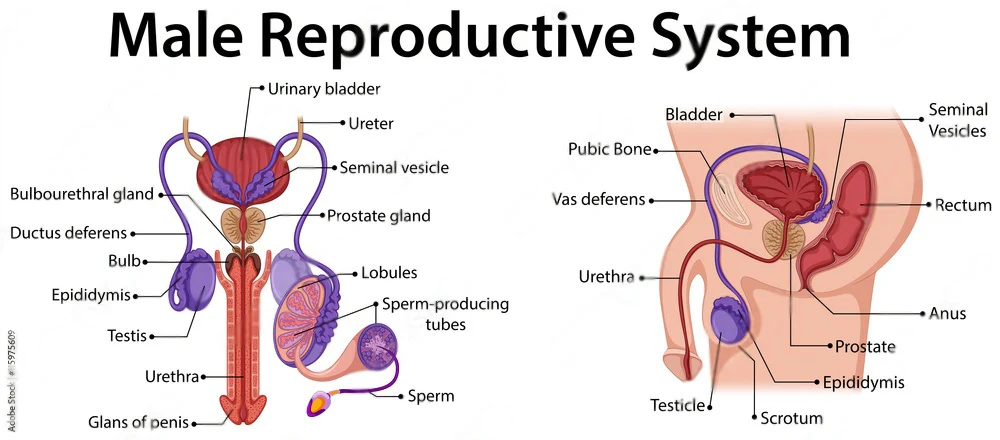
Genitourinary System Male Reproductive System
The Genitourinary System: An Introduction
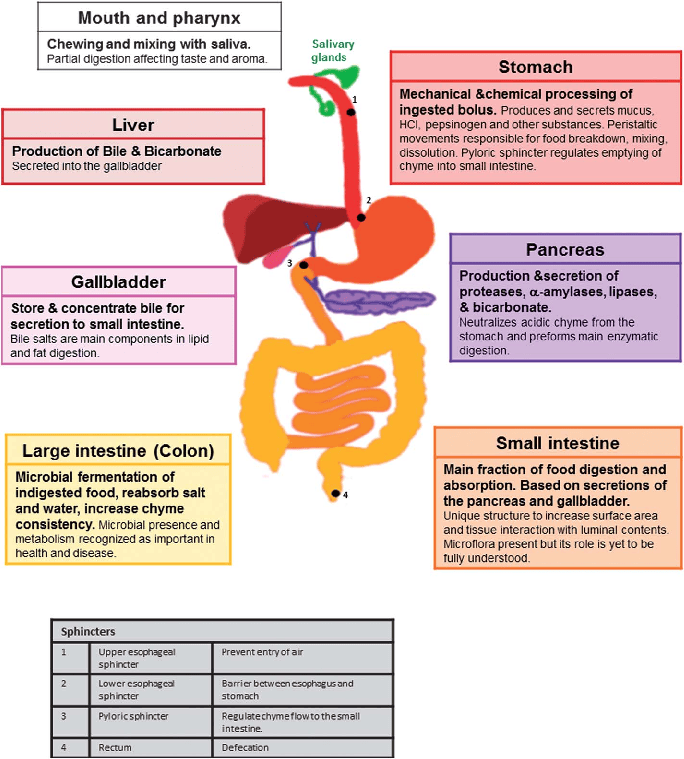
The genitourinary system encompasses the reproductive organs and the urinary system. While functionally distinct in many ways, they share developmental origins and anatomical proximity, particularly in males, hence their common classification. This section will focus on the reproductive system.
The Reproductive System: Core Principles
The reproductive system is a unique and fundamental biological system with several distinguishing characteristics:
- Sexual Dimorphism: It exhibits marked differences between males and females in terms of anatomy, physiology, and hormonal regulation. This is the most obvious difference from other organ systems.
- Latency in Development: Unlike most other body systems which are functional from birth, the reproductive system undergoes a prolonged period of dormancy (childhood) before maturing and becoming fully functional at puberty.
- Not Essential for Individual Survival: While crucial for the propagation of the species, an individual can survive perfectly well without a functional reproductive system. This contrasts with systems like the cardiovascular, respiratory, or nervous systems, which are indispensable for individual life.
Functional Classification of the Reproductive System:
1. Primary Reproductive Organs (Gonads)
- Males: Testes (produce sperm and male hormones).
- Females: Ovaries (produce ova/eggs and female hormones).
- These organs are responsible for gamete (sex cell) production and hormone synthesis.
2. Accessory Reproductive Organs (Ducts and Glands)
These structures are responsible for the transport, maturation, and nourishment of gametes, and facilitate fertilization.
- Males: Epididymis, vas deferens, ejaculatory ducts, seminal vesicles, prostate gland, bulbourethral glands.
- Females: Uterine tubes (fallopian tubes), uterus, vagina.
3. Secondary Sexual Characteristics
These are physical traits that distinguish males from females but are not directly involved in reproduction. They develop under the influence of sex hormones at puberty.
- Males: Deepening voice, facial and body hair growth, increased muscle mass, broader shoulders.
- Females: Breast development, widening of hips, growth of pubic and axillary hair.
- These characteristics also serve as "sexual attractants" or signals for reproductive fitness.
Anatomical Classification of the Reproductive System:
- External Genitalia (Perineal Structures):
- Males: Penis and Scrotum (containing the testes).
- Females: Vulva (comprising the labia majora, labia minora, clitoris, vestibule, and associated glands).
- Internal Genitalia:
- Males: Testes (though partially in scrotum, functionally internal), epididymis, vas deferens, ejaculatory ducts, seminal vesicles, prostate gland, bulbourethral glands.
- Females: Ovaries, uterine tubes, uterus, vagina.
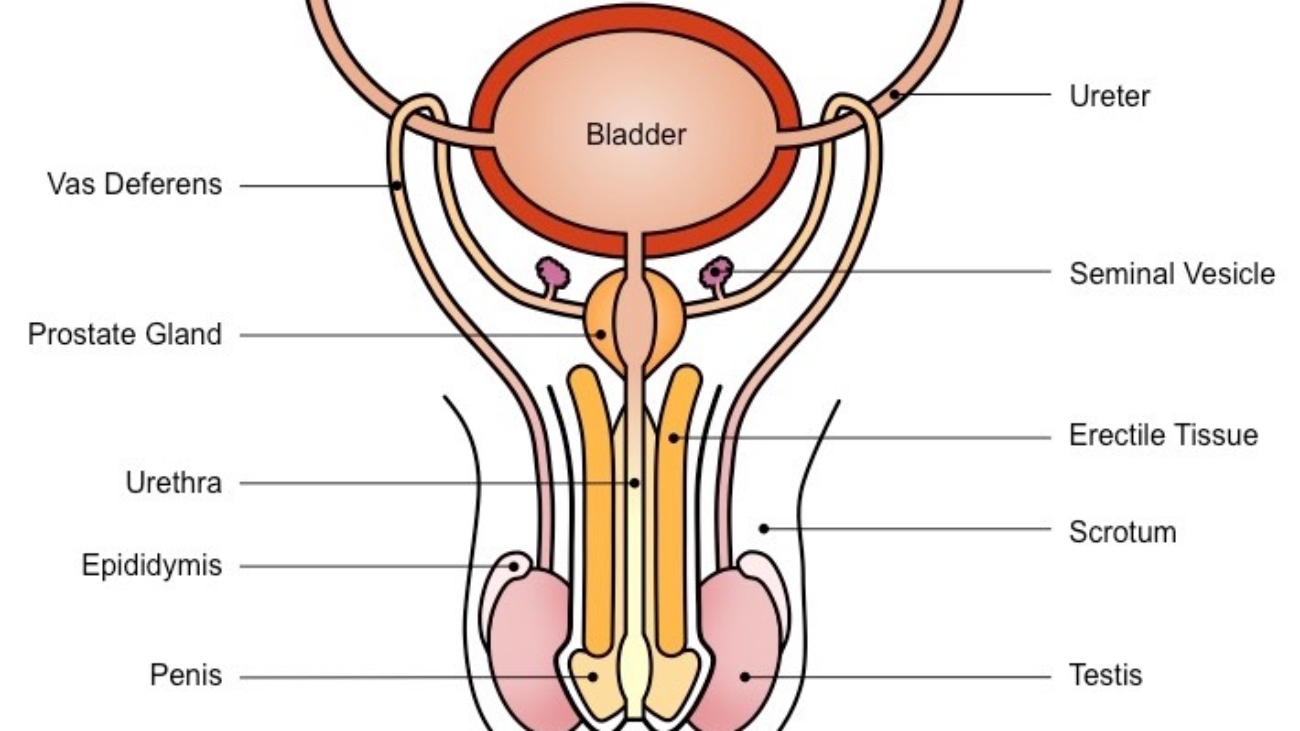
Male External Genitalia
The male external genitalia are located in the perineum and consist of the penis and the scrotum.
The Penis
The penis is the male organ of copulation and urination.
- Analogue to the Female Clitoris: Both develop from the same embryonic structure (genital tubercle), hence they are homologous organs. However, their primary functions in adulthood differ significantly.
- Primary Function: To deliver sperm into the female reproductive tract during sexual intercourse and to serve as the exit for urine.
Structure of the Penis:
The penis consists of a fixed root and a free, pendulous body (shaft), terminating in the glans penis.
1. Root of the Penis
The internal, attached portion of the penis. Composed of three erectile tissue structures:
- Bulb of the Penis: A single, ventrally placed, rounded mass of erectile tissue. It is the proximal continuation of the corpus spongiosum and is attached to the perineal membrane. It is covered externally by the bulbospongiosus muscle.
- Crura of the Penis (Crus, singular): Two diverging masses of erectile tissue (right and left) that are the proximal continuations of the corpora cavernosa. They are firmly attached to the inferior rami of the pubis and ischium. Each crus is covered externally by an ischiocavernosus muscle.
2. Body (Shaft) of the Penis
The free, movable portion of the penis, covered by skin. Composed of three cylindrical masses of erectile tissue arranged longitudinally:
- Corpus Spongiosum (Corpus Cavernosum Urethrae):
- A single, ventrally placed column of erectile tissue.
- It is the distal continuation of the bulb of the penis.
- Crucially, it is traversed throughout its length by the penile (spongy) urethra.
- Distally, it expands to form the glans penis.
- Corpora Cavernosa (Corpus Cavernosum, singular):
- Two dorsally placed (relative to the corpus spongiosum and urethra), parallel columns of erectile tissue.
- They are the distal continuations of the crura of the penis.
- Their distal ends are covered by the expanded glans penis.
- These are the primary erectile bodies responsible for penile rigidity during erection.
3. Glans Penis:
- The expanded, conical distal end of the corpus spongiosum.
- Covers the distal ends of the corpora cavernosa.
- Highly sensitive due to abundant nerve endings.
- Corona: The prominent rim at the base of the glans.
- Prepuce (Foreskin): A retractable fold of skin that covers the glans penis in uncircumcised males.
Coverings of the Penis:
- Skin: Thin, loose, and usually hairless, allowing for movement during erection.
- Superficial (Dartos) Fascia: A layer of loose connective tissue and smooth muscle (dartos muscle) just beneath the skin.
- Deep (Buck's) Fascia of the Penis: A strong, inelastic, fibrous sheath that binds the three corpora together and surrounds the neurovascular structures. This fascia plays a crucial role in maintaining erection by compressing emissary veins.
Neurovascular Supply:
- Arterial Supply: Primarily from the internal pudendal arteries, which give rise to the:
- Dorsal arteries of the penis: Supply the skin, fascia, and glans.
- Deep arteries of the penis (Cavernosal arteries): Run within the corpora cavernosa and are essential for erection.
- Bulbourethral arteries: Supply the bulb of the penis and corpus spongiosum.
- Venous Drainage: Deep veins (e.g., deep dorsal vein of the penis) drain the erectile tissue, and superficial veins drain the skin.
- Innervation:
- Somatic (Pudendal Nerve): Provides sensory innervation to the skin of the penis and glans.
- Autonomic (Pelvic Splanchnic Nerves - Parasympathetic; Hypogastric Plexus - Sympathetic): Essential for the erectile and ejaculatory reflexes. Parasympathetic stimulation causes vasodilation leading to erection, while sympathetic stimulation leads to ejaculation and detumescence.
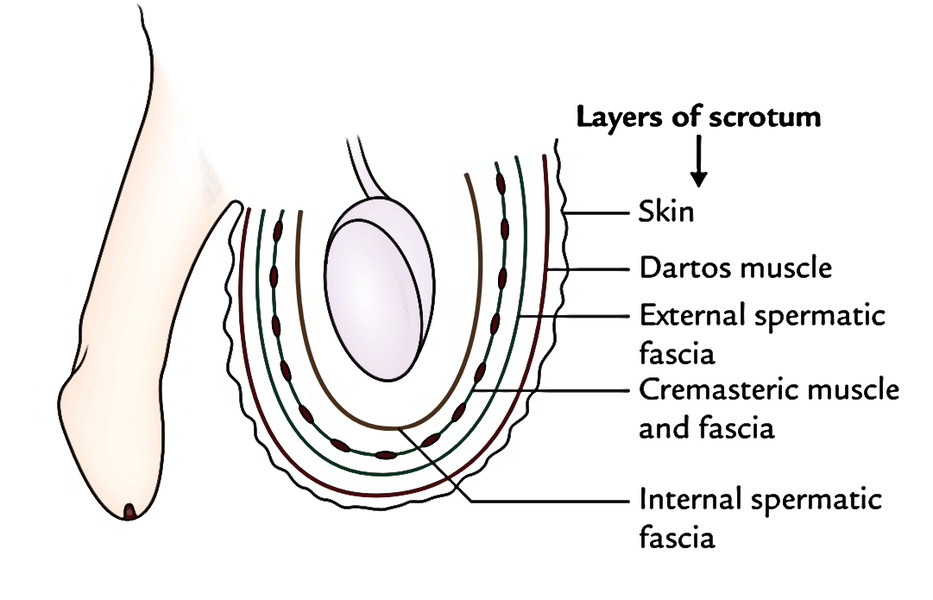
The Scrotum
The scrotum is a cutaneous fibromuscular sac that encloses and protects the testes, epididymides, and the inferior parts of the spermatic cords.
- Analogue to the Female Labia Majora: Both develop from the same embryonic structures (labioscrotal folds), hence they are homologous.
- Location: An outpouching of the anterior abdominal wall, located in the perineum, inferior to the pubic symphysis and anterior to the anus.
Layers of the Scrotum:
The scrotum is formed by several layers derived from the anterior abdominal wall during testicular descent.
- Skin:
- Thin, wrinkled, and typically darker pigmented than surrounding skin.
- Presence of sebaceous glands and hair follicles (often sparse).
- A median ridge, the scrotal raphe, marks the line of fusion of the embryonic labioscrotal folds.
- Dartos Fascia (Superficial Fascia):
- Consists of a mixture of loose connective tissue and smooth muscle fibers known as the dartos muscle.
- Key Function: The dartos muscle contracts in response to cold, causing the scrotal skin to wrinkle and reducing the surface area to conserve heat, and relaxes when warm to increase surface area for heat dissipation. Note: This layer represents both the fatty and membranous layers of the superficial fascia of the anterior abdominal wall. It does not contain Colle's fascia, which is the membranous layer of the superficial perineal pouch.
- External Spermatic Fascia: Derived from the aponeurosis of the external oblique muscle of the anterior abdominal wall.
- Cremasteric Fascia and Muscle:
- Derived from the internal oblique muscle and its fascia.
- Cremaster muscle: Striated skeletal muscle fibers that form loops around the testis and spermatic cord.
- Key Function: The cremaster muscle contracts in response to cold or sexual arousal, pulling the testis closer to the body for warmth or protection (cremasteric reflex).
- Internal Spermatic Fascia: Derived from the fascia transversalis.
- Tunica Vaginalis (Parietal Layer):
- A remnant of the peritoneum that invaginates with the testis during its descent.
- The parietal layer lines the inner surface of the scrotal sac.
- The visceral layer covers the testis and epididymis.
- A potential space exists between the parietal and visceral layers (cavity of the tunica vaginalis), which normally contains a small amount of fluid to allow the testis to move freely.
Important Note: The deep fascia, the transversus abdominis muscle, and extraperitoneal fat of the anterior abdominal wall are not represented in the scrotal layers.
Neurovascular Supply of the Scrotum:
- Arterial Supply: Branches from the internal pudendal, external pudendal, and cremasteric arteries.
- Venous Drainage: Follows the arteries.
- Innervation: The skin of the scrotum is highly sensitive and receives innervation from several sources:
- Ilioinguinal nerve (L1): Anterior aspects.
- Genital branch of the genitofemoral nerve (L1, L2): Anterolateral aspects.
- Perineal branches of the pudendal nerve (S2-S4): Posterior aspects.
- Posterior scrotal nerves (from the perineal nerve, a branch of the pudendal nerve): Posterior aspects.
- Perineal branches of the posterior cutaneous nerve of the thigh (S1-S3): Inferior aspects (as mentioned in the original text, but typically it's the posterior scrotal nerves that are the primary supply to the posterior scrotum).
Lymphatic Drainage of the Scrotum:
- Lymph from the skin and superficial fascia of the scrotum drains primarily to the superficial inguinal lymph nodes. (This is distinct from the lymphatic drainage of the testes, which goes to lumbar/aortic lymph nodes).
Function of the Scrotum:
- Temperature Regulation: The primary and most critical function of the scrotum is to maintain the testes at a temperature approximately 2-3°C (or about 2-5°C) below core body temperature. This cooler temperature is essential for viable spermatogenesis (sperm formation) and storage.
- It achieves this through:
- Its position outside the abdominal cavity.
- The dartos muscle (wrinkling/smoothing the skin).
- The cremaster muscle (raising/lowering the testes).
- The pampiniform plexus (a network of veins surrounding the testicular artery in the spermatic cord that acts as a countercurrent heat exchanger, cooling arterial blood entering the testes).
- It achieves this through:
- Protection: Provides a protective sac for the testes and epididymides.
The Testis (Testes, plural)
The testes are the male gonads, the primary reproductive organs in males. They are responsible for two main functions: spermatogenesis (production of male gametes, sperm) and steroidogenesis (production of male sex hormones, primarily testosterone).
General Characteristics:
- Location: Located within the scrotum, which is crucial for their temperature regulation.
- Shape and Size: Ovoid in shape, typically about 4-5 cm long, 2.5 cm wide, and 3 cm thick.
- Consistency and Mobility: They are firm, smooth, and somewhat mobile within the scrotum.
- Development and Descent:
- The testes develop in the posterior abdominal wall, near the kidneys (at approximately the L1 vertebral level).
- They typically descend into the scrotum during the 7th month of fetal development, guided by the gubernaculum.
- This descent can occur at birth or within a few hours or days after birth. Failure of descent (cryptorchidism) is a significant clinical condition.
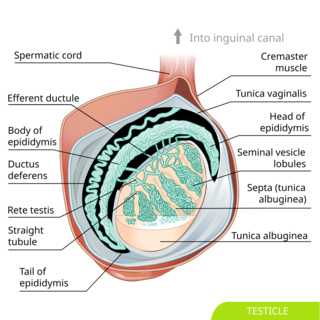
Gross Anatomy of the Testis:
Each testis is covered by a series of layers:
- Tunica Vaginalis:
- A serous sac derived from the peritoneum during testicular descent.
- It has two layers: a parietal layer (lining the inner surface of the scrotum) and a visceral layer (covering the outer surface of the testis, epididymis, and lower part of the spermatic cord).
- A small amount of serous fluid between these layers allows for smooth movement.
- Clinical Relevance: Excess fluid in this space leads to a hydrocele.
- Tunica Albuginea:
- A dense, white, fibrous connective tissue capsule that directly surrounds the testis beneath the visceral tunica vaginalis.
- At the posterior border of the testis, the tunica albuginea thickens and projects into the gland as the mediastinum testis.
- From the mediastinum testis, numerous fibrous septa extend inward, dividing the testis into approximately 250-300 conical compartments called testicular lobules.
- Testicular Lobules:
- Each lobule contains 1-4 highly convoluted seminiferous tubules, which are the sites of spermatogenesis ("sperm factories").
- Between the seminiferous tubules within the lobules are clusters of interstitial cells (Leydig cells).
Duct System within the Testis:
- Seminiferous Tubules:
- The primary site of sperm production.
- They are highly coiled and convoluted.
- At the apex of each lobule, they straighten to become the tubuli recti (straight tubules).
- Rete Testis:
- A network of anastomosing channels located within the mediastinum testis.
- The tubuli recti drain into the rete testis.
- From the rete testis, about 12-20 efferent ductules emerge.
- Efferent Ductules:
- These small, ciliated ducts pierce the tunica albuginea and connect the rete testis to the epididymis.
- They play a role in sperm transport and fluid absorption.
Histology of the Testis:
The cellular composition of the testis is specialized for its dual functions:
1. Seminiferous Tubules
Lined by a complex stratified epithelium known as the germinal epithelium. This epithelium contains two main cell types:
- Spermatogenic Cells: These cells are in various stages of spermatogenesis (spermatogonia, primary spermatocytes, secondary spermatocytes, spermatids, and finally spermatozoa/sperm, which are the male gametes). They are arranged in an orderly fashion from the basement membrane towards the lumen.
- Sertoli Cells (Sustentacular Cells): Tall, columnar cells that extend from the basement membrane to the lumen of the tubule. They are sometimes called "nurse cells" due to their critical support functions:
- Support and Nourish: Provide physical and nutritional support for developing sperm.
- Blood-Testis Barrier: Form tight junctions with adjacent Sertoli cells, creating a blood-testis barrier that isolates the developing sperm from the immune system.
- Phagocytosis: Phagocytose residual bodies from maturing spermatids.
- Hormone Production: Produce inhibin (regulates FSH release) and androgen-binding protein (maintains high local testosterone levels).
2. Interstitial Cells of Leydig
- Located in the connective tissue (interstitium) between the seminiferous tubules.
- Function: Produce and secrete male sex hormones, primarily testosterone, under the influence of Luteinizing Hormone (LH) from the anterior pituitary. Testosterone is crucial for spermatogenesis, development of secondary sexual characteristics, and maintaining male reproductive organs.
Blood Supply of the Testis:
- Arterial Supply:
- The main supply is the testicular artery (gonadal artery).
- It arises directly from the abdominal aorta at approximately the L1-L2 vertebral level.
- It descends in the retroperitoneum, passes through the deep inguinal ring, and becomes a component of the spermatic cord.
- Venous Drainage:
- The veins form a network around the testicular artery called the pampiniform plexus within the spermatic cord.
- The pampiniform plexus serves as a countercurrent heat exchange system, cooling arterial blood before it reaches the testis.
- The pampiniform plexus eventually consolidates into the single testicular vein on each side.
- Right Testicular Vein: Drains directly into the inferior vena cava (IVC).
- Left Testicular Vein: Drains into the left renal vein (which then drains into the IVC).
- Clinical Relevance: The different drainage patterns explain why varicoceles (dilated veins of the pampiniform plexus) are more common on the left side due to the longer course and perpendicular entry into the left renal vein, which can increase hydrostatic pressure.
Lymphatic Drainage of the Testis:
- Lymph from the testes follows the testicular vessels back along their developmental path.
- It drains to the para-aortic (lumbar) lymph nodes at the L1-L2 vertebral level, near the origin of the testicular arteries.
- Clinical Relevance: This is extremely important in the metastasis of testicular cancer, as these are the primary nodes to check. This is distinct from the lymphatic drainage of the scrotum, which drains to inguinal nodes.
Innervation of the Testis:
- Autonomic Nerves: The testis receives both sympathetic and parasympathetic innervation from the testicular plexus, which accompanies the testicular artery.
- Sympathetic: From T10-T11 spinal cord segments (via aorticorenal ganglion).
- Parasympathetic: From the vagus nerve (CN X).
- Sensory: Afferent fibers accompany the sympathetic nerves back to T10-T11, which explains referred pain from the testis to the lower abdominal/groin region.
The Spermatic Cord
The spermatic cord is a complex structure that suspends the testis in the scrotum and provides its neurovascular supply and a conduit for sperm transport. It begins at the deep inguinal ring, passes through the inguinal canal, and ends at the posterior border of the testis.
Contents of the Spermatic Cord:
- Ductus Deferens (Vas Deferens): A thick-walled muscular tube that transports sperm from the epididymis to the ejaculatory duct.
- Testicular Artery: The primary arterial supply to the testis.
- Pampiniform Plexus of Veins: A network of veins that drains the testis and epididymis, and provides a countercurrent heat exchange mechanism.
- Lymphatic Vessels: Draining lymph from the testis and epididymis to the para-aortic lymph nodes.
- Autonomic Nerves: Accompanying the testicular artery and ductus deferens.
- Artery to the Ductus Deferens: Typically a branch of the superior vesical artery.
- Cremasteric Artery: A branch of the inferior epigastric artery, supplying the cremaster muscle and the coverings of the spermatic cord.
- Genital Branch of the Genitofemoral Nerve (L1, L2): Supplies the cremaster muscle (motor) and sensory innervation to the scrotal skin.
- Remains of the Processus Vaginalis: A peritoneal diverticulum that typically obliterates after testicular descent. Its persistence can lead to congenital inguinal hernias or hydroceles.
Coverings of the Spermatic Cord:
These are continuous with the layers of the anterior abdominal wall that are traversed by the descending testis:
- External Spermatic Fascia: Derived from the external oblique aponeurosis.
- Cremasteric Fascia and Muscle: Derived from the internal oblique muscle.
- Internal Spermatic Fascia: Derived from the fascia transversalis.
Clinical Correlates (Genitourinary System - General & Male External Genitalia)
The developmental complexities of the genitourinary system can lead to various congenital anomalies.
Hypospadias
- A congenital condition in males where the urethral opening (meatus) is located on the underside (ventral surface) of the penis, rather than at the tip of the glans.
- Can range from mild (meatus near the glans) to severe (meatus in the perineum).
- Often associated with chordee (a downward curvature of the penis) and an abnormally formed prepuce (dorsal hood).
- Requires surgical correction.
Epispadias
- A rare congenital anomaly where the urethral opening is located on the upper side (dorsal surface) of the penis.
- Often associated with bladder exstrophy (see below) due to a more severe developmental defect.
- More difficult to repair than hypospadias and often leads to urinary incontinence.
Exstrophy of the Bladder (Bladder Exstrophy)
- A severe congenital anomaly where the bladder is open to the outside of the body through a defect in the abdominal wall and anterior bladder wall.
- The inner surface of the bladder is exposed.
- Often associated with other anomalies, including epispadias, widely separated pubic bones, and malformed genitalia.
- Requires complex surgical repair shortly after birth.
Micropenis
- A penis that is abnormally small (more than 2.5 standard deviations below the mean for age) but otherwise normally formed.
- Often caused by hormonal deficiencies (e.g., testosterone deficiency) during fetal development.
- May be treated with hormonal therapy.
Bifid Penis (Diphallia) or Double Penis
- A rare congenital malformation where there is a duplication of the penis.
- The degree of duplication can vary, from a partially bifid glans to two completely separate penises.
- Often associated with other genitourinary and gastrointestinal anomalies.
- Requires surgical correction.
Additional Clinical Correlates for the Testis:
Cryptorchidism (Undescended Testis)
- Failure of one or both testes to descend into the scrotum.
- If not corrected, it carries increased risks of infertility (due to higher abdominal temperature), testicular cancer, and inguinal hernia.
- Often requires surgical intervention (orchidopexy) to bring the testis into the scrotum.
- A surgical emergency where the spermatic cord twists, cutting off the blood supply to the testis.
- Causes sudden, severe scrotal pain, swelling, and tenderness.
- If not treated promptly (within hours), it can lead to testicular ischemia and necrosis, requiring removal of the testis.
Epididymitis/Orchitis
- Epididymitis: Inflammation of the epididymis, often caused by bacterial infection.
- Orchitis: Inflammation of the testis, often due to viral infection (e.g., mumps).
- Both cause scrotal pain and swelling.
Testicular Cancer
- Relatively rare but the most common cancer in young men (ages 15-35).
- Often presents as a painless lump or swelling in the testis.
- Highly curable if detected early.
- Risk factors include cryptorchidism.
Hydrocele
- A collection of fluid in the tunica vaginalis, causing swelling of the scrotum.
- Can be congenital (due to a patent processus vaginalis) or acquired.
- Usually benign but can cause discomfort.
Varicocele
- Dilated, tortuous veins of the pampiniform plexus within the spermatic cord, often described as feeling like a "bag of worms."
- More common on the left side.
- Can cause discomfort and is a common cause of male infertility (due to impaired thermoregulation and blood flow).
Disorders of Male External Genitalia and Related Conditions
This section outlines various congenital anomalies, acquired conditions, and diseases affecting the male external genitalia (penis and scrotum) and associated structures like the testis and spermatic cord.
Congenital Anomalies:
Congenital conditions are present at birth and result from developmental errors during embryogenesis.
1. Bifid Penis (Diphallia):
- Description: A rare congenital condition characterized by the duplication of the penis. The degree of duplication can range from a partially bifid glans to two completely separate penises, each with its own urethra.
- Etiology: Results from incomplete or abnormal fusion of the genital tubercles during embryonic development.
- Associated Anomalies: Often coexists with other genitourinary defects (e.g., bladder exstrophy, duplicated bladder or urethra) and sometimes gastrointestinal anomalies.
- Clinical Significance: Depending on the severity, it can lead to difficulties with urination, sexual function, and psychological distress.
- Treatment: Surgical correction is usually required to reconstruct a single functional penis, or in some cases, remove one of the duplicated structures.
2. Hypospadias:
- Description: A common congenital condition where the urethral opening (meatus) is located on the ventral (underside) aspect of the penis, rather than at the tip of the glans. The location can vary from near the glans (glandular or coronal) to the shaft (penile) or even the scrotum or perineum (penoscrotal or perineal).
- Associated Features: Often accompanied by:
- Chordee: A downward curvature of the penis, especially during erection, caused by fibrous tissue replacing normal skin/fascia on the ventral side.
- Dorsal Hood: An incomplete foreskin that forms a "hood" on the dorsal aspect of the glans, leaving the ventral glans uncovered.
- Clinical Significance: Can lead to problems with:
- Urination: Difficulty directing the urine stream, requiring sitting to urinate.
- Sexual Function: Chordee can make intercourse difficult or painful.
- Fertility: In severe cases, abnormal sperm deposition can affect fertility.
- Treatment: Surgical correction (urethroplasty) is typically performed between 6-18 months of age to reposition the meatus and correct chordee. Circumcision is contraindicated in infants with hypospadias, as the foreskin may be needed for surgical reconstruction.
3. Epispadias:
- Description: A rare congenital anomaly where the urethral opening is located on the dorsal (upper) aspect of the penis. It results from a more severe failure of fusion of the anterior urethral plate.
- Associated Features: Often associated with a more extensive developmental defect of the lower abdominal wall and bladder, most notably bladder exstrophy.
- Clinical Significance: Patients almost always experience urinary incontinence due to a defect in the urinary sphincter mechanism, in addition to the cosmetic and functional issues of the penile malformation.
- Treatment: Complex surgical reconstruction, often involving multiple stages, aimed at creating a functional urethra, correcting penile curvature, and achieving continence.
4. Micropenis:
- Description: Refers to a penis that is abnormally small (length significantly below the average for age, usually more than 2.5 standard deviations below the mean), but otherwise structurally normal.
- Etiology: Typically due to insufficient androgen (testosterone) production or action during fetal development, often related to hypothalamic-pituitary-gonadal axis dysfunction.
- Clinical Significance: Primarily concerns regarding sexual function, body image, and potential fertility issues.
- Treatment:
- Hormonal Therapy: Testosterone supplementation, particularly if initiated in infancy or early childhood, can promote penile growth.
- Plastic Surgery: While the original text mentions "plastic surgery" as treatment, surgical lengthening procedures for true micropenis are complex, often with limited cosmetic and functional success, and are generally reserved for specific cases after hormonal therapy has been optimized. The primary approach remains hormonal.
5. Macropenis:
- Description: An abnormally large penis, significantly above the average size for age.
- Etiology: Can be caused by excessive androgen production during fetal development or childhood (e.g., congenital adrenal hyperplasia) or certain genetic conditions.
- Clinical Significance: While sometimes perceived as desirable, extreme macropenis can lead to discomfort, difficulty with clothing, and sometimes psychological issues. Rarely requires medical intervention for reduction.
6. Disorders of Sexual Development (DSD) / Intersex Conditions (formerly Hermaphroditism):
- Description: These are congenital conditions where there is a discrepancy between the external genitalia and the internal reproductive organs or chromosomal sex. The term "hermaphroditism" is now considered outdated and replaced by DSD.
- Types: Include conditions like 46,XX DSD (XX karyotype with virilized external genitalia), 46,XY DSD (XY karyotype with undervirilized or ambiguous external genitalia), and sex chromosome DSDs (e.g., Klinefelter syndrome, Turner syndrome variants).
- Clinical Significance: Presentation can range from ambiguous genitalia at birth to delayed or absent puberty. Management involves complex medical, genetic, psychological, and surgical considerations, often by a multidisciplinary team, with careful consideration of gender identity.
Acquired Conditions & Diseases:
These conditions develop after birth due to various factors like infection, trauma, lifestyle, or aging.
1. Testicular Torsion:
- Description: A surgical emergency characterized by the twisting of the spermatic cord, which suspends the testis. This twisting causes kinking of the testicular artery and veins, obstructing blood flow to the testis.
- Etiology: Can occur spontaneously, after trauma, or with vigorous activity (including sexual activity or exercise), though often no specific trigger is identified. It's more common in adolescents but can occur at any age. "Bell-clapper deformity" (where the testis has a higher attachment and can swing freely) is a predisposing anatomical factor.
- Clinical Significance:
- Sudden, severe, unilateral scrotal pain: Often radiating to the groin or lower abdomen.
- Swelling and tenderness: Of the affected testis, which may be elevated and in an abnormal horizontal lie ("transverse lie").
- Nausea and vomiting: Common systemic symptoms.
- Ischemic Necrosis: If blood flow is not restored promptly (typically within 4-6 hours), the testis will suffer irreversible damage and become necrotic, requiring surgical removal (orchiectomy).
- Treatment: Urgent surgical exploration (orchiopexy to untwist and fix the testis, and often fix the contralateral testis to prevent future torsion).
2. Phimosis & Paraphimosis:
Phimosis
- Description: A condition where the foreskin (prepuce) of the penis cannot be fully retracted over the glans.
- Types:
- Physiologic phimosis: Normal in infants and young boys due to adhesions, which usually resolve with age.
- Pathologic phimosis (severe phimosis): Results from scarring (often due to recurrent infections or forceful retraction) that makes retraction difficult or impossible.
- Clinical Significance:
- Difficulty with hygiene: Can lead to accumulation of smegma under the foreskin, predisposing to infection (balanitis).
- Obstruction of urine flow: In severe cases, can cause ballooning of the foreskin during urination or even difficulty voiding.
- Painful erections/intercourse: Due to stretching of the tight foreskin.
- Treatment: Steroid creams can sometimes soften the foreskin. If unsuccessful or severe, circumcision (surgical removal of the foreskin) is the definitive treatment.
Paraphimosis
- Description: A true medical emergency where a retracted foreskin becomes trapped behind the glans and cannot be returned to its normal position.
- Clinical Significance: The tight band of foreskin acts like a tourniquet, leading to venous and lymphatic engorgement, swelling of the glans, pain, and eventually arterial occlusion and necrosis of the glans.
- Treatment: Urgent manual reduction (squeezing the glans to reduce swelling and then pulling the foreskin forward). If unsuccessful, surgical incision of the constricted band is required, followed by emergency circumcision once swelling subsides.
3. Cancer of the Penis (Penile Carcinoma):
- Description: A rare malignancy, most commonly a squamous cell carcinoma, that affects the skin or tissues of the penis.
- Risk Factors:
- Uncircumcised males: Accumulation of smegma (a cheesy white substance composed of shed skin cells, oils, and moisture) under the foreskin is believed to be carcinogenic. Chronic inflammation and infections also contribute.
- Human Papillomavirus (HPV) infection: Especially high-risk types.
- Smoking: A significant independent risk factor.
- Poor hygiene.
- Presentation: Often begins as a painless lump, ulcer, or discoloration on the glans or foreskin.
- Clinical Significance: Can be disfiguring and lead to significant morbidity and mortality if not treated early.
- Treatment:
- Early Stages: Treatment aims for organ preservation. Options include topical chemotherapy, laser ablation, cryosurgery, radiation therapy, or wide local excision.
- Advanced Stages: May require partial or total penectomy (amputation), often with inguinal lymph node dissection, depending on the extent of the cancer.
4. Penile Warts (Condylomata Acuminata):
- Description: Benign epithelial growths on the penis (and other genital areas) caused by infection with the Human Papillomavirus (HPV), particularly low-risk types (e.g., HPV 6 and 11).
- Transmission: Primarily through sexual contact.
- Clinical Significance: Can be asymptomatic but may cause itching, irritation, or bleeding. While generally harmless, they are a sign of HPV infection, which can sometimes be associated with higher-risk types that cause cancer.
- Treatment: Aims to remove the visible warts and alleviate symptoms, but does not eradicate the underlying HPV infection. Treatment options include:
- Topical agents: Podophyllin (a cytotoxic agent, often used in a paint or gel), imiquimod (immune response modifier), trichloroacetic acid (TCA).
- Cryotherapy: Freezing the warts with liquid nitrogen.
- Electrocautery: Burning off the warts.
- Surgical excision: Cutting out the warts.
- Laser therapy: Vaporizing the warts with a laser.
5. Pubic Lice (Pthirus Pubis, "Crabs"):
- Description: Small insects that infest the pubic hair and other coarse body hair (e.g., armpits, eyelashes, beard).
- Transmission: Primarily through close physical (often sexual) contact, but can also spread via infested bedding or clothing.
- Clinical Significance: Cause intense itching in the affected areas, leading to skin irritation and sometimes secondary bacterial infections from scratching. Visible nits (lice eggs) or adult lice may be seen on the hair shafts.
- Treatment: Topical insecticides (e.g., permethrin, pyrethrins) applied to the affected areas. Decontaminating clothing and bedding is also important.

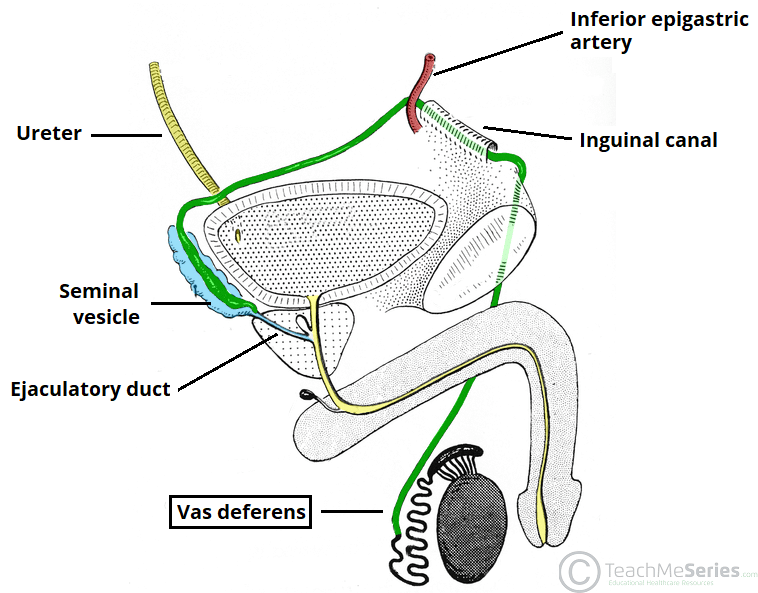
Male Internal Genitalia
The male internal genitalia comprise the reproductive organs located within the pelvic cavity, extending into the perineum. They are responsible for the maturation, transport, and nourishment of sperm, as well as the production of seminal fluid, which, along with sperm, constitutes semen. The main components are the seminal vesicles, ejaculatory ducts, vas deferens, prostate gland, and bulbourethral glands (which were not in your list, but are crucial for completeness). The male urethra, while serving both urinary and reproductive functions, is functionally an integral part of the internal genitalia during ejaculation.
1. Seminal Vesicles (Glands):
- Description: Two elongated, coiled, and lobulated glandular organs. They are not simply "vesicles" (sacs) but rather highly convoluted tubes that appear sac-like.
- Location: Situated on the posterior surface of the urinary bladder, superior to the prostate gland, and lateral to the ampulla of the vas deferens.
- Anatomical Relations:
- Anteriorly: Posterior wall of the urinary bladder.
- Posteriorly: Rectum.
- Medially: Ampulla of the vas deferens.
- Superiorly: Peritoneum (some parts).
- Inferiorly: Prostate gland.
- Duct System: Each seminal vesicle has an excretory duct that joins the corresponding ampulla of the vas deferens to form the ejaculatory duct.
- Blood Supply:
- Inferior Vesical Artery: A branch of the internal iliac artery.
- Middle Rectal Artery: Also a branch of the internal iliac artery.
- Some anastomoses with the superior vesical artery.
- Innervation: Primarily sympathetic from the inferior hypogastric plexus.
- Function:
- Produce a viscous, slightly alkaline, yellowish fluid that constitutes approximately 60-70% of the semen volume.
- This fluid is rich in:
- Fructose: The primary energy source for sperm motility.
- Prostaglandins: Potent local hormones. They are believed to stimulate smooth muscle contractions in the female reproductive tract (uterus and fallopian tubes) to aid sperm transport, and may also suppress female immune response to sperm.
- Coagulation Proteins (Fibrinogen-like proteins): These proteins cause the semen to coagulate shortly after ejaculation, forming a temporary plug in the vagina, which may prevent sperm reflux.
- Ascorbic Acid (Vitamin C), Inositol, Citric Acid: The role of citric acid here is not fully understood, but it contributes to the overall chemical composition and pH.
2. Ejaculatory Ducts:
- Description: Short, straight ducts (about 2 cm long) formed by the union of the duct of the seminal vesicle and the ampulla of the vas deferens.
- Location: They begin near the superior pole of the prostate gland and pass obliquely downwards and forwards through the substance of the prostate.
- Termination: They open into the prostatic urethra on either side of the utricle prostaticus (a small blind-ended pouch on the seminal colliculus).
- Function: Transport sperm and seminal vesicle fluid into the prostatic urethra during ejaculation.
3. Vas Deferens (Ductus Deferens):
- Description: Two thick-walled, muscular tubes that are continuations of the epididymis. Each is approximately 45 cm long.
- Course:
- Ascends from the tail of the epididymis, passes superiorly as a component of the spermatic cord.
- Travels through the inguinal canal.
- Crosses the external iliac vessels and turns medially into the pelvic cavity.
- Descends on the lateral pelvic wall, then turns medially to lie on the posterior surface of the bladder, medial to the seminal vesicle.
- Before joining the seminal vesicle duct, it widens to form the ampulla of the vas deferens, which stores sperm.
- Structure: Has a narrow lumen surrounded by a thick muscular wall (three layers of smooth muscle: inner longitudinal, middle circular, outer longitudinal), which is responsible for the powerful peristaltic contractions that rapidly propel sperm during ejaculation.
- Blood Supply: Artery to the vas deferens, usually a branch of the superior vesical artery.
- Function: Rapidly propels mature sperm from the epididymis and ampulla towards the ejaculatory ducts during ejaculation.
4. Prostate Gland:
- Description: A single, firm, fibromuscular and glandular organ of the male reproductive system. It is the largest accessory gland of the male reproductive system.
- Shape: Commonly described as cone-shaped or an inverted pyramid, with its base superior and apex inferior.
- Location: Surrounds the initial part of the urethra (prostatic urethra) and lies inferior to the urinary bladder.
- Anatomical Relations:
- Superiorly (Base): Continuous with the neck of the urinary bladder.
- Inferiorly (Apex): Rests on the urogenital diaphragm (perineal membrane and external urethral sphincter).
- Anteriorly: Separated from the pubic symphysis by the retropubic space (space of Retzius), which contains loose areolar tissue and fat.
- Posteriorly: Related to the anterior wall of the rectum, separated by the rectovesical septum (fascia of Denonvilliers). This close proximity allows for digital rectal examination (DRE) of the prostate.
- Laterally: Related to the levator ani muscles (puboprostatic ligaments) and its own fibrous capsule.
- Internal Structure (Zones/Lobes):
- Traditionally described as having five lobes (anterior, posterior, median, and two lateral lobes), this anatomical division is largely superseded by the zonal anatomy which is more relevant clinically.
- Zonal Anatomy:
- Peripheral Zone (70% of glandular tissue): Posterior and lateral parts, most common site for prostate cancer.
- Central Zone (25%): Surrounds the ejaculatory ducts, resistant to carcinoma and inflammation.
- Transitional Zone (5%): Surrounds the urethra, site of origin for benign prostatic hyperplasia (BPH).
- Anterior Fibromuscular Stroma: Non-glandular part.
- Blood Supply:
- Branches from the inferior vesical artery (main supply).
- Branches from the middle rectal artery and internal pudendal artery also contribute.
- Innervation: Primarily sympathetic and parasympathetic nerves from the inferior hypogastric plexus.
- Function:
- Produces a thin, milky, slightly acidic fluid that constitutes about 20-30% of the semen volume.
- This fluid contains:
- Citric Acid: Serves as a nutrient for sperm.
- Acid Phosphatase: Function not fully understood but used as a marker for prostatic tissue.
- Prostate-Specific Antigen (PSA): A proteolytic enzyme that acts as a fibrinolysin, breaking down the coagulation proteins from the seminal vesicles. This liquefaction of the seminal coagulum occurs about 15-30 minutes after ejaculation, allowing sperm to become motile and escape the semen clot.
- Seminalplasmin: An antibiotic that may prevent urinary tract infections in males.
- The slightly acidic nature of prostatic fluid, combined with the alkaline fluid from seminal vesicles, results in a final semen pH of 7.2-7.8, which neutralizes the acidic environment of the vagina, optimizing sperm motility and survival.
5. Bulbourethral Glands (Cowper's Glands):
- Description: Two small (pea-sized) exocrine glands.
- Location: Located within the deep perineal pouch (urogenital diaphragm), lateral to the membranous urethra. Their ducts are long and open into the spongy (penile) urethra.
- Function:
- Produce a clear, alkaline mucus-like fluid (pre-ejaculate) that is released during sexual arousal, before ejaculation.
- Lubrication: Lubricates the glans penis for intercourse.
- Neutralization: Neutralizes any acidic urine residue in the urethra, protecting sperm.
6. Male Urethra:
The male urethra serves as a common passageway for both urine and semen. It is approximately 20 cm (8 inches) long and is divided into three main parts:
- Preprostatic Urethra:
- Description: Shortest, about 1 cm. Extends from the internal urethral orifice of the bladder to the base of the prostate. Surrounded by the internal urethral sphincter (smooth muscle).
- Prostatic Urethra:
- Description: About 3 cm long. It traverses through the prostate gland.
- Characteristic: It is the widest and most dilatable part of the entire male urethra.
- Key Features:
- Urethral Crest: A median longitudinal ridge on its posterior wall.
- Seminal Colliculus (Verumontanum): An elevation on the urethral crest, containing the opening of the prostatic utricle (a blind-ended remnant of the paramesonephric duct) and the openings of the ejaculatory ducts.
- Openings of the prostatic glands.
- Membranous Urethra:
- Description: The shortest (about 1.25 cm long) and least dilatable part of the male urethra.
- Location: Passes through the urogenital diaphragm (deep perineal pouch).
- Characteristic: Surrounded by the external urethral sphincter (voluntary striated muscle), which provides voluntary control over urination. It is also the most vulnerable part to injury.
- Spongy (Penile) Urethra:
- Description: The longest part (about 15 cm long).
- Location: Traverses the entire length of the corpus spongiosum of the penis.
- Dilatations: Contains two dilatations:
- Bulb of the Urethra: In the bulb of the penis.
- Navicular Fossa: A terminal expansion within the glans penis.
- External Urethral Meatus: The external opening of the urethra at the tip of the glans penis. This is the narrowest part of the entire male urethra and thus the most common site for strictures and difficulty passing instruments.
- Openings: Receives the ducts of the bulbourethral glands.
Disorders of the Urethra
While hypospadias and epispadias were covered under congenital disorders of the external genitalia, their direct impact on the urethra warrants their mention here.
- Hypospadias: (As described previously) The urethral opening is on the ventral surface of the penis. This is a malformation of the spongy (penile) urethra or its meatus.
- Epispadias: (As described previously) The urethral opening is on the dorsal surface of the penis. This is also a malformation of the penile urethra.
Other Urethral Disorders:
Urethral Stricture
- Description: Narrowing of the urethra due to scarring.
- Etiology: Most commonly caused by trauma (e.g., catheterization, pelvic fracture), infection (e.g., gonorrhea), or inflammation.
- Clinical Significance: Causes obstruction to urine flow, leading to weak stream, straining, incomplete emptying, and increased risk of urinary tract infections.
- Treatment: Dilation, internal urethrotomy, or open surgical repair (urethroplasty).
Urethritis
- Description: Inflammation of the urethra.
- Etiology: Most commonly caused by sexually transmitted infections (e.g., Chlamydia trachomatis, Neisseria gonorrhoeae) or bacterial infections.
- Clinical Significance: Causes dysuria (painful urination), urgency, frequency, and urethral discharge.
Urethral Diverticulum
- Description: An outpouching or sac-like projection from the urethral wall.
- Etiology: Can be congenital or acquired (e.g., due to trauma, infection).
- Clinical Significance: Can cause recurrent UTIs, post-void dribbling, dysuria, or pain.
Male Reproductive Quiz
Systems Anatomy
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Male Reproductive Quiz
Systems Anatomy
Preparing questions...
Exam Completed!
See your performance breakdown below.