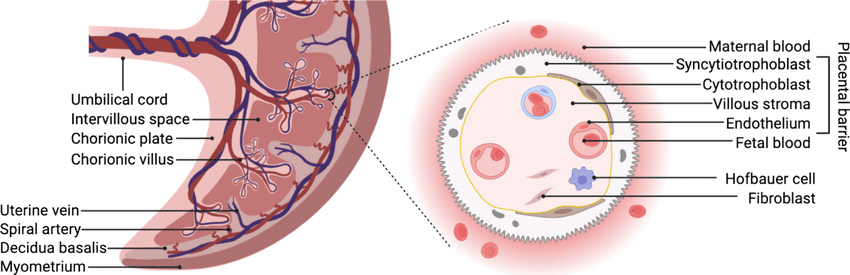
B. The Placental Barrier
This is not a true, impenetrable barrier but rather a highly selective, semi-permeable membrane across which all feto-maternal exchange occurs. Initially, it consists of four layers, which significantly thin out as pregnancy progresses to increase exchange efficiency:
- Syncytiotrophoblast (The outermost layer directly touching maternal blood).
- Cytotrophoblast (This layer becomes discontinuous and largely thins out by the 3rd trimester).
- Connective tissue of the villus core.
- Endothelium of the fetal capillaries.
Functions of the Placenta
The placenta is a transient but indispensable organ that acts as the lifeline between the mother and the developing fetus. It performs multiple critical functions, broadly categorized into metabolic, transfer (gas, nutrient, waste), barrier, and endocrine (hormonal) roles.
I. Metabolic Functions
The placenta is a metabolically highly active organ, performing synthesis, storage, and transfer of various substances essential for both fetal development and maternal adaptation to pregnancy.
Synthesis and Storage
- Glycogen Synthesis & Storage: The placenta actively synthesizes and stores glycogen (a polymer of glucose), especially in early pregnancy. This serves as a readily available energy reserve for the growing embryo/fetus when maternal glucose supply might be fluctuating or insufficient, particularly before the fetal liver is fully mature enough to store its own glycogen.
- Cholesterol Synthesis: The placenta synthesizes cholesterol, which is a vital precursor for steroid hormone production (estrogens, progesterone). While it can utilize maternal cholesterol from LDLs, its own synthetic capacity is highly important.
- Fatty Acid Synthesis: The placenta can synthesize some fatty acids, which are crucial for the massive expansion of fetal cell membranes and neural development.
- Protein Synthesis: The placenta synthesizes various proteins, including structural proteins for its own growth, enzymes required for its metabolic activities, and various growth factors and cytokines.
Nutrient Transfer (Feeding the Fetus)
The placenta acts as the primary organ for transferring nutrients from the maternal circulation to the fetal circulation. Mechanisms vary based on the molecule:
- Glucose:
- Mechanism: Primarily facilitated diffusion via glucose transporters (GLUTs), especially GLUT1 and GLUT3, located on both maternal and fetal sides of the syncytiotrophoblast.
- How it Works: Maternal glucose levels directly influence fetal glucose supply. The fetus relies almost entirely on maternal glucose for its rapid energy needs. The placenta extracts glucose from maternal blood and passes it efficiently down the concentration gradient to the fetal side.
- Amino Acids:
- Mechanism: Primarily secondary active transport, requiring cellular energy (ATP). There are multiple amino acid transporter systems (e.g., A, L, ASC systems).
- How it Works: Fetal amino acid concentrations are generally higher than maternal concentrations, demonstrating the active "pumping" action of the placenta against a gradient. These are crucial for massive fetal protein synthesis, muscle growth, and organ development.
- Fatty Acids & Lipids:
- Mechanism: Simple diffusion for smaller free fatty acids; facilitated diffusion and receptor-mediated endocytosis for larger fatty acids and cholesterol. Lipoprotein lipase in the placenta hydrolyzes maternal triglycerides into free fatty acids to cross.
- How it Works: Essential fatty acids (e.g., omega-3 DHA and omega-6 ARA) are absolutely vital for fetal brain myelinization and retinal development.
- Vitamins:
- Mechanism: Simple/facilitated diffusion and active transport.
- How it Works: All fat-soluble (A, D, E, K) cross via diffusion, but can cause toxicity if maternal levels are too high. Water-soluble vitamins (B, C) cross via active transport, ensuring higher concentrations in the fetus than in the mother.
- Minerals:
- Mechanism: Primarily active transport (Iron, Calcium, Phosphorus).
- How it Works: Iron is actively transported against a gradient for fetal erythropoiesis (RBC creation). Calcium and Phosphorus are actively pumped across, crucial for 3rd-trimester fetal skeletal mineralization.
II & III. Gas Exchange and Waste Excretion
Gas Exchange (O₂ & CO₂)
The placenta acts identically to the fetal lungs.
- Mechanism: Simple passive diffusion, strictly driven by partial pressure gradients.
- Oxygen: Diffuses from maternal blood (high PO2) into fetal blood. To maximize this, Fetal hemoglobin (HbF) has a much higher affinity for O₂ than adult HbA, shifting the oxygen-dissociation curve to the left, actively pulling oxygen from the mother's red blood cells even at lower pressures.
- Carbon Dioxide: Fetal blood has high CO₂. It releases it into maternal blood (which has lower CO₂). The maternal lungs then exhale it.
Waste Excretion
- Substances: Urea, Creatinine, Bilirubin, Uric Acid.
- Mechanism: Primarily simple diffusion, driven by concentration gradients. These metabolic waste products generated by the fetus are transferred to maternal blood. The maternal kidneys and liver then clear them from her system.
IV. Barrier Function (Protective Role)
The placenta acts as a selective barrier, protecting the fetus from potentially harmful substances while allowing essential nutrients to pass. However, it is fundamentally an imperfect barrier.
1. Antibodies (Immunological Protection)
- Mechanism: Active transport (pinocytosis) via special Fc receptors (FcRn) located on the syncytiotrophoblast.
- How it Works: Only maternal IgG antibodies are actively transported across the placenta (occurring predominantly in the 3rd trimester). This provides essential passive immunity to the newborn against many diseases the mother has encountered (measles, rubella, tetanus) for the first 6 months of life. Other immunoglobulins (IgM, IgA, IgD, IgE) are too large or lack receptors and do not cross significantly.
2. TORCHES Infections (Barrier Failure)
The placenta is generally highly effective against most bacteria, but a specific group of pathogens known by the acronym "TORCHES" can cross the placental barrier, leading to devastating congenital defects, miscarriages, or severe neonatal illness.
| Pathogen (TORCHES) | Mechanism / Source | Specific Fetal / Neonatal Consequences |
|---|---|---|
| Toxoplasmosis (Toxoplasma gondii) | Parasite. Ingested via raw meat or contact with infected cat feces. | Classic Triad: Chorioretinitis (blindness), Hydrocephalus (fluid in brain), Intracranial calcifications. |
| Other (Syphilis) | Treponema pallidum. Transplacental transmission. | Maculopapular rash, snuffles (bloody nasal discharge), saddle nose deformity, Hutchinson teeth (notched), saber shins, deafness. |
| Other (Varicella-Zoster) | Chickenpox virus. | Congenital Varicella Syndrome: Cutaneous scarring (cicatricial lesions), limb hypoplasia, cataracts, brain damage. |
| Other (Parvovirus B19) | Respiratory droplets. Causes "Fifth disease" in kids. | Virus aggressively attacks fetal red blood cell precursors leading to profound fetal anemia, heart failure, and Hydrops Fetalis (massive total body edema). |
| Rubella | German measles virus. Very high risk in 1st trimester. | Classic Triad: Cataracts, Sensorineural deafness, Congenital Heart Disease (specifically Patent Ductus Arteriosus / PDA). Also "blueberry muffin" rash. |
| Cytomegalovirus (CMV) | Most common congenital viral infection. Body fluids. | Microcephaly, periventricular calcifications, profound hearing loss, seizures, petechial rash. |
| Herpes Simplex Virus (HSV) | Usually transmitted ascending through birth canal during delivery, but can cross placenta. | Skin/eye/mouth vesicles, severe viral encephalitis, disseminated multiorgan disease. |
| Enteroviruses / Strep (Group B) | GBS colonizes maternal vagina/rectum. Transmitted during birth. | GBS is the leading cause of neonatal sepsis, meningitis, and pneumonia. |
3. Drugs, Toxins, and Teratogens
Most drugs (especially lipid-soluble ones), alcohol, nicotine, and environmental toxins (lead, mercury) easily cross via simple diffusion. While the placenta possesses enzymes (like Cytochrome P450s) to metabolize some toxins, it is easily overwhelmed. Alcohol causes Fetal Alcohol Syndrome (facial abnormalities, severe mental retardation), and smoking severely restricts placental blood flow causing Intrauterine Growth Restriction (IUGR).
4. Maternal Thyroid Hormones
Maternal T3 and T4 cross the placenta via active transport in early pregnancy. This is absolutely critical for early fetal brain development and neurogenesis before the fetal thyroid gland is fully formed and functional (around week 12).
V. Hormonal (Endocrine) Functions
The placenta is a massive, autonomous endocrine organ, producing a wide array of hormones that hijack maternal physiology, maintain the pregnancy, and promote fetal growth.
hCG (Human Chorionic Gonadotropin)
- Source: Syncytiotrophoblast.
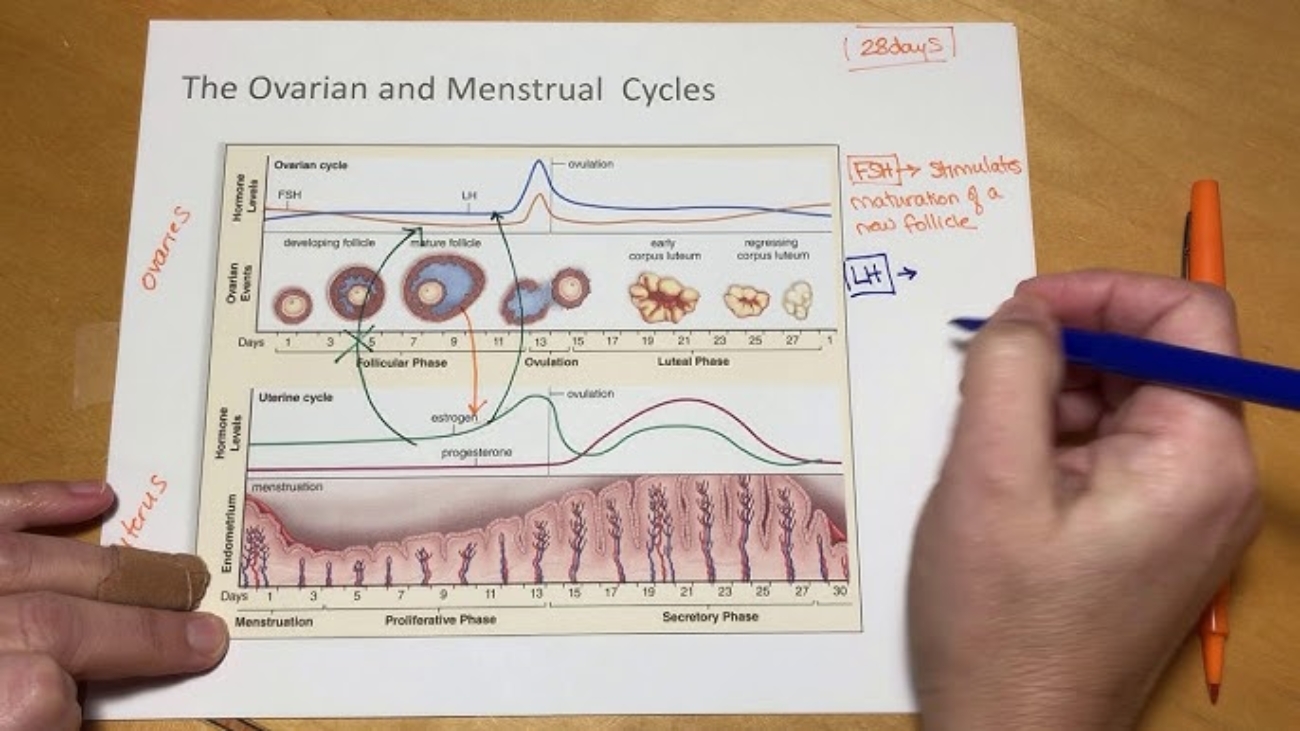
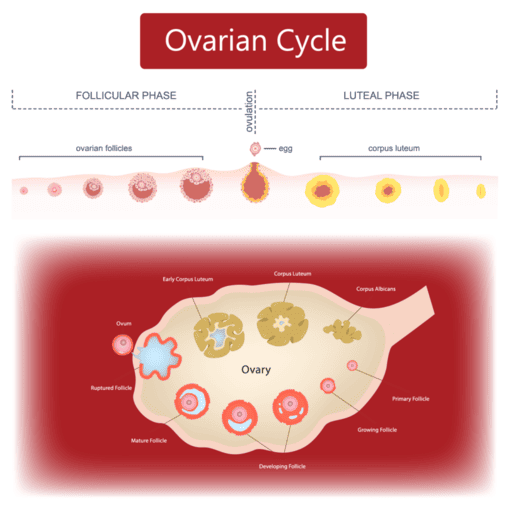
- Function: Acts identical to LH. Its primary role is to "rescue" and maintain the Corpus Luteum in the ovary during early pregnancy. The corpus luteum pumps out progesterone to prevent the endometrium from shedding (menstruation). After 7-10 weeks, the placenta makes its own progesterone, and hCG levels drop.
- Clinical: It is the basis of all maternal blood and urine pregnancy tests. It also stimulates fetal testes to produce testosterone for male differentiation.
hPL (Human Placental Lactogen)
- Source: Syncytiotrophoblast.
- Function: Causes profound maternal metabolic adaptation. It acts as an anti-insulin hormone (creating maternal insulin resistance). This decreases maternal glucose use, effectively diverting all the free glucose to the fetus. It also causes lipolysis, mobilizing maternal fatty acids for her own energy.
- Clinical: Overproduction is a major driver of Gestational Diabetes.
Progesterone
- Source: Syncytiotrophoblast (takes over primary production from the corpus luteum around 7-10 weeks). Synthesized from maternal cholesterol.
- Function: The "Hormone of Pregnancy." Maintains massive uterine quiescence by relaxing the myometrium (prevents premature contractions and labor). It maintains the highly vascular secretory endometrium (decidua) and thickens the cervical mucus plug to create an infection barrier.
Estrogens (Estrone, Estradiol, Estriol)
- Source: The Feto-Placental Unit. The placenta lacks specific enzymes to make estrogen from scratch. It relies on the fetal adrenal gland to make DHEAS, which travels to the fetal liver to be hydroxylated (16-OH-DHEAS), and then travels to the placenta. The placenta uses the enzyme Aromatase to convert this into Estriol (E3).
- Function: Stimulates massive uterine growth and blood flow. Promotes mammary gland ductal growth. Towards term, it increases myometrial sensitivity to oxytocin and prostaglandins to prepare for labor. Maternal Estriol levels are a direct clinical marker of fetal health.
Other Important Hormones:
- CRH (Corticotropin-Releasing Hormone): Known as the "Placental Clock." Rising exponential levels in late pregnancy trigger the onset of labor. It also stimulates the fetal adrenal gland to produce cortisol, which matures the fetal lungs (surfactant production).
- Relaxin: Softens the pelvic ligaments and the pubic symphysis to widen the birth canal for childbirth. Aids in cervical ripening (effacement and dilation) and heavily relaxes uterine muscle.

The Umbilical Cord
The umbilical cord develops from the connecting stalk and serves as the vital, physical connection between the fetus and the placenta, facilitating all exchange.
- Two Umbilical Arteries: Wrap spirally around the vein. They carry DEOXYGENATED blood and metabolic waste away from the fetus to the placenta. Clinical note: A Single Umbilical Artery (SUA) is an anomaly associated with cardiovascular and renal defects in the fetus.
- One Umbilical Vein: Carries highly OXYGENATED blood and vital nutrients directly from the placenta to the fetus.
- Wharton's Jelly: A thick, specialized, gelatinous connective tissue (rich in hyaluronic acid) that tightly surrounds the vessels. It acts as a physical shock absorber, protecting the vital vessels from kinking, compression, or knotting during fetal movements.
Fetal Circulation
In utero, the fetus relies entirely on the placenta for respiration, nutrition, and excretion, as its own lungs and GI tract are non-functional and filled with fluid. Fetal circulation is ingeniously designed with a series of bypasses (shunts) to accommodate this reality, ensuring the most highly oxygenated blood reaches the most critical organs (the developing brain and heart).
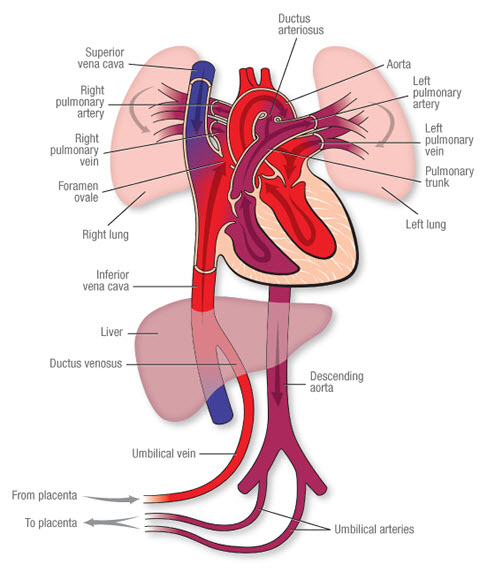
The Pathway of Fetal Blood Flow
- Oxygenated Blood from Placenta: Rich blood (roughly 80% O2 saturation) flows from the placenta to the fetus via the single Umbilical Vein.
- Bypassing the Liver (First Shunt): Blood travels toward the fetal liver. Because the liver does not need all this oxygen yet, about 50% of this blood completely bypasses the liver sinusoids by flowing through a special shunt called the Ductus Venosus. This shunt empties directly into the Inferior Vena Cava (IVC), mixing with deoxygenated blood from the lower body.
- Bypassing the Lungs (Second Shunt): This mixed blood enters the Right Atrium. Because the fetal lungs are collapsed and filled with fluid, pulmonary vascular resistance is extremely high. To avoid pumping blood into closed lungs, most of the oxygenated blood is directed by a valve (Eustachian valve) straight across the atrial septum through a hole called the Foramen Ovale, moving directly into the Left Atrium.
- Supplying Vital Organs: From the left atrium, blood enters the left ventricle and is pumped into the ascending aorta. This highly oxygenated blood is strategically prioritized to supply the coronary arteries (heart) and the carotid arteries (brain) first.
- Bypassing the Lungs (Third Shunt): Meanwhile, heavily deoxygenated blood returning from the fetal upper body (via the Superior Vena Cava) enters the right atrium and drops into the right ventricle. It is pumped into the pulmonary artery. Since the lungs are high-resistance, this blood meets a wall. It is instantly shunted away from the pulmonary artery, through a wide vessel called the Ductus Arteriosus, directly into the descending aorta (post-carotid arteries, so it doesn't dilute the brain's oxygen).
- Return to Placenta: This deoxygenated blood from the descending aorta flows down into the internal iliac arteries, enters the two Umbilical Arteries, and travels back to the placenta for re-oxygenation and waste clearance.
A simple way to remember the key structures in exact order of blood flow:
P-U-D-I-F-D-U
(Placenta → Umbilical Vein → Ductus Venosus → IVC → Foramen Ovale → Ductus Arteriosus → Umbilical Arteries)
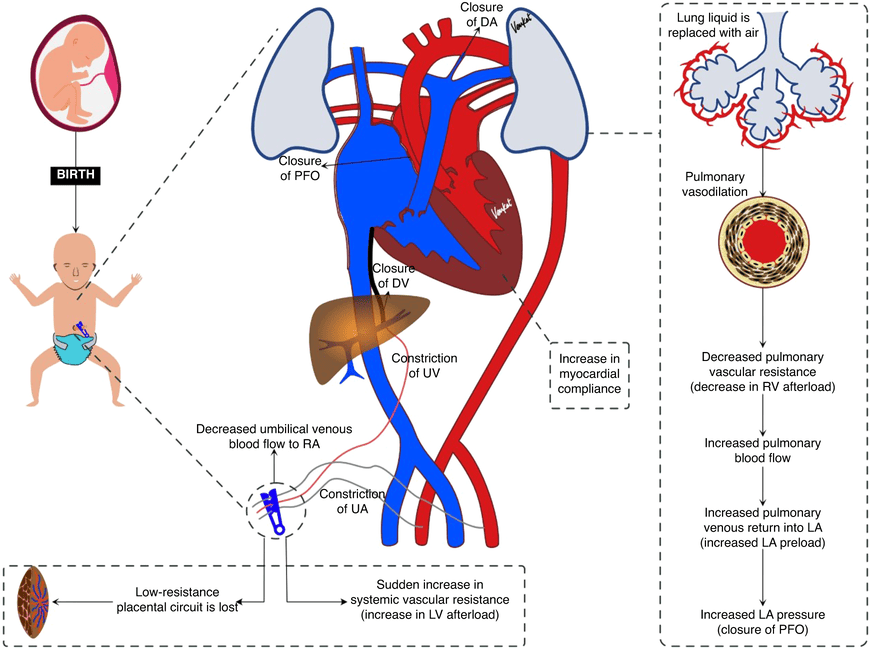
Changes After Birth: Adaptation to Extrauterine Life
At birth, with the neonate's first breath and the physical clamping of the umbilical cord, a series of rapid, profound, and permanent physiological changes occur to instantly transition the circulatory system from fetal to adult patterns.
Closure of Fetal Shunts
- Foramen Ovale Closure: The baby takes its first breath. The lungs expand, dramatically dropping pulmonary vascular resistance. Blood rushes into the lungs and returns massively to the left atrium. This sudden increase in left atrial pressure physically pushes the flap of the septum primum against the septum secundum, instantly closing the Foramen Ovale.
- Ductus Arteriosus Closure: The expansion of the lungs greatly increases systemic blood oxygen (PO2). High oxygen levels, combined with a massive drop in circulating maternal prostaglandins (which normally kept the ductus open), cause the thick muscular walls of the Ductus Arteriosus to aggressively constrict and close within hours.
- Ductus Venosus Closure: When the umbilical cord is clamped, blood flow through the umbilical vein immediately ceases. The ductus venosus sphincter contracts, and the vessel rapidly closes.
Formation of Ligaments (Adult Remnants)
The fetal vascular structures, no longer in use, undergo fibrosis and remain in the adult body as non-functional ligaments:
| Fetal Structure | Adult Remnant |
|---|---|
| Umbilical Vein | Ligamentum Teres (Round ligament of the liver, sitting in the free edge of the falciform ligament). |
| Ductus Venosus | Ligamentum Venosum (Attached to the liver). |
| Foramen Ovale | Fossa Ovalis (The oval depression visible in the right atrium). |
| Ductus Arteriosus | Ligamentum Arteriosum (Connecting the pulmonary artery to the aortic arch). |
| Umbilical Arteries | Medial Umbilical Ligaments (Located on the inner abdominal wall). *Note: The proximal parts remain open to supply the superior bladder. |
Test Your Knowledge
Check your understanding of the concepts covered in this post.
1. Which fetal membrane directly surrounds the embryo/fetus and is filled with amniotic fluid?
- Chorion
- Yolk sac
- Amnion
- Allantois
2. The primary function of amniotic fluid includes all of the following EXCEPT:
- Cushioning the fetus from trauma
- Providing nutrients for fetal growth
- Allowing for fetal movement
- Maintaining fetal body temperature
3. The placenta is formed from tissues derived from both the mother and the fetus. Which fetal component primarily contributes to the formation of the placenta?
- Amnion
- Yolk sac
- Trophoblast
- Inner cell mass
4. Which part of the placenta is the site of nutrient, gas, and waste exchange between mother and fetus?
- Decidua basalis
- Chorionic villi
- Amniotic sac
- Umbilical vein
5. The umbilical cord typically contains how many blood vessels?
- One artery, one vein
- Two arteries, one vein
- One artery, two veins
- Two arteries, two veins
6. Which of the following fetal shunts bypasses the liver, directing oxygenated blood from the umbilical vein directly to the inferior vena cava?
- Foramen ovale
- Ductus arteriosus
- Ductus venosus
- Patent foramen ovale
7. In fetal circulation, the highest oxygen saturation is found in the blood within the:
- Umbilical arteries
- Pulmonary artery
- Umbilical vein
- Aorta
8. The foramen ovale is a shunt that allows blood to bypass which fetal organ?
- Liver
- Lungs
- Kidneys
- Brain
9. What is the primary reason why fetal lungs receive only a small amount of blood flow in utero?
- They are not yet fully developed.
- The fetus breathes amniotic fluid.
- High pulmonary vascular resistance.
- The lungs are filled with meconium.
10. After birth, the ductus arteriosus typically closes to become the:
- Ligamentum teres
- Ligamentum venosum
- Ligamentum arteriosum
- Medial umbilical ligaments
11. The fetal component of the placenta, characterized by its finger-like projections, is called the _____________.
12. The gelatinous substance that surrounds the blood vessels within the umbilical cord, protecting them from compression, is known as _____________.
13. The fetal shunt that connects the pulmonary artery to the aorta, bypassing the fetal lungs, is the _____________.
14. The part of the maternal endometrium that forms the maternal portion of the placenta is the _____________.
15. The small, usually non-functional, sac that extends from the embryonic gut into the connecting stalk, contributing to early blood formation and primordial germ cell migration, is the _____________.
Quiz Complete!
Your Score:
0%
0 / 0 correct