Growth Hormone Physiology
GROWTH HORMONE (SOMATOTROPHIN)
Growth Hormone (GH), also known as Somatotrophin, is a crucial hormone responsible for the growth and development of the body's tissues.
- Structure: It is a relatively small protein molecule, composed of a single chain of 191 amino acids, with a molecular weight of approximately 22,005.
- Half-Life: In the bloodstream, GH has a relatively short half-life of less than 20 minutes. This is because it binds only weakly to plasma proteins, allowing for rapid turnover.
- Primary Function: GH causes the growth of almost all tissues of the body that are capable of growing.
- It promotes an increase in the sizes of cells (hypertrophy) and an increase in mitosis (cell division), leading to the development of greater numbers of cells (hyperplasia).
- It also contributes to the specific differentiation of certain types of cells, such as bone growth cells (chondrocytes and osteoblasts) and early muscle cells (myoblasts).
- Mechanism of Action: In contrast to many other hormones that act through specific target glands (e.g., TSH acting on the thyroid), GH is unique because it does not function through a single target gland. Instead, it exerts its effects directly on all or almost all tissues of the body, acting as a widespread metabolic hormone.
ROLE OF HYPOTHALAMUS IN SECRETION OF GROWTH HORMONE
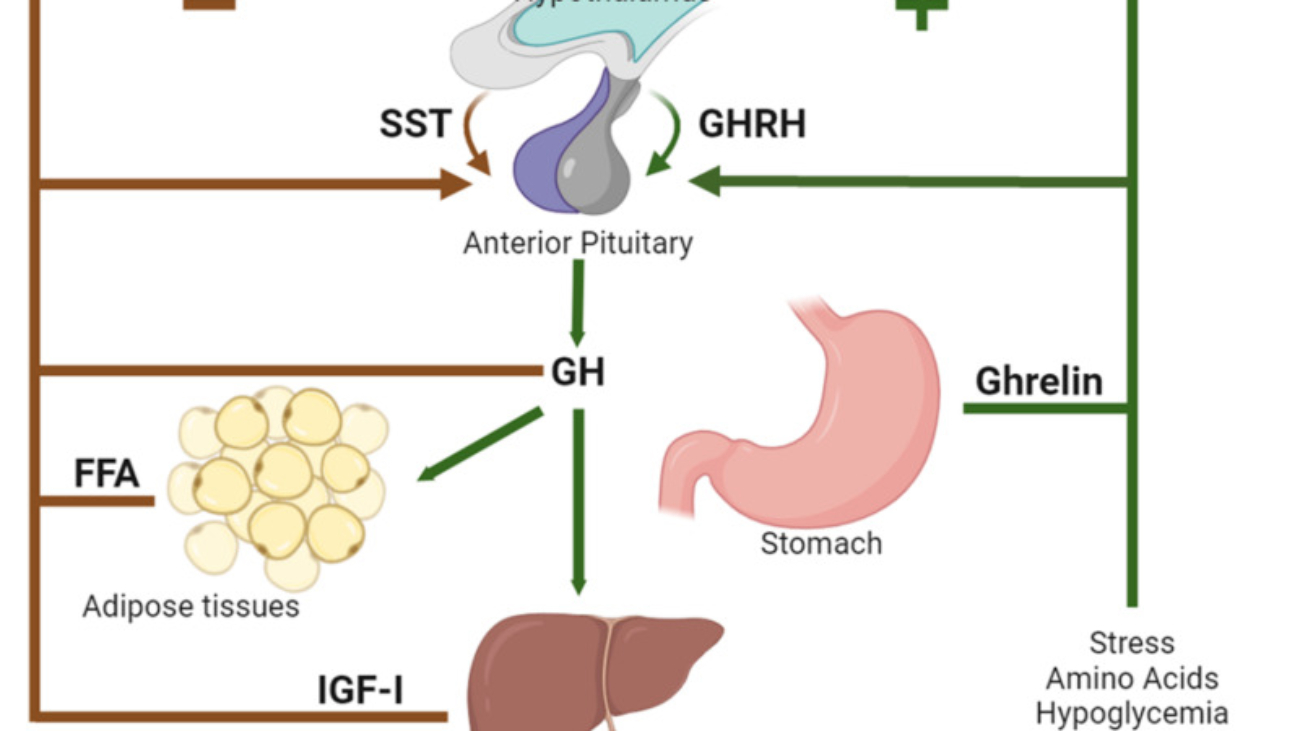
The secretion of Growth Hormone from the anterior pituitary gland is meticulously controlled by the hypothalamus through a dual regulatory system involving both stimulating and inhibiting hormones.
- Growth Hormone-Releasing Hormone (GHRH):
- The hypothalamus secretes Growth Hormone-Releasing Hormone (GHRH).
- GHRH is a peptide hormone that travels through the hypophyseal portal system to the anterior pituitary gland.
- Upon reaching the anterior pituitary, GHRH acts on the somatotrophs (GH-secreting cells) to stimulate the release of Growth Hormone.
- Growth Hormone-Inhibitory Hormone (GHIH) / Somatostatin:
- When growth hormone levels in the blood rise above a certain normal threshold, or in response to other physiological cues, the hypothalamus releases Somatostatin, also known as Growth Hormone-Inhibitory Hormone (GHIH).
- Somatostatin also travels to the anterior pituitary via the portal system.
- There, it acts on the somatotrophs to inhibit the release of Growth Hormone. This provides a crucial negative feedback mechanism to prevent excessive GH secretion.
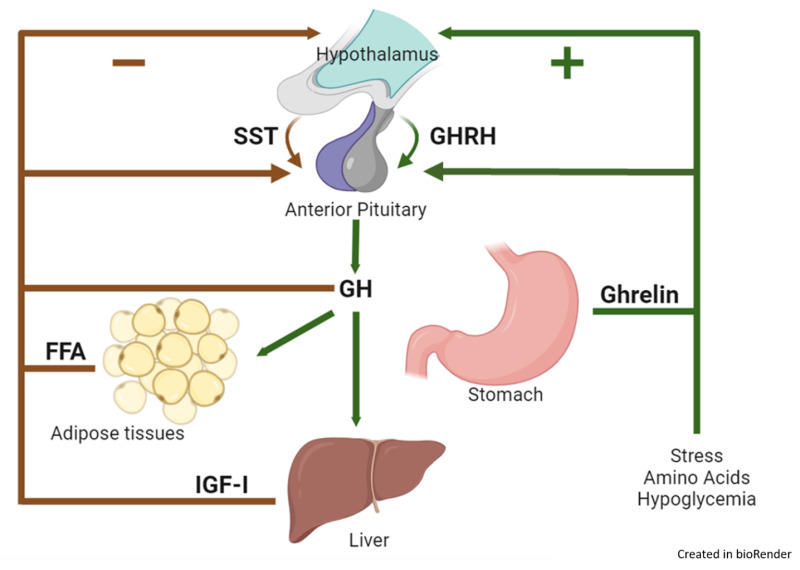
REGULATION OF GROWTH HORMONE SECRETION: FACTORS THAT STIMULATE OR INHIBIT
The secretion of Growth Hormone is complex and pulsatile, influenced by a variety of physiological, metabolic, and hormonal factors, operating through the hypothalamic GHRH and GHIH system.
Factors That Stimulate Growth Hormone Secretion:
These factors generally indicate a need for energy mobilization, tissue repair, or active growth.
- Decreased Blood Glucose (Hypoglycemia): A fall in blood sugar is a potent stimulus for GH release, helping to mobilize glucose from the liver.
- Decreased Blood Free Fatty Acids: Low levels of free fatty acids also stimulate GH secretion, as GH promotes fat breakdown.
- Starvation or Fasting, Protein Deficiency: These states signal a need for metabolic adaptation, with GH promoting protein conservation and fat utilization.
- Trauma, Stress, Excitement: Acute stress (physical or psychological) can trigger GH release, potentially aiding in recovery and energy mobilization.
- Exercise: Physical activity is a strong stimulus for GH secretion, contributing to muscle repair and growth.
- Hormones (Testosterone, Estrogen): Sex hormones, particularly during puberty, contribute to growth spurts and stimulate GH secretion.
- Deep Sleep (Stages II and IV): The majority of daily GH secretion occurs in bursts during the early stages of deep sleep, highlighting its role in growth and repair.
- Growth Hormone-Releasing Hormone (GHRH): As mentioned, this hypothalamic hormone is the primary physiological stimulator of GH release.
Factors That Inhibit Growth Hormone Secretion:
These factors typically signal sufficient energy stores or act as part of a negative feedback loop to prevent overproduction.
- Increased Blood Glucose (Hyperglycemia): High blood sugar levels inhibit GH release, as there is no immediate need to mobilize more glucose.
- Increased Blood Free Fatty Acids: Abundant free fatty acids indicate sufficient energy stores, suppressing GH secretion.
- Aging: As individuals age, basal and stimulated GH secretion generally decline, contributing to some of the metabolic changes associated with aging.
- Obesity: Obese individuals often exhibit lower GH secretion, which may contribute to their metabolic profile.
- Growth Hormone Inhibitory Hormone (GHIH) / Somatostatin: This hypothalamic hormone is the primary physiological inhibitor of GH release.
- Growth Hormone (Exogenous): Administration of exogenous GH provides a negative feedback signal to the hypothalamus and pituitary, inhibiting endogenous GH secretion.
- Somatomedins (Insulin-like Growth Factors - IGFs): These are peptide hormones, primarily IGF-1, produced largely by the liver in response to GH. IGFs act as a crucial negative feedback signal, directly inhibiting GH release from the pituitary and also stimulating GHIH release from the hypothalamus.
PHYSIOLOGICAL FUNCTIONS OF GROWTH HORMONE
As established, Growth Hormone (GH) is unique in that it does not function through a single target gland but rather exerts its pervasive effects directly on all or almost all tissues of the body that are capable of growing. Its diverse actions can be broadly categorized into:
- Promotes growth of many tissues: This is its most prominent and well-known function.
- Enhances fat utilization for energy: Shifting the body's fuel source.
- Decreases carbohydrate utilization: Conserving glucose, which has implications for blood sugar.
- Promotes protein deposition in tissues: Essential for tissue repair and growth.
GH PROMOTES PROTEIN DEPOSITION IN TISSUES
Growth Hormone is a potent anabolic hormone, meaning it promotes the building up of complex molecules from simpler ones, particularly proteins. While the precise mechanisms are still being fully elucidated, several key effects are known:
- Increased Nuclear Transcription of DNA to form RNA: GH stimulates the machinery within the cell nucleus to increase the transcription of DNA into various types of RNA (mRNA, tRNA, rRNA). This effectively ramps up the production of the templates and components necessary for protein synthesis.
- Enhancement of Amino Acid Transport Through the Cell Membranes: GH increases the active transport of amino acids from the extracellular fluid into the cells. This ensures a readily available supply of the building blocks for protein synthesis within the cells.
- Enhancement of RNA Translation to Cause Protein Synthesis by the Ribosomes: Once inside the cell, GH further promotes the translation of RNA into protein by the ribosomes. This means that not only are more protein blueprints being made, but they are also being utilized more efficiently to produce actual proteins.
- Decreased Catabolism of Protein and Amino Acids: Beyond promoting synthesis, GH also reduces the breakdown (catabolism) of existing proteins and amino acids. This dual action—increasing synthesis and decreasing breakdown—maximizes protein accumulation in tissues.
In summary: GH enhances almost all facets of amino acid uptake and protein synthesis by cells, while at the same time reducing the breakdown of proteins. This collective action leads to a positive nitrogen balance and overall tissue growth.
GH ENHANCES FAT UTILIZATION FOR ENERGY
One of the significant metabolic effects of GH is its ability to shift the body's primary fuel source away from carbohydrates and proteins and towards fats.
- Release of Fatty Acids from Adipose Tissue: GH directly stimulates adipose tissue (fat cells) to release fatty acids into the bloodstream. This significantly increases the concentration of free fatty acids in the body fluids.
- Enhanced Conversion to Acetyl Coenzyme A (Acetyl-CoA): These increased free fatty acids are then readily taken up by cells, where they are converted into acetyl coenzyme A (acetyl-CoA) through beta-oxidation. Acetyl-CoA is a central molecule in energy metabolism, entering the Krebs cycle for subsequent utilization to produce ATP (energy).
- Preference for Fat as Fuel: The consequence of this is that fat is used for energy in preference to the use of carbohydrates and proteins. This "protein-sparing" effect is crucial during periods of growth or when nutrient intake is limited, allowing proteins to be used for structural purposes and growth rather than for energy. This overall leads to an increase in lean body mass.
However, there are potential downsides:
- Ketosis: Sometimes, the mobilization of fat from adipose tissue can be so rapid and extensive that the liver processes large quantities of fatty acids into acetyl-CoA, exceeding the capacity of the Krebs cycle. This leads to the excessive formation and release of acetoacetic acid and other ketone bodies into the body fluids, potentially causing ketosis.
- Fatty Liver: This excessive mobilization of fat from the adipose tissue can also frequently cause a fatty liver, as the liver takes up large amounts of fatty acids, which can accumulate if their oxidation or export is not balanced.
GH DECREASES CARBOHYDRATE UTILIZATION
GH has significant effects on carbohydrate metabolism, generally leading to an increase in blood glucose levels and earning it the label of a "diabetogenic" hormone. Several effects contribute to this:
- Decreased Glucose Uptake in Tissues: GH reduces the uptake of glucose by peripheral tissues, such as skeletal muscle and fat cells. This means that these cells rely more on fatty acids for energy, leaving more glucose in the bloodstream.
- Increased Glucose Production by the Liver: GH stimulates the liver to increase its output of glucose, primarily through gluconeogenesis (synthesis of glucose from non-carbohydrate precursors) and possibly glycogenolysis (breakdown of glycogen).
- Increased Insulin Secretion: As a consequence of the rising blood glucose levels, the pancreas is stimulated to increase insulin secretion in an attempt to normalize blood sugar.
Mechanism: GH-induced "Insulin Resistance": Each of these changes results from GH-induced "insulin resistance," which attenuates the action of insulin. This means that cells become less responsive to insulin's signals to take up glucose. The overall outcome is an increased blood glucose concentration and a compensatory increase in insulin secretion. This mirrors the characteristics of Type 2 Diabetes Mellitus (T2DM), hence GH is said to have diabetogenic effects.
Unclear Mechanisms: The precise mechanisms of this insulin resistance are still unclear, but it may be attributed to increased blood concentrations of fatty acids. Elevated fatty acids can interfere with insulin signaling pathways in various tissues.
GH STIMULATES CARTILAGE AND BONE GROWTH
This is perhaps the most obvious and defining effect of Growth Hormone, particularly during childhood and adolescence. Several interconnected effects contribute to this:
- Increased Deposition of Protein by Chondrocytic and Osteogenic Cells: GH stimulates chondrocytes (cartilage cells) and osteogenic cells (bone-forming cells) to increase the synthesis and deposition of protein, especially collagen, which forms the organic matrix of cartilage and bone.
- Increased Rate of Reproduction of These Cells: GH promotes the proliferation (mitosis) of both chondrocytes and osteogenic cells. This leads to an increased number of cells actively involved in growth.
- Specific Effect of Converting Chondrocytes into Osteogenic Cells: GH also plays a role in the differentiation of chondrocytes into osteogenic cells. This conversion is crucial in the process of endochondral ossification, where cartilage is replaced by bone.
Two main mechanisms govern bone growth under GH influence:
- Stimulation of Long Bones to Grow in Length at the Epiphyseal Cartilages:
- In growing individuals, the long bones (e.g., femur, tibia) grow in length at the epiphyseal growth plates (cartilages), which are located at the ends of the bone, separating the epiphyses from the shaft.
- GH directly stimulates the chondrocytes within these growth plates to proliferate and enlarge, pushing the epiphyses further from the diaphysis. Subsequently, this cartilage is calcified and replaced by bone, leading to an increase in bone length. This process continues until the growth plates fuse after puberty, at which point longitudinal growth ceases.
- Stimulation of Osteoblasts (Deposition of New Bone):
- GH strongly stimulates osteoblasts, the cells responsible for depositing new bone. This leads to an increase in bone thickness and density, especially in membranous bones (e.g., skull bones, jawbone).
- In this context, osteoblast activity is stimulated to be greater than osteoclast activity, resulting in a net increase in bone mass.
GH AND THE ROLE OF SOMATOMEDINS (INSULIN-LIKE GROWTH FACTORS - IGFs)
While GH has direct effects on tissues, many of its growth-promoting actions are mediated indirectly through a group of small proteins called somatomedins, now more commonly known as Insulin-like Growth Factors (IGFs).
- Formation: GH causes the liver (and, to a much lesser extent, other tissues like cartilage) to form these somatomedins.
- Potent Effect on Growth: These somatomedins have a potent effect of increasing all aspects of bone growth and general tissue growth.
- "Insulin-like" Activity: Their effects on growth are very similar to those of insulin, hence the name Insulin-like Growth Factors.
- Types of Somatomedins: Four main types have been isolated, but somatomedin C is the most potent and clinically significant, often referred to as IGF-I.
- Somatomedin C (IGF-I):
- It has a molecular weight of about 7500.
- Its concentration in the plasma closely follows the rate of growth hormone secretion, making it a good clinical indicator of GH activity.
- Binding to Carrier Proteins: A critical feature of Somatomedin C is that it attaches strongly to specific carrier proteins in the blood. This binding has several important consequences:
- Prolonged Half-Life: It is released only slowly from the blood to the tissues, with a significantly longer half-life time of about 20 hours (compared to GH's <20 minutes).
- Sustained Growth-Promoting Effects: This greatly prolongs the growth-promoting effects of the pulsatile bursts of GH, providing a more continuous stimulus for tissue growth.
- Unclear Details: While the role of somatomedins/IGFs in mediating GH's actions is well-established, the precise details of their interaction and regulation are still areas of active research. It's understood that GH primarily stimulates IGF-I production, and IGF-I then carries out many of the anabolic and growth-promoting effects attributed to GH.
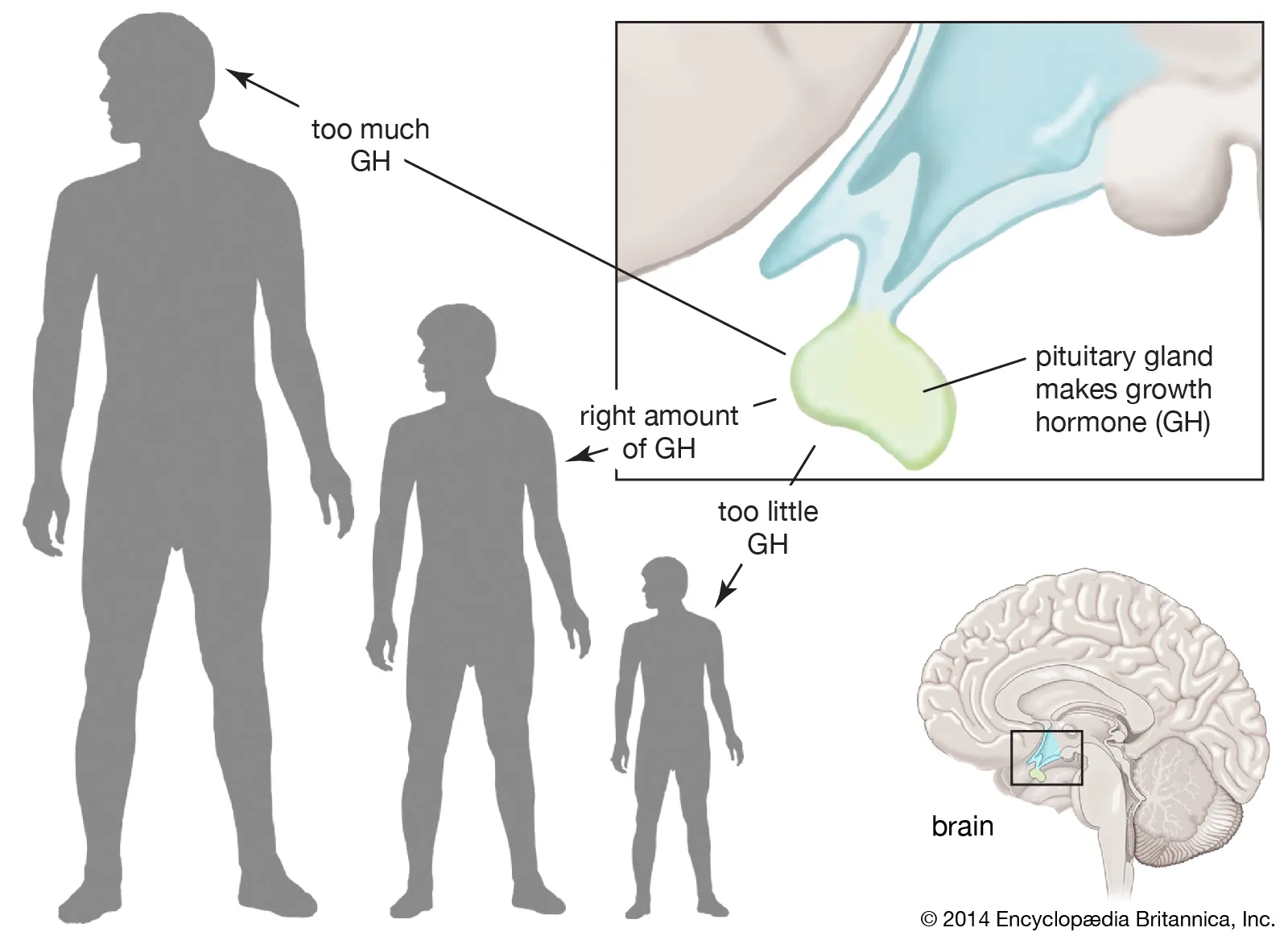
ABNORMALITIES OF GROWTH HORMONE SECRETION
Disruptions in the normal production or action of Growth Hormone (GH) can lead to a variety of clinical syndromes, ranging from stunted growth to excessive growth and metabolic disturbances. These abnormalities highlight the critical role GH plays throughout life. We will discuss four main conditions:
- Panhypopituitarism
- Dwarfism
- Gigantism
- Acromegaly
PANHYPOPITUITARISM
Panhypopituitarism refers to a condition characterized by decreased secretion of all or almost all the anterior pituitary hormones. This global deficiency impacts not just Growth Hormone but also TSH, ACTH, FSH, LH, and prolactin, leading to widespread endocrine dysfunction.
- Onset: This decrease in pituitary hormone secretion can be congenital (present from birth) or may develop suddenly or slowly at any time during life. The clinical manifestations will vary depending on the age of onset and the severity of the deficiency.
- Etiology (Causes):
- Pituitary Tumors: The most common cause in adults is a pituitary tumor (e.g., a non-functional adenoma) that grows and compresses or destroys the normal pituitary gland tissue.
- Craniopharyngiomas: In children, tumors like craniopharyngiomas can cause similar widespread pituitary dysfunction.
- Infarction: Ischemic necrosis of the pituitary, such as Sheehan's syndrome (postpartum pituitary necrosis due to severe hemorrhage and hypovolemia during childbirth), is another cause.
- Trauma, Radiation, Surgery: Head trauma, radiation therapy to the head, or surgery involving the pituitary region can also damage the gland.
- Infiltrative Diseases: Conditions like sarcoidosis or hemochromatosis can infiltrate and damage pituitary tissue.
- Genetic Mutations: Rare genetic mutations affecting pituitary development can lead to congenital panhypopituitarism.
- Clinical Manifestations (if GH is affected):
- Children: If panhypopituitarism occurs during childhood, it will lead to dwarfism (as discussed below), along with delayed puberty, hypothyroidism, and adrenal insufficiency.
- Adults: In adults, symptoms include hypothyroidism, adrenal insufficiency, hypogonadism, and often subtle signs of GH deficiency, such as reduced muscle mass, increased central adiposity, and fatigue.
DWARFISM
Dwarfism specifically refers to significantly stunted growth and short stature, often resulting from a deficiency in Growth Hormone.
- Etiology: It is mostly due to a generalized deficiency of anterior pituitary secretion during childhood, which implies that not only GH but often other pituitary hormones (leading to varying degrees of panhypopituitarism) are also deficient.
- GH Deficiency: The most direct cause is an insufficient secretion of GH itself, often due to a pituitary lesion, genetic factors, or idiopathic reasons.
- GHRH Deficiency: Problems with hypothalamic GHRH production can also lead to secondary GH deficiency.
- GH Insensitivity (Laron Syndrome): In some cases, the problem isn't a lack of GH, but rather that the body's tissues are unresponsive to GH. This is due to defects in the GH receptor, leading to a failure to produce IGF-I.
- Clinical Features:
- Proportional Development: Despite their short stature, individuals with pituitary dwarfism generally exhibit all the body physical parts developing in appropriate proportion to one another. They are essentially miniature adults.
- Slow Growth Rate: Their growth rate is significantly slowed. For example, a child who has reached the age of 10 years may have the bodily development and size of a child aged 4 to 5 years. Similarly, a person at age 20 years might have the bodily development of a child aged 7 to 10 years.
- Sexual Maturity: Unless treated, individuals with generalized panhypopituitarism may also have delayed or absent sexual development due to deficiencies in gonadotropins (FSH and LH).
- Mental Development: Importantly, mental development is typically normal, distinguishing them from other forms of dwarfism (e.g., cretinism due to severe hypothyroidism).
- Specific Forms of Dwarfism:
- African Pygmies and Levi-Lorain Dwarfs: In these genetically distinct groups, the rate of growth hormone secretion is often normal or even high. However, the underlying issue is a hereditary inability to form Somatomedin C (IGF-I), which is a key step for the promotion of growth by growth hormone. Their tissues are insensitive to GH due to a defect in the GH receptor or post-receptor signaling, leading to a lack of IGF-I, which is the primary mediator of GH's growth-promoting effects.
GIGANTISM
Gigantism is a condition characterized by excessive growth and abnormally tall stature, resulting from overproduction of Growth Hormone during childhood or adolescence.
- Etiology: Gigantism is typically caused by an acidophilic tumor (adenoma) of the anterior pituitary gland, which secretes large quantities of Growth Hormone. These tumors are often composed of somatotroph cells.
- Timing is Key: The critical factor differentiating gigantism from acromegaly is that the condition occurs before adolescence, specifically before the epiphyses of the long bones have become fused with the shafts.
- Clinical Features:
- Rapid and Excessive Growth: All body tissues grow rapidly, including the bones, leading to an extreme increase in height. Individuals can become exceptionally tall, often reaching heights of up to 8 feet.
- Proportional Growth (initially): While overall size is exaggerated, the body proportions generally remain relatively normal in the early stages, although later stages may show some disproportion.
- Metabolic Complications:
- Hyperglycemia and Diabetes Mellitus: Giants are often hyperglycemic due to the anti-insulin effects of excessive GH. This chronic strain on the pancreatic beta cells can lead to their degeneration, eventually resulting in diabetes mellitus in a significant percentage of these individuals.
- Weakness: Despite their large size, individuals with gigantism often experience generalized body weakness, likely due to the catabolic effects of very high GH levels on muscles and other tissues, or related to the metabolic burden.
- Cardiovascular Issues: Enlargement of organs and increased metabolic demand can strain the cardiovascular system, leading to heart failure over time.
- Treatment: Once gigantism is diagnosed, further effects can often be blocked by:
- Microsurgical Removal of the Tumor: This is the primary and most effective treatment to remove the source of excess GH.
- Irradiation of the Pituitary Gland: Radiation therapy can be used as an alternative or adjuvant treatment, particularly if surgery is not feasible or not completely successful.
- Pharmacological Agents: Medications like somatostatin analogues (which inhibit GH release) or GH receptor antagonists can also be used to control GH levels.
ACROMEGALY
Acromegaly is a condition resulting from the overproduction of Growth Hormone, similar to gigantism, but it occurs after adolescence.
- Etiology: Like gigantism, acromegaly is almost invariably caused by an acidophilic tumor (adenoma) of the anterior pituitary gland that secretes excessive GH.
- Timing is Key: The crucial distinction is that this excessive GH secretion occurs after the epiphyses of the long bones have fused with the shaft. Once the growth plates are closed, longitudinal bone growth is no longer possible.
- Clinical Features (Growth of Bones and Soft Tissues):
- No Increase in Height: The person cannot grow taller.
- Thickening of Bones: Instead, the bones become thicker and denser, particularly in the extremities and membranous bones.
- Soft Tissue Growth: The soft tissues throughout the body continue to grow and proliferate.
- Characteristic Enlargement Patterns:
- Hands and Feet: Enlargement is most marked in the bones of the hands and feet, making them appear broad and large. Patients often report needing larger shoe and ring sizes. The fingers become extremely thickened, often described as "spade-like" (hands can be up to twofold normal size).
- Face and Skull: Significant changes occur in the membranous bones of the skull. This includes:
- Protrusion of the Lower Jaw (Prognathism): The lower jawbone (mandible) grows forward, often by half an inch or more, creating a characteristic prognathic appearance.
- Enlarged Nose: The nose increases significantly in size, sometimes up to twice its normal size.
- Prominent Forehead and Supraorbital Ridges: The forehead slants forward, and the bony ridges above the eyes (supraorbital ridges) become very prominent, creating a heavy brow.
- Bosses on the Forehead: Bony protuberances develop on the forehead.
- Increased Skull Thickness: The cranium generally thickens.
- Spine: Growth of portions of the vertebrae can lead to an exaggerated outward curvature of the thoracic spine, known as kyphosis (hunchback).
- Organomegaly: Internal organs also undergo significant enlargement. The tongue (macroglossia), the liver (hepatomegaly), and especially the kidneys become greatly enlarged.
- Other Soft Tissue Changes: Skin thickens and becomes oily, hair growth may increase, and vocal cords thicken, leading to a deeper voice.
- Metabolic and Systemic Effects: Similar to gigantism, patients with acromegaly also experience:
- Hyperglycemia and Diabetes Mellitus: Due to chronic GH excess causing insulin resistance.
- Cardiovascular Disease: Hypertension, cardiomyopathy, and an increased risk of heart failure.
- Arthritis: Due to joint overgrowth and degeneration.
- Headaches and Visual Field Defects: From the growing pituitary tumor compressing surrounding structures.
- Diagnosis and Treatment: Diagnosis involves measuring elevated GH and IGF-I levels, along with imaging (MRI) of the pituitary gland. Treatment strategies are similar to gigantism:
- Transsphenoidal Surgery: Surgical removal of the pituitary adenoma is the first-line treatment.
- Radiation Therapy: Used as an adjunct or alternative.
- Pharmacological Agents: Somatostatin analogues, GH receptor antagonists, and dopamine agonists are used to control GH and IGF-I levels.
Growth Hormone Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Growth Hormone Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Parathyroid Gland & Calcium Metabolism
Parathyroid Gland & Calcium Metabolism
INTRODUCTION TO CALCIUM METABOLISM
Calcium (Ca²⁺) is the most abundant mineral in the human body, playing a pivotal role far beyond its primary association with bone health. It is an indispensable second messenger in virtually every cell, a key player in nerve impulse transmission, muscle contraction, and blood coagulation. Similarly, phosphate (PO₄³⁻) is a crucial component of bones, cell membranes (phospholipids), genetic material (DNA, RNA), and energy currency (ATP).
The body maintains extremely tight control over the levels of these ions, particularly calcium, in the extracellular fluid (ECF) and plasma. Deviations, even slight ones, can have profound and immediate physiological consequences. This section will explore the regulation of calcium and phosphate, their distribution in the body, and the critical physiological roles they play.
CALCIUM REGULATION IN ECF AND PLASMA
The concentration of calcium ions in the extracellular fluid (ECF) and plasma is precisely and tightly regulated. It rarely deviates significantly from normal levels, highlighting its critical importance for life.
- Normal Value: The normal value of total calcium in the ECF is approximately 9.4 mg/dL (or 2.4 mEq/L). This represents a very small fraction, about 0.1%, of the total calcium in the body.
- Vital Physiological Processes: Calcium ions are absolutely vital to numerous physiological processes, including:
- Contraction of muscles: Essential for the excitation-contraction coupling in skeletal, cardiac, and smooth muscles.
- Blood clotting: A critical cofactor in several steps of the coagulation cascade, facilitating the formation of a stable blood clot.
- Transmission of nerve impulses: Involved in the release of neurotransmitters from presynaptic terminals and influencing neuronal excitability.
- Enzyme activation, hormone secretion, and cell signaling.
- Impact of Deviations: Any significant deviations from the normal ECF calcium levels have immediate and direct effects:
- Low Ca²⁺ (Hypocalcemia): Directly excites neuromuscular systems, leading to increased neuronal excitability, tetany, and muscle spasms.
- High Ca²⁺ (Hypercalcemia): Directly depresses neuromuscular and cardiac systems, leading to muscle weakness, lethargy, and cardiac arrhythmias.
- Distribution of Total Body Calcium:
- Approximately 99% of total body calcium is stored in the bones, serving as a large and readily available reservoir.
- About 1% of total calcium is found in cells, where it functions as a crucial intracellular messenger. The remaining very small fraction is in the ECF and plasma.
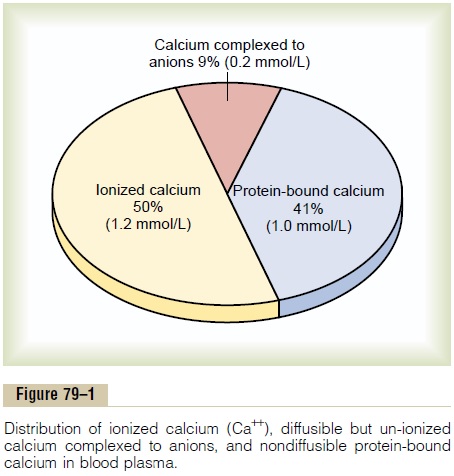
CALCIUM IN PLASMA AND INTERSTITIAL FLUID
In plasma and interstitial fluid, calcium exists in three distinct forms, contributing to the total calcium level:
- 41% Combined with Plasma Proteins: This fraction is primarily bound to albumin and, to a lesser extent, globulins. This protein-bound calcium is non-diffusible through capillary membranes and therefore not physiologically active in terms of directly influencing cell excitability.
- 9% Diffusible, Combined with Anionic Substances: This portion is bound to various anionic substances present in plasma and interstitial fluid, such as citrates and phosphates. This calcium is diffusible across capillary membranes but is not ionized, meaning it is not biologically active in the same way as free calcium ions.
- 50% Diffusible and Ionized (Free Ca²⁺): This is the most crucial form of calcium. It is diffusible across capillary membranes and, most importantly, exists as free calcium ions (Ca²⁺). This ionized calcium is the physiologically active form that participates in muscle contraction, nerve impulse transmission, blood clotting, and other vital cellular processes. Its normal level in plasma is approximately 1.2 mmol/L (or 2.4 mEq/L), which corresponds to roughly 4.7 mg/dL.
The ionized calcium fraction is the one that is tightly regulated by hormones like parathyroid hormone (PTH), vitamin D, and calcitonin.
PHOSPHATE REGULATION IN ECF AND PLASMA
Phosphate is also a vital mineral, but its regulation in the ECF is generally less precise and less tightly controlled than calcium.
- Distribution of Total Body Phosphate:
- Approximately 85% of the body's phosphate is found in bones, predominantly as hydroxyapatite crystals.
- 14-15% is located within cells, where it is integral to intracellular processes (e.g., ATP, DNA, RNA, phospholipids).
- Less than 1% is in the ECF, indicating its relatively minor extracellular presence compared to its intracellular and bone stores.
- ECF Concentration: The concentration of inorganic phosphate in the ECF is typically around 4 mg/dL. This level can vary slightly:
- Adults: Generally 3 to 4 mg/dL.
- Children: Tend to have slightly higher levels, typically 4 to 5 mg/dL, due to higher growth rates.
- Forms in ECF: Inorganic phosphate exists in the ECF in two primary forms:
- HPO₄²⁻ (Divalent Phosphate Ion): Approximately 1.05 mmol/L.
- H₂PO₄⁻ (Monovalent Phosphate Ion): Approximately 0.26 mmol/L.
- Relationship with pH:
- An increase in total ECF phosphate will generally increase the concentrations of both forms.
- A low pH (acidosis) increases the concentration of H₂PO₄⁻ and decreases HPO₄²⁻.
- A high pH (alkalosis) has the reverse effect, increasing HPO₄²⁻ and decreasing H₂PO₄⁻.
- Regulation: Although less tightly regulated than calcium, many of the same factors that regulate ECF calcium concentration (e.g., PTH, Vitamin D) also influence phosphate levels, mainly by affecting its renal excretion and intestinal absorption.
NON-BONE EFFECTS OF ALTERED CA AND PHOSPHATE CONCENTRATIONS IN THE BODY FLUIDS
The immediate physiological impact of altered calcium and phosphate levels differs significantly:
- Phosphate: Changing the level of phosphate in the ECF from far below normal to two to three times normal does not cause major immediate effects on the body. While chronic alterations can have serious consequences, acute changes are often well-tolerated because most phosphate is intracellular or in bone.
- Calcium: In stark contrast, even slight increases or decreases of ionized calcium in the ECF can cause extreme immediate physiological effects. This underscores the body's meticulous regulatory mechanisms for calcium.
- Hypocalcemia: (Low ECF ionized calcium) leads to increased neuromuscular excitability, manifesting as tetany, muscle cramps, tingling, and potentially seizures.
- Hypercalcemia: (High ECF ionized calcium) leads to depressed neuromuscular activity, manifesting as muscle weakness, lethargy, constipation, confusion, and cardiac arrhythmias.
Hence, clinical conditions are primarily discussed in terms of:
- Hypocalcemia vs. Hypercalcemia (which are acutely life-threatening due to effects on excitable tissues)
- Hypophosphatemia vs. Hyperphosphatemia (which tend to have more chronic and metabolic implications, rather than immediate severe effects on excitability).
ALTERED CALCIUM LEVELS
HYPOCALCEMIA
Hypocalcemia occurs when the extracellular fluid (ECF) calcium ion concentration falls below its normal range (normally 9.4 mg/dL). This condition has profound and immediate effects on the nervous and muscular systems due to the role of calcium in regulating cell excitability.
- Increased Nervous System Excitability: As ECF [Ca²⁺] falls, the nervous system becomes progressively more excitable. This is because calcium ions normally stabilize nerve membranes. When calcium is low, nerve fibers become more permeable to sodium ions, making them more likely to depolarize and fire action potentials spontaneously.
- Tetany: At about 6 mg/dL (approximately 50% below the normal ionized calcium level), the peripheral nerve fibers become so excitable that they begin to fire spontaneously, causing generalized muscle contractions known as tetanic contractions (tetany). This can manifest as carpopedal spasm (spasms of the hands and feet) and laryngospasm (spasm of the vocal cords, which can be life-threatening).
- Seizures: Hypocalcemia can also lead to seizures due to its action of increasing excitability in the brain.
- Lethal Level: If ECF [Ca²⁺] drops to about 4 mg/dL, severe hypocalcemia can lead to respiratory arrest (due to laryngospasm or severe muscle spasms) and cardiac arrhythmias, resulting in death.
HYPERCALCEMIA
Hypercalcemia occurs when the level of calcium in the body fluids rises above normal. Unlike hypocalcemia, which excites the nervous system, hypercalcemia tends to depress it.
- Depressed Nervous System: The nervous system becomes depressed, and reflex activities of the central nervous system (CNS) become sluggish. This is because high calcium levels decrease the permeability of nerve membranes to sodium ions, making them less excitable.
- Cardiac Effects: Hypercalcemia decreases the QT interval of the heart on an electrocardiogram (ECG), which can lead to arrhythmias.
- Gastrointestinal Effects: It can cause lack of appetite (anorexia) and constipation due to decreased smooth muscle activity in the gastrointestinal tract.
- Severity:
- Effects begin to appear at about 12 mg/dL.
- Become marked above 15 mg/dL.
- Very high levels (e.g., above 17 mg/dL) can lead to lethargy, coma, and cardiac arrest.
LINES OF DEFENCE FROM CHANGES IN [CA++]
The body employs two main lines of defense to prevent significant alterations in ECF calcium concentration, ensuring its tight regulation:
- Buffer Function of the Exchangeable Calcium in Bones—The First Line of Defense:
- Bones contain a large reservoir of calcium, a small portion of which is in a readily exchangeable form. This exchangeable calcium is in dynamic equilibrium with the ECF.
- If ECF [Ca²⁺] begins to fall, calcium can be rapidly released from this exchangeable pool in the bones into the ECF.
- Conversely, if ECF [Ca²⁺] rises, calcium can be rapidly taken up by the bone.
- This rapid exchange acts as an immediate, short-term buffer system to minimize acute fluctuations in ECF calcium.
- Hormonal Control of Calcium Ion Concentration—The Second Line of Defense:
- For long-term and fine-tuned regulation, the body relies on specific hormones that control calcium homeostasis. These hormones primarily act on the gut, kidneys, and bone.
- The three main hormones involved are:
- Parathyroid Hormone (PTH): The most critical regulator, increasing ECF [Ca²⁺].
- Calcitriol (active Vitamin D): Works synergistically with PTH, increasing intestinal absorption of calcium.
- Calcitonin: Generally decreases ECF [Ca²⁺], though its role in adult human calcium homeostasis is less dominant than PTH and Vitamin D.
ABSORPTION AND EXCRETION OF CA AND PHOSPHATE
Calcium and phosphate balance in the body is a result of the interplay between:
- Intestinal Absorption: The uptake of these minerals from the diet into the bloodstream.
- Renal Excretion: The removal of excess minerals from the bloodstream via the kidneys into the urine.
- Bone Turnover: The continuous process of bone formation (deposition of calcium and phosphate) and bone resorption (release of calcium and phosphate) from the skeleton.
These processes are tightly regulated by the hormonal control system.
VITAMIN D
Vitamin D is a fat-soluble vitamin that plays a critical role in calcium and phosphate homeostasis. However, Vitamin D itself is not the active substance that directly causes these effects. Instead, it must be metabolized into its active form.
- Potent Effect: Its most potent and well-known effect is to increase calcium absorption from the intestinal tract.
SYNTHESIS AND METABOLISM OF VITAMIN D
Vitamin D exists in several forms and undergoes a series of hydroxylations to become biologically active:
- Sources of Precursor Vitamin D:
- Skin Synthesis: Vitamin D₃ (cholecalciferol) is synthesized in the skin when 7-dehydrocholesterol is exposed to ultraviolet B (UVB) radiation from sunlight.
- Dietary Sources:
- Vitamin D₂ (ergocalciferol): Obtained in the diet primarily from plant sources (e.g., fortified foods, some mushrooms).
- Vitamin D₃ (cholecalciferol): Also obtained in the diet from animal sources (e.g., fatty fish, fish liver oil, fortified dairy).
- First Hydroxylation (in the Liver):
- Both dietary Vitamin D₂ and D₃, as well as D₃ synthesized in the skin, are transported to the liver.
- In the liver, they undergo hydroxylation at the 25-position by the enzyme 25-hydroxylase, converting them into 25-hydroxyvitamin D (25(OH)D), also known as calcidiol.
- Calcidiol is the main circulating form of Vitamin D and is used as an indicator of a person's Vitamin D status.
- Second Hydroxylation (in the Kidney):
- Calcidiol (25(OH)D) then travels to the kidneys.
- In the kidneys, it is converted to the most active form, 1,25-dihydroxyvitamin D (1,25(OH)₂D), also known as calcitriol, by the enzyme 1-alpha-hydroxylase.
- This step is tightly regulated, primarily by Parathyroid Hormone (PTH). Elevated serum PTH increases the hydroxylation of Vitamin D in the kidney, thus increasing the production of calcitriol.
PHYSIOLOGICAL EFFECTS OF VITAMIN D (CALCITRIOL)
The active form of Vitamin D, calcitriol, has several critical physiological effects on calcium and phosphate homeostasis:
- Facilitates Intestinal Absorption: It is the primary hormone that facilitates the uptake of calcium from the intestinal epithelium into the bloodstream. This is its most crucial role in raising plasma calcium levels.
- Enhances Cellular Transport: It enhances the transport of calcium through and out of cells in various tissues, including the intestine and bone.
- Bone Turnover: It is important for normal bone turnover, working in concert with PTH to facilitate bone remodeling. While it promotes calcium and phosphate deposition into bone, it can also, under certain conditions (especially in the presence of PTH), mobilize calcium from bone.
- Promotes Phosphate Absorption: In addition to calcium, it also promotes phosphate absorption by the intestines, thereby increasing plasma phosphate levels.
- Decreases Renal Excretion: It decreases renal calcium and phosphate excretion, promoting their reabsorption in the kidneys and reducing their loss in urine. This also contributes to increasing plasma levels of both minerals.
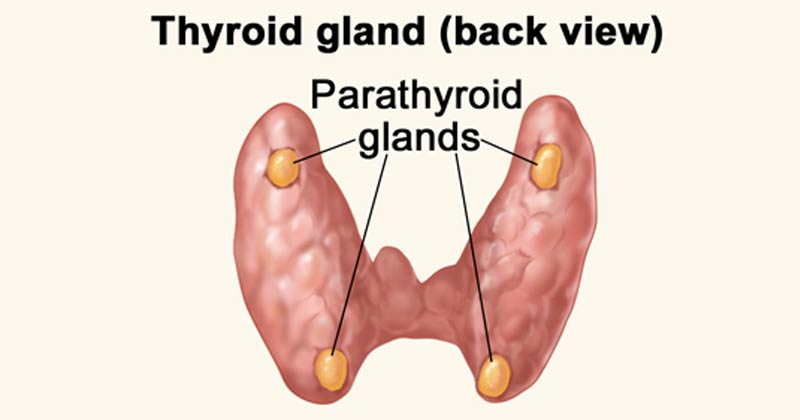
PARATHYROID GLANDS
The parathyroid glands are small endocrine glands that play a central role in maintaining calcium homeostasis.
Physiological Anatomy of Parathyroid Glands:
- Number and Location: Humans typically have four parathyroid glands. They are located immediately behind the thyroid gland, with one gland situated behind each of the upper poles and each of the lower poles of the thyroid.
- Size and Appearance: Each parathyroid gland is quite small, typically about 6 mm long, 3 mm wide, and 2 mm thick. Macroscopically, they have a characteristic dark brown, fatty appearance, which can make them challenging to identify during surgery.
Histology of Parathyroid Glands:
The parathyroid gland of the adult human being primarily consists of two main cell types:
- Chief Cells (or Principal Cells):
- These are the most numerous cells and are believed to be responsible for secreting most, if not all, of the Parathyroid Hormone (PTH).
- They are characterized by a relatively clear cytoplasm in their inactive state and a more granular cytoplasm when actively synthesizing and secreting PTH.
- Oxyphil Cells:
- These cells are present in small to moderate numbers in adult human parathyroid glands.
- However, oxyphil cells are often absent in many animals and in young humans.
- Their function is not entirely certain, but they are generally believed to be modified or depleted chief cells that no longer secrete hormone. They typically appear later in life and increase with age.
PARATHYROID HORMONE (PTH)
Parathyroid Hormone (PTH) is the single most important hormone for the minute-to-minute regulation of ECF calcium concentration. It provides a powerful mechanism for controlling both ECF calcium and phosphate levels.
Chemistry:
- Polypeptide Structure: PTH is a polypeptide composed of 84 amino acids. It has a molecular weight (MW) of approximately 9500.
- Active Fragment: Interestingly, smaller compounds, specifically the first 34 amino acids adjacent to the N-terminus of the molecule, can also exhibit full PTH activity. This N-terminal fragment is the biologically active portion.
- Metabolism and Measurement: The full-length PTH (84 amino acids) is rapidly cleared by the kidneys. However, the inactive C-terminal fragments of PTH are cleared much more slowly, allowing them to circulate for hours. Therefore, a large share of measured PTH function in clinical assays often reflects these circulating fragments. Measuring intact PTH (1-84) is usually preferred for more accurate assessment of parathyroid function.
Overall Regulatory Role:
PTH primarily regulates ECF calcium and phosphate by acting on:
- Intestinal Reabsorption: Indirectly through its effects on Vitamin D activation.
- Renal Excretion: Directly affecting the reabsorption and secretion of calcium and phosphate in the kidneys.
- Exchange Between ECF and Bone: Directly stimulating bone cells to release or take up calcium and phosphate.
EFFECTS OF PTH ON [CA++] AND [PHOSPHATE] IN ECF
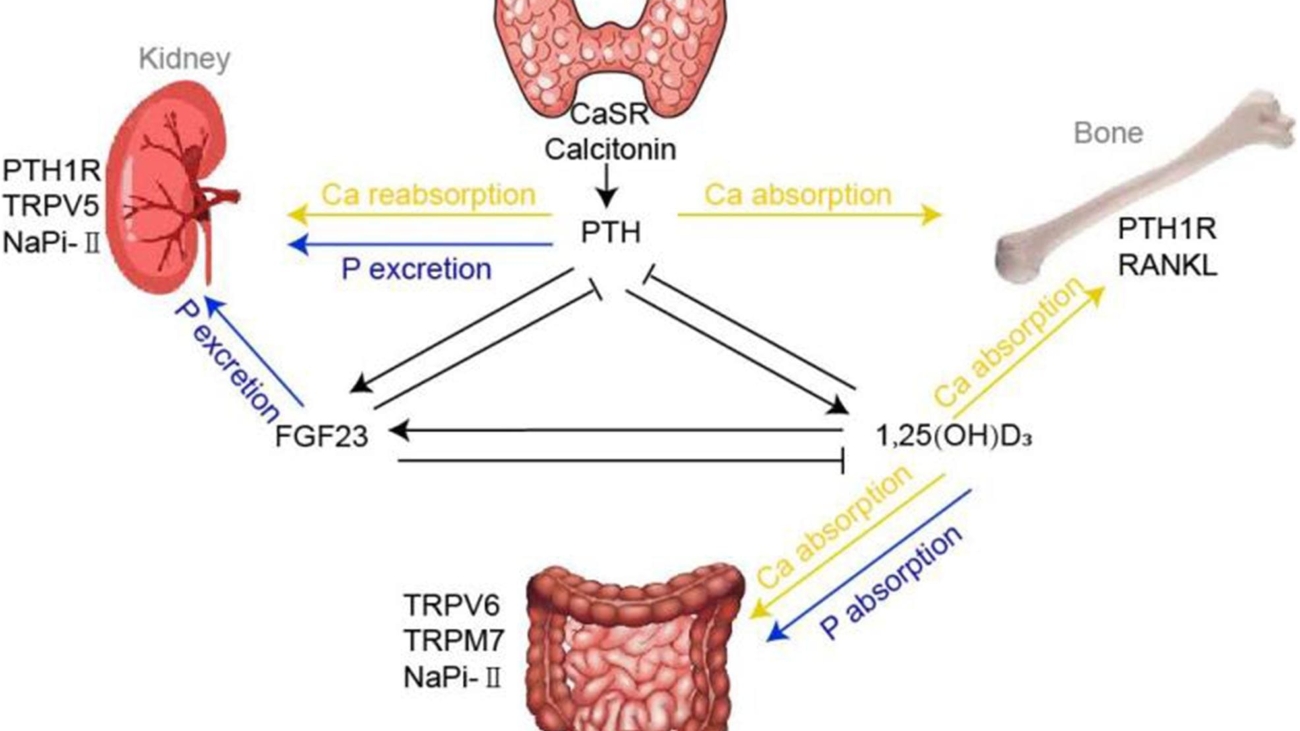
PTH exerts three main effects to increase ECF calcium concentration and generally decrease ECF phosphate concentration:
- Increases Calcium and Phosphate Absorption from the Bone.
- Decreases Calcium Excretion and Increases Phosphate Excretion by the Kidneys.
- Increases Intestinal Absorption of Calcium and Phosphate (indirectly, via Vitamin D activation).
Let's look at each of these in more detail:
1. Increases Calcium and Phosphate Absorption from the Bone
PTH has two phases of action on bone, both leading to the release of calcium and phosphate into the ECF:
- Rapid Phase (Minutes to Hours):
- This phase involves the activation of already existing bone cells, primarily the osteocytes (bone cells embedded within the bone matrix) and potentially osteoblasts (bone-forming cells).
- PTH stimulates these cells to promote the rapid transfer of calcium and phosphate from the bone fluid, which surrounds the bone crystals, into the ECF. This process is thought to involve the osteocytic-osteoblastic pump and increased permeability of the osteocyte membrane.
- Slow Phase (Days to Weeks):
- This phase involves the stimulation of osteoclasts (large cells that resorb bone tissue).
- PTH directly stimulates osteoblasts, which then produce signaling molecules (like RANKL) that activate osteoclasts.
- This leads to the proliferation of osteoclasts and a marked increase in osteoclastic resorption of bone itself, not just absorption from bone fluid. This breaks down the bone matrix, releasing large quantities of calcium and phosphate into the ECF.
2. Decreases Calcium Excretion and Increases Phosphate Excretion by the Kidneys
PTH has opposing effects on calcium and phosphate handling by the kidneys, which is crucial for maintaining their balance:
- Diminishes Proximal Tubular Reabsorption of Phosphate Ions: PTH acts on the renal tubules, particularly the proximal tubule, to decrease the reabsorption of phosphate. This leads to increased phosphate excretion in the urine (phosphaturia), which helps to lower ECF phosphate levels.
- Increases Renal Tubular Reabsorption of Calcium: At the same time that it promotes phosphate excretion, PTH significantly increases the reabsorption of calcium in the renal tubules.
- This increased Ca²⁺ reabsorption occurs mainly in the late distal tubules, the collecting tubules, the early collecting ducts, and possibly the ascending loop of Henle to a lesser extent.
- Importance of Renal Effect: This dual effect on the kidneys is vital. Were it not for the effect of PTH on the kidneys to increase Ca²⁺ reabsorption, the continuous loss of Ca²⁺ into the urine would eventually deplete both the ECF and the bones of this essential mineral, even with PTH's bone-resorbing effects.
3. Increases Intestinal Absorption of Calcium and Phosphate
PTH does not directly act on the intestines. Instead, it exerts this effect indirectly by stimulating the production of active Vitamin D (calcitriol):
- PTH increases the formation in the kidneys of 1,25-dihydroxycholecalciferol (calcitriol) from inactive Vitamin D precursors.
- As discussed earlier, calcitriol is then responsible for directly increasing the absorption of both calcium and phosphate from the gastrointestinal tract.
ROLE OF CAMP IN PTH ACTIONS
Many of the cellular effects of PTH are mediated by the cyclic AMP (cAMP) second messenger system.
- Mechanism: When PTH binds to its receptors on target cells (e.g., osteocytes, osteoclasts, renal tubular cells), it activates adenylate cyclase, leading to an accumulation of cAMP within the cell.
- Resulting Actions: This increase in intracellular cAMP then triggers a cascade of events that result in:
- Osteoclastic secretion of enzymes and acids to cause bone resorption (as part of the slow phase of bone effect).
- Formation of 1,25-dihydroxycholecalciferol in the kidneys (activation of 1-alpha-hydroxylase).
- Altered transport mechanisms in the renal tubules leading to increased Ca²⁺ reabsorption and decreased phosphate reabsorption.
- Other Mechanisms: However, it is also believed that other direct effects of PTH on cells may occur independent of cAMP, indicating that PTH signaling can be complex.
CONTROL OF PTH SECRETION BY [CA++]
The secretion of PTH is under an extremely potent and sensitive negative feedback mechanism, directly regulated by the concentration of ionized calcium in the ECF:
- Decrease in ECF [Ca²⁺]: A decrease in ECF [Ca²⁺] is the primary stimulus for increasing PTH production and secretion by the chief cells of the parathyroid glands.
- If this decrease in calcium is prolonged, it can lead to hypertrophy of the parathyroid glands (an increase in their size and cell number) to produce more PTH. This is observed in conditions like rickets (due to chronic low calcium/vitamin D) and also occurs physiologically during pregnancy and lactation, when calcium demands are high.
- Increase in ECF [Ca²⁺]: Conversely, an increase in ECF [Ca²⁺] directly decreases PTH production and secretion.
- If this increase is prolonged, it can lead to atrophy of the parathyroid glands (a decrease in their size and activity). Examples include:
- Excess quantities of calcium in the diet.
- Increased vitamin D in the diet (leading to increased intestinal calcium absorption).
- Bone absorption caused by other factors not involving PTH (e.g., certain bone cancers releasing calcium).
- If this increase is prolonged, it can lead to atrophy of the parathyroid glands (a decrease in their size and activity). Examples include:
This sensitive feedback loop ensures that PTH levels are precisely adjusted to maintain ECF calcium within its narrow physiological range.
CALCITONIN
Calcitonin is a hormone that, in some ways, acts as an antagonist to PTH, primarily by lowering blood calcium levels.
- Chemistry: Calcitonin is a peptide hormone composed of 32 amino acids, with a molecular weight of approximately 3400.
- Source: It is secreted by the Parafollicular cells (C-cells) of the thyroid gland. These C-cells are located in the interstitial fluid (ISF) between the follicles of the thyroid gland.
- Developmental Origin: C-cells constitute a small percentage (about 0.1%) of the thyroid gland and are considered remnants of the ultimobranchial glands of lower animals (such as fish, amphibians, reptiles, and birds), which play a more prominent role in calcium regulation in those species.
- Stimulus for Secretion: Calcitonin is secreted primarily in response to an increase in extracellular fluid (ECF) calcium concentration.
- Effects on Calcium and Phosphate: Calcitonin generally has effects opposite to those of PTH, meaning it tends to decrease ECF calcium levels.
- Decreases Osteoclastic Activity: It primarily acts to inhibit osteoclastic bone resorption, thus preventing the release of calcium and phosphate from bone into the ECF.
- Increases Renal Calcium Excretion: It also slightly increases renal excretion of calcium, though this effect is less pronounced than its action on bone.
- Quantitative Role in Adults: The quantitative role of calcitonin in regulating ECF [Ca²⁺] in healthy adult humans is considered far less significant than that of PTH. Its effects are often weak in adults and are frequently overridden by the more powerful regulatory mechanisms of PTH.
- Significant Effects in Specific Conditions: However, calcitonin can have more potent and clinically relevant effects in certain situations:
- Children: It is more active in children due to their rapid bone remodeling and growth.
- Paget's Disease: It is used therapeutically in conditions like Paget's disease, which is characterized by accelerated and disorganized osteoclastic activity, where calcitonin can help to reduce bone resorption.
PATHOPHYSIOLOGY OF CALCIUM AND PHOSPHATE DISORDERS
The balance of calcium and phosphate can be disrupted by various pathophysiological conditions, primarily involving:
- Parathyroid Hormone (PTH) Abnormalities: Either too much (hyperparathyroidism) or too little (hypoparathyroidism).
- Vitamin D Abnormalities: Deficiency or disorders of its metabolism.
- Bone Diseases: Conditions that directly affect bone structure and metabolism.
HYPOPARATHYROIDISM
Hypoparathyroidism is a condition characterized by insufficient secretion of PTH.
- Etiology: It most commonly results from accidental removal or damage to the parathyroid glands during thyroid surgery.
- Consequences of PTH Deficiency:
- Decreased Bone Resorption: Without sufficient PTH, the osteocytic reabsorption of exchangeable Ca²⁺ decreases, and the osteoclasts become almost totally inactive. As a result, Ca²⁺ and phosphate reabsorption from the bones is severely depressed.
- Hypocalcemia: This leads to a significant decrease in body fluid [Ca²⁺] (hypocalcemia).
- Hyperphosphatemia: The renal tubules fail to excrete phosphate effectively, leading to increased blood phosphate levels (hyperphosphatemia).
- Strong Bones: Paradoxically, in the absence of PTH, bone resorption is minimal, and the bones usually remain strong, often denser than normal, as calcium and phosphate are not being adequately mobilized.
- Clinical Manifestations:
- Rapid Calcium Drop: Following removal of the parathyroid glands, ECF [Ca²⁺] can fall rapidly from the normal 9.4 mg/dL to 6-7 mg/dL within 2 to 3 days.
- Blood Phosphate Doubles: Concurrently, blood phosphate levels can double due to decreased renal excretion.
- Tetany: At calcium levels of 6-7 mg/dL, the characteristic signs of tetany begin to develop due to increased neuromuscular excitability. This is particularly dangerous if it affects the laryngeal muscles, causing spasm and potentially obstructing respiration, which can lead to death.
Treatment of Hypoparathyroidism:
- PTH Administration: While PTH can be administered, it is not usually the primary long-term treatment due to its high cost, short half-life, and potential for immune reactions.
- Vitamin D and Calcium Supplementation (Primary Treatment): The most common and effective treatment involves:
- Large Quantities of Vitamin D: Administering high doses of Vitamin D (e.g., 100,000 units per day) to stimulate intestinal calcium absorption.
- Oral Calcium Intake: Augmenting this with high oral intake of calcium (e.g., 1 to 2 grams per day). This combination helps to keep ECF [Ca²⁺] within the normal range.
- 1,25-Dihydroxycholecalciferol (Calcitriol): Sometimes, 1,25-dihydroxycholecalciferol (the active form of Vitamin D) is administered. It is much more potent and acts faster. However, its high potency can make it difficult to control, leading to potential hypercalcemia if not carefully monitored.
PRIMARY HYPERPARATHYROIDISM
Primary hyperparathyroidism results from an abnormality of the parathyroid glands causing inappropriate and excess PTH secretion.
- Etiology:
- Parathyroid Adenoma: In the vast majority of cases (85-90%), it is caused by a benign tumor (adenoma) of one of the parathyroid glands. Less commonly, it can be due to hyperplasia of all glands or, rarely, carcinoma.
- Gender Predisposition: These tumors occur much more frequently in women than in men or children, possibly due to the increased stress on calcium metabolism during pregnancy and lactation, which can predispose the parathyroid glands to hyperactivity.
- Consequences of Excess PTH:
- Extreme Osteoclastic Activity: The excessive PTH leads to extreme osteoclastic activity in the bones, causing continuous and significant release of calcium and phosphate from bone into the ECF.
- Hypercalcemia: This consistently elevates ECF [Ca²⁺].
- Hypophosphatemia: Simultaneously, the high PTH levels cause increased renal excretion of phosphate, leading to usually depressed concentrations of phosphate ions in the ECF.
Effects of Primary Hyperparathyroidism:
- Bone Disease (Osteitis Fibrosa Cystica):
- In severe hyperparathyroidism, the osteoclastic absorption of bone significantly outstrips osteoblastic deposition. This leads to bone demineralization, fibrous replacement of bone tissue, and the formation of bone cysts, a condition known as osteitis fibrosa cystica. Bones become fragile and prone to fractures.
- Hypercalcemia:
- Plasma calcium levels rise, typically to 12-15 mg/dL, and rarely even higher. The symptoms of hypercalcemia ensue as discussed earlier (depressed nervous system, sluggish reflexes, muscle weakness, constipation, cardiac arrhythmias, polyuria, and polydipsia).
- Metastatic Calcification:
- When extreme quantities of PTH are secreted, ECF [Ca²⁺] rises rapidly to very high values. While PTH normally decreases phosphate, if calcium levels are excessively high, and phosphate levels are not sufficiently decreased (or are increased by other factors), the product of calcium and phosphate concentrations can exceed the solubility constant.
- This leads to supersaturation of CaHPO₄, and crystals of calcium phosphate are deposited in soft tissues throughout the body, a process called metastatic calcification. Common sites include the alveoli of the lungs, renal tubules, thyroid gland, artery walls, and stomach. This can be fatal within days if severe.
- Formation of Kidney Stones (Nephrolithiasis):
- The excess calcium and phosphate absorbed from the intestines (due to PTH-induced Vitamin D activation) or mobilized from the bones leads to significantly increased concentrations of these minerals in the urine.
- This increased urinary concentration, especially of calcium, often results in the precipitation of calcium phosphate or calcium oxalate crystals in the kidney tubules, leading to the formation of kidney stones.
SECONDARY HYPERPARATHYROIDISM
Secondary hyperparathyroidism refers to high levels of PTH that occur as a compensation for chronic hypocalcemia, rather than an intrinsic problem with the parathyroid glands themselves.
- Mechanism: Any condition that consistently lowers ECF [Ca²⁺] will stimulate the parathyroid glands to hypertrophy and secrete more PTH in an attempt to normalize calcium levels.
- Common Causes:
- Vitamin D Deficiency: Insufficient Vitamin D leads to poor intestinal calcium absorption, causing hypocalcemia and stimulating PTH secretion.
- Chronic Renal Disease: Damaged kidneys are unable to produce sufficient amounts of 1,25-dihydroxycholecalciferol (the active form of Vitamin D) due to impaired 1-alpha-hydroxylase activity. This results in impaired intestinal calcium absorption and hypocalcemia, leading to compensatory PTH elevation. The damaged kidneys also retain phosphate, which further contributes to stimulating PTH secretion.
RICKETS (VITAMIN D DEFICIENCY IN CHILDREN)
Rickets is a bone-softening disease that occurs in children due to a deficiency of Vitamin D, which is essential for proper calcium and phosphate absorption and bone mineralization.
- Etiology: Lack of sufficient Vitamin D, often due to inadequate dietary intake or insufficient exposure to sunlight (UVB radiation needed for skin synthesis).
- Preventive Measure: Adequate exposure to sunlight is crucial for prevention.
- Effects:
- Decreased Plasma Calcium and Phosphate: Vitamin D deficiency leads to impaired intestinal absorption of calcium and phosphate, causing plasma concentrations of both minerals to decrease.
- Weakens Bones: The lower calcium and phosphate levels mean insufficient mineralization of growing bones, leading to soft, weak, and deformed bones.
- Compensatory Secondary Hyperparathyroidism: The hypocalcemia stimulates a compensatory increase in PTH secretion (secondary hyperparathyroidism) which attempts to normalize calcium by resorbing bone, further weakening it, and increasing renal phosphate excretion.
- Tetany: In severe rickets, if ECF [Ca²⁺] falls below 7 mg/dL despite compensatory PTH, tetany can occur.
- Treatment:
- Supplementation: Supplying adequate calcium and phosphate in the diet.
- Vitamin D Administration: Administering large amounts of Vitamin D to restore proper calcium and phosphate absorption and bone mineralization.
ADULT RICKETS (OSTEOMALACIA)
Osteomalacia is the adult equivalent of rickets, characterized by defective bone mineralization leading to soft bones.
- Etiology: Adults seldom have a serious dietary deficiency of Vitamin D or calcium. However, serious deficiencies can occasionally occur, particularly due to:
- Malabsorption Syndromes: Conditions like steatorrhea (failure to absorb fat) are significant causes. Since Vitamin D is fat-soluble, its absorption is impaired in steatorrhea. Additionally, calcium tends to form insoluble soaps with unabsorbed fat in the gut, which are then passed in feces, further exacerbating calcium deficiency.
- Clinical Presentation: Adult rickets (osteomalacia) causes bone pain, muscle weakness, and increased risk of fractures. It typically never proceeds to the stage of tetany in adults as the skeletal system is already mature, and the calcium demands are different compared to growing children. However, it often is a cause of severe bone disability.
RENAL RICKETS
Renal rickets is a type of osteomalacia that results from prolonged kidney damage, often seen in chronic kidney disease.
- Mechanism: The damaged kidneys are unable to perform their critical role in converting 25-hydroxyvitamin D to 1,25-dihydroxycholecalciferol (the active form of Vitamin D) due to impaired 1-alpha-hydroxylase activity. This leads to Vitamin D deficiency (even if intake is adequate), impaired intestinal calcium absorption, hypocalcemia, and subsequent secondary hyperparathyroidism.
- Severity: This condition is particularly severe in patients undergoing hemodialysis, as their kidney function is severely compromised.
- Vitamin D-Resistant Rickets: Renal rickets can also be caused by congenital hypophosphatemia, which results from congenitally reduced reabsorption of phosphates by the renal tubules. This form of rickets is often referred to as Vitamin D-resistant rickets because it doesn't respond to typical doses of Vitamin D and requires specialized treatment.
OSTEOPOROSIS
Osteoporosis is the most common of all bone diseases in adults, especially prevalent in old age.
- Key Characteristic: It results primarily from diminished organic bone matrix (e.g., collagen, proteoglycans) rather than from poor bone calcification. While the bone that is present is normally mineralized, there is simply less of it.
- Pathophysiology:
- Imbalance in Bone Remodeling: Normally, bone undergoes continuous remodeling, with osteoblastic activity (bone formation) balanced by osteoclastic activity (bone resorption). In osteoporosis, osteoblastic activity is often less than normal, and consequently, the rate of bone osteoid deposition is depressed. This leads to a net loss of bone mass over time.
- Common Causes:
- Lack of Physical Stress on the Bones: Inactivity and a sedentary lifestyle reduce the mechanical stress on bones, which is a critical stimulus for osteoblastic activity and bone formation.
- Malnutrition: Insufficient protein intake means that a sufficient protein matrix (collagen) cannot be formed, which is essential for building new bone.
- Postmenopausal Lack of Estrogen Secretion: Estrogen plays a crucial role in inhibiting osteoclastic activity and promoting bone formation. After menopause, the sharp decline in estrogen levels in women leads to accelerated bone loss, making it a major risk factor for osteoporosis.
- Lack of Vitamin C: Vitamin C (ascorbic acid) is essential for collagen synthesis. Deficiency can impair the formation of the organic bone matrix.
- Old Age: With aging, there is a natural decline in osteoblastic activity and an increase in bone resorption, contributing to age-related bone loss.
- Cushing's Syndrome: Excess glucocorticoids (as in Cushing's syndrome or long-term corticosteroid therapy) directly inhibit osteoblast function and promote osteoclast activity, leading to bone loss.
Parathyroid & Calcium Metabolism Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Parathyroid & Calcium Metabolism Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Hypothalamus & Pituitary Physiology
Hypothalamus & Pituitary Gland Physiology
INTRODUCTION TO THE HYPOTHALAMUS & PITUITARY GLAND
The hypothalamus and pituitary gland form a crucial functional unit at the base of the brain, acting as the primary link between the nervous system and the endocrine system. Together, they regulate virtually all hormonal functions of the body, maintaining homeostasis, governing growth, metabolism, reproduction, and stress responses.
- The hypothalamus, a small but immensely powerful region of the diencephalon, serves as the command center, integrating neural signals from the brain and translating them into hormonal signals.
- The pituitary gland (also known as the hypophysis), often dubbed the "master gland," receives these signals from the hypothalamus and, in turn, secretes hormones that control other endocrine glands throughout the body.
FUNCTIONS OF THE HYPOTHALAMUS
The hypothalamus is a highly specialized region of the brain responsible for maintaining various homeostatic functions and integrating responses to internal and external stimuli. Its diverse functions include:
1. Autonomic Nervous System Regulation
The hypothalamus is a major control center for the autonomic nervous system (ANS), influencing both its sympathetic and parasympathetic divisions. It regulates involuntary functions such as heart rate, blood pressure, digestion, respiration, and pupil dilation, adapting the body's internal environment to changing conditions.
2. Hormone Production
The hypothalamus itself produces several hormones. These include:
- Releasing hormones and inhibiting hormones that control the secretion of hormones from the anterior pituitary.
- Antidiuretic hormone (ADH, or vasopressin) and oxytocin, which are synthesized in the hypothalamic nuclei and then transported to the posterior pituitary for storage and release.
3. Endocrine Regulation
This is a primary function. Through its production of releasing and inhibiting hormones, the hypothalamus controls the secretion of nearly all anterior pituitary hormones, thereby indirectly regulating many other endocrine glands (e.g., thyroid, adrenal cortex, gonads).
4. Circadian Rhythm Regulation
The suprachiasmatic nucleus (SCN) within the hypothalamus acts as the body's primary biological clock, regulating circadian rhythms such as the sleep-wake cycle, body temperature, and hormone secretion patterns in response to light-dark cues.
5. Limbic System Interaction
The hypothalamus is intimately connected with the limbic system, the part of the brain involved in emotion, motivation, and memory. This connection allows the hypothalamus to integrate emotional responses with physiological functions, influencing behaviors like feeding, aggression, and sexual drive.
6. Integration of Basic Drives
It is involved in regulating fundamental physiological drives and behaviors, such as thirst, hunger, satiety, sexual behavior, and defensive reactions.
7. Temperature Regulation
The hypothalamus contains specialized thermoreceptors and serves as the body's thermostat. It initiates physiological responses (e.g., sweating, shivering, vasodilation/vasoconstriction) to maintain a stable core body temperature within a narrow range.
8. Feeding
Specific nuclei within the hypothalamus (e.g., ventromedial nucleus for satiety, lateral hypothalamus for hunger) play critical roles in regulating food intake and energy balance.
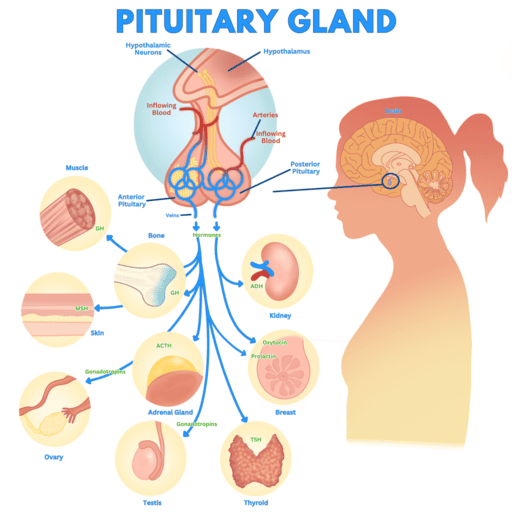
THE PITUITARY GLAND (HYPOPHYSIS, MASTER GLAND)
The pituitary gland is a small, pea-sized endocrine gland, approximately 1 cm in diameter and weighing about 0.5 to 1 gram. It is strategically located within the sella turcica, a bony cavity at the base of the brain, protecting it from injury.
The pituitary gland is functionally and anatomically connected to the hypothalamus by the pituitary stalk (or hypophysial stalk/infundibulum), a slender structure containing blood vessels and nerve fibers.
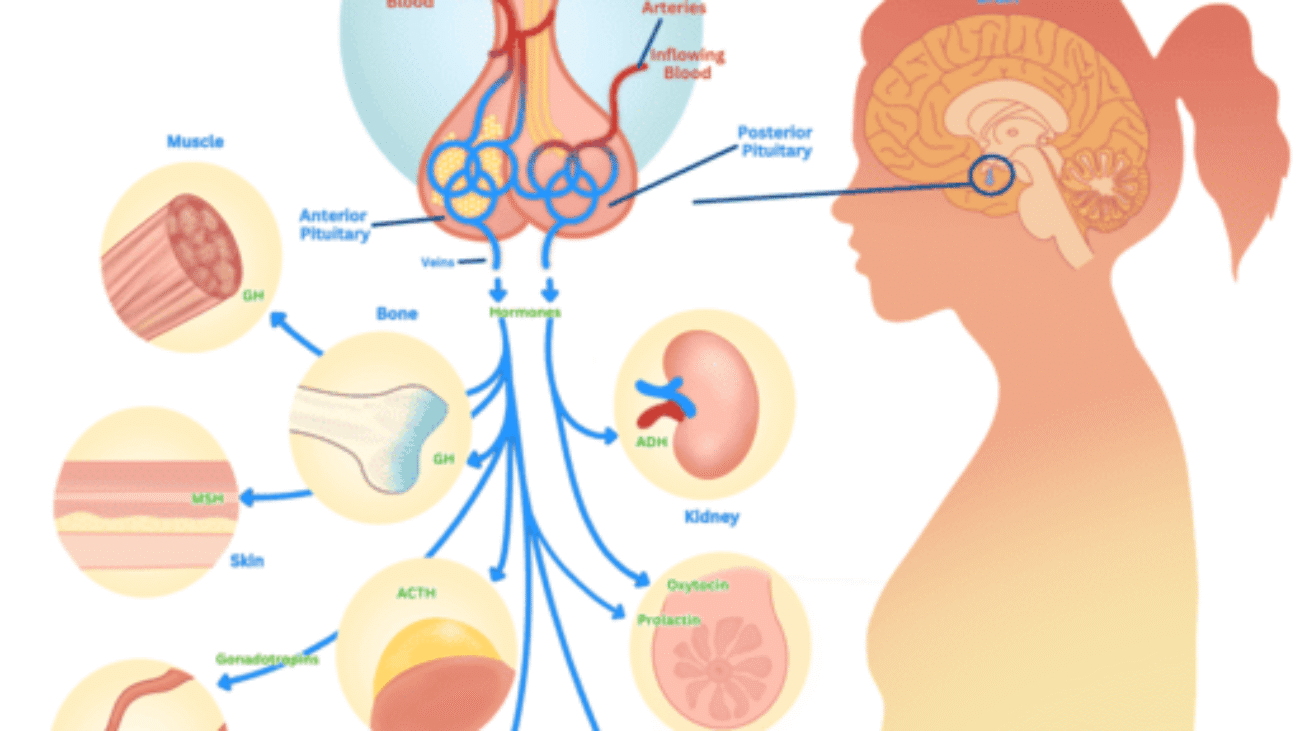
Structurally and functionally, the pituitary gland is divided into two distinct lobes:
- Anterior Pituitary Lobe (Adenohypophysis): Constitutes about two-thirds of the gland.
- Posterior Pituitary Lobe (Neurohypophysis): Constitutes about one-third of the gland.
A. THE ANTERIOR PITUITARY LOBE (ADENOHYPOPHYSIS)
The anterior pituitary is an endocrine gland in its own right, synthesizing and secreting a variety of vital hormones.
- Pars Intermedia: In the fetus, there is a small, avascular tissue called the pars intermedia located between the anterior and posterior lobes. It is much more functional in some lower animals (fish, amphibians, reptiles) but is largely vestigial and no longer present as a distinct functional unit in adult humans, though some of its cells may be dispersed within the anterior lobe.
- Adult Structure: In adults, the anterior pituitary consists of two main parts:
- Pars Distalis: This is the rounded, major endocrine part of the gland, responsible for secreting most of the anterior pituitary hormones. This is what is commonly referred to as the "anterior pituitary."
- Pars Tuberalis: A thin, upward extension that wraps around the infundibulum (pituitary stalk). Its precise function in humans is less understood but is believed to contribute to seasonal and circadian rhythms.
The Anterior Pituitary Gland Cells:
Histologically, the anterior pituitary contains various types of secretory cells, traditionally classified by their staining properties as chromophils (acidophils and basophils) or chromophobes. Each cell type is typically responsible for producing a specific hormone or hormones:
Chromophils: Acidophils
- Somatotropes: Constitute approximately 30-40% of anterior pituitary cells and secrete human Growth Hormone (hGH).
- Lactotropes (or Mammotropes): Constitute approximately 3-5% of anterior pituitary cells and secrete Prolactin (PRL).
Chromophils: Basophils
- Corticotropes: Constitute approximately 20% of anterior pituitary cells and secrete Adrenocorticotropic Hormone (ACTH).
- Thyrotropes: Constitute approximately 3-5% of anterior pituitary cells and secrete Thyroid-Stimulating Hormone (TSH).
- Gonadotropes: Constitute approximately 3-5% of anterior pituitary cells and secrete the gonadotropic hormones: Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH).
Embryological Origin: The anterior pituitary is embryologically derived from Rathke's pouch, an upward invagination of epithelial tissue from the roof of the primitive pharynx (mouth). This epithelial origin distinguishes it from the posterior pituitary, which has a neural origin.
B. THE POSTERIOR PITUITARY LOBE (NEUROHYPOPHYSIS)
The posterior pituitary is functionally an extension of the hypothalamus, serving as a storage and release site for hormones produced by hypothalamic neurons.
- Embryological Origin: It is embryologically derived from a downward outgrowth of nervous tissue from the hypothalamus. This neural origin explains its structural and functional connection to the brain.
- Structure: It is in direct contact with the infundibulum (pituitary stalk) and physically associated with the adenohypophysis. It consists mainly of the Pars Nervosa, which is essentially a collection of axons and nerve terminals originating from the hypothalamus, along with specialized glial cells called pituicytes.
- Neural Part: The posterior pituitary is distinctly the neural part of the pituitary gland. It does not synthesize hormones itself but stores and releases hormones produced by the hypothalamus.
HYPOTHALAMIC CONTROL OF PITUITARY SECRETIONS
The hypothalamus exerts profound control over almost all secretions by both lobes of the pituitary gland. This control is achieved through distinct mechanisms:
- Anterior Pituitary: Controlled primarily by hormonal signals from the hypothalamus.
- Posterior Pituitary: Controlled primarily by nervous signals from the hypothalamus.
A. RELATIONSHIP WITH THE ANTERIOR PITUITARY GLAND: THE HYPOTHALAMIC-HYPOPHYSIAL PORTAL SYSTEM
The communication between the hypothalamus and the anterior pituitary is vascular, through a specialized portal system:
- Superior Hypophysial Artery: Branches off the internal carotid artery and supplies the upper part of the pituitary stalk and the median eminence (the inferior extension of the hypothalamus).
- First Capillary Network (at the Median Eminence): These arteries form a primary capillary plexus in the median eminence, where neurosecretory neurons in the hypothalamus release their hypothalamic releasing and inhibitory hormones into the blood.
- Hypophysial Portal Vessels: These capillaries then coalesce to form the hypophysial portal veins, which descend along the pituitary stalk.
- Second Capillary Network (in the Anterior Pituitary): The portal veins branch into a secondary capillary plexus within the anterior pituitary. Here, the hypothalamic hormones diffuse out of the capillaries and act directly on the specific secretory cells of the anterior pituitary, stimulating or inhibiting their hormone release.
- Venous Flow to the Heart: The anterior pituitary hormones then enter the systemic circulation via venous drainage to the heart, reaching their target organs.
This portal system ensures that the hypothalamic hormones reach the anterior pituitary in high concentrations before being diluted in the general circulation, allowing for precise control.
B. HYPOTHALAMIC CONTROL OF ANTERIOR PITUITARY SECRETIONS: RELEASING & INHIBITING HORMONES
The hypothalamus secretes a number of peptide hormones, collectively known as "hypothalamic releasing hormones" and "hypothalamic inhibitory hormones," which directly regulate the secretion of anterior pituitary hormones. Each anterior pituitary hormone generally has at least one hypothalamic regulatory hormone.
Here are some key hypothalamic nuclei and the hormones they release:
| NUCLEUS | HORMONE RELEASED |
|---|---|
| Pre-Optic Nucleus | Gonadotropin-Releasing Hormone (GnRH): Stimulates LH and FSH release. |
| Ventromedial Nucleus |
Growth Hormone-Releasing Hormone (GHRH): Stimulates GH release. Somatostatin (Growth Hormone-Inhibiting Hormone, GHIH): Inhibits GH release. |
| Paraventricular Nucleus |
Oxytocin (90%) - ADH (10%): Synthesizes these, which are released from posterior pituitary. Thyrotropin-Releasing Hormone (TRH): Stimulates TSH and Prolactin release. |
| Arcuate Nucleus | Prolactin-Inhibiting Factor (PIF), which is Dopamine: Inhibits Prolactin release. |
| Supra-Optic Nucleus | ADH (90%) - Oxytocin (10%): Synthesizes these, which are released from posterior pituitary. |
RELATIONSHIP WITH THE POSTERIOR PITUITARY GLAND
Unlike the anterior pituitary, which communicates via a vascular portal system, the posterior pituitary has a direct neural connection with the hypothalamus. This makes the posterior pituitary essentially an extension of the brain itself.
- Neural Connection: The posterior pituitary gland is connected to the hypothalamus by unmyelinated nerve fibers. These nerve fibers form the hypothalamohypophysial tract.
- Location of Hormone Synthesis: The cell bodies of the neurons that produce the posterior pituitary hormones are located in specific nuclei within the hypothalamus:
- Supraoptic Nucleus: Primarily responsible for synthesizing Antidiuretic Hormone (ADH), also known as Vasopressin.
- Paraventricular Nucleus: Primarily responsible for synthesizing Oxytocin. It is crucial to understand that these hormones are synthesized in the hypothalamus, NOT in the posterior pituitary gland itself.
- Axonal Transport: The nerve fibers (axons) from these hypothalamic nuclei extend down through the infundibulum (pituitary stalk), alongside small glial-like cells called pituicytes, into the posterior pituitary.
- Storage and Release: The synthesized hormones (ADH and Oxytocin) are then transported down these axons by axoplasmic flow to the nerve terminals located in the posterior pituitary gland. They are stored in secretory granules within these nerve terminals until an appropriate stimulus triggers their release directly into the bloodstream.
In summary, the posterior pituitary gland acts as a storage and release site for hormones that are secreted (synthesized) from the hypothalamus. It does not produce its own hormones. This direct neural pathway allows for rapid and precise release of ADH and Oxytocin in response to hypothalamic signals.
ANTERIOR PITUITARY GLAND HORMONES
The anterior pituitary gland secretes a variety of hormones that are often referred to as trophic hormones. The term 'trophic' (from Greek trophos, meaning "to feed" or "nourish") signifies their role in stimulating the growth, development, and function of other endocrine glands or target tissues.
- High [Hormone]: A consistently high concentration of a trophic hormone typically causes its target organ to hypertrophy (increase in size) and often leads to hyperfunction (increased activity).
- Low [Hormone]: Conversely, a consistently low concentration of a trophic hormone can cause its target organ to atrophy (decrease in size) and often leads to hypofunction (decreased activity).
These hormones are essential for orchestrating a wide range of physiological processes.
Hormones and Their Characteristics:
While all anterior pituitary hormones are crucial, some share structural and functional similarities.
- Structurally and Functionally Related Group:
- Growth Hormone (GH)
- Prolactin (PRL)
- Human Placental Lactogen (hPL) (Note: hPL is produced by the placenta, not the anterior pituitary, but shares structural and functional similarities with GH and Prolactin).
- Similar Alpha Peptide Units (Glycoproteins): These hormones are glycoproteins consisting of two subunits: an alpha subunit and a beta subunit. The alpha subunit is virtually identical across this group, while the beta subunit is different and confers hormone-specific biological activity.
- Thyroid-Stimulating Hormone (TSH)
- Follicle-Stimulating Hormone (FSH)
- Luteinizing Hormone (LH)
- Human Chorionic Gonadotropin (hCG) (Note: hCG is produced by the placenta, not the anterior pituitary, but is structurally similar to LH, FSH, and TSH).
The Seven Key Anterior Pituitary Hormones:
Let's look at the individual anterior pituitary hormones in more detail:
1. Thyroid-Stimulating Hormone (TSH), or Thyrotropin:
- Target Tissue: Primarily the thyroid gland (indirect).
- Function: Stimulates the thyroid gland to release thyroid hormones (T3 and T4).
- Regulation: Its production is influenced by stress (which can increase production) and, more importantly, by Thyrotropin-Releasing Hormone (TRH) from the hypothalamus and negative feedback from thyroid hormones.
2. Adrenocorticotropic Hormone (ACTH), or Corticotropin:
- Target Tissue: The adrenal cortex.
- Function: Stimulates the release of steroid hormones by the adrenal glands. Specifically targets cells producing glucocorticoids (like cortisol), which affect glucose metabolism, stress response, and immune function.
- Regulation: Heavily influenced by stress (which increases its production) and by Corticotropin-Releasing Hormone (CRH) from the hypothalamus.
3. Follicle-Stimulating Hormone (FSH):
- Target Tissue: The gonads (ovaries in females, testes in males).
- Function (Females): Promotes egg development within ovarian follicles and stimulates the secretion of estrogens by ovarian cells.
- Function (Males): Supports sperm production (spermatogenesis) in the testes.
- Regulation: Regulated by Gonadotropin-Releasing Hormone (GnRH) from the hypothalamus and negative feedback from gonadal steroids.
4. Luteinizing Hormone (LH):
- Target Tissue: The gonads (ovaries in females, testes in males).
- Function (Females): Induces ovulation (the release of a mature egg from the ovary) and promotes the ovarian secretion of estrogens and progesterone, which prepare the body for the possibility of pregnancy.
- Function (Males): Stimulates the interstitial cells (Leydig cells) in the testes to produce testosterone.
- Regulation: Regulated by Gonadotropin-Releasing Hormone (GnRH) from the hypothalamus and negative feedback from gonadal steroids.
- Note: Both FSH and LH are collectively known as Gonadotropic Hormones because they target the gonads.
5. Prolactin (PRL):
- Target Tissue: Primarily breast tissue (mammary glands).
- Function (Females): Stimulates the development of the mammary glands and, crucially, the production of milk (lactogenesis) after childbirth.
- Function (Males): Historically thought to have no effect on human males, but recent research suggests roles in immune function, prostate health, and reproductive behavior, though not milk production.
- Regulation: Its release is primarily under tonic inhibition by Dopamine (Prolactin-Inhibiting Factor, PIF) from the hypothalamus. Mechanical stimulation of breast tissue (nursing) causes a rapid increase in prolactin production by inhibiting dopamine release. High levels of sex hormones (estrogens) can cause sensitivity of breast tissue prior to the flow phase of the menstrual cycle, and prolactin works with other hormones to stimulate breast development.
6. Growth Hormone (GH), or Somatotropin:
- Target Tissue: Nearly all body cells, especially bone, cartilage, and muscle.
- Function: Stimulates cell growth and replication primarily by increasing the rate of protein synthesis. It also has metabolic effects: it promotes the breakdown of fat (lipolysis) and causes the liver to break down glycogen reserves, releasing glucose into the circulation, thus raising blood glucose levels (diabetogenic effect). Its growth-promoting effects are largely mediated by insulin-like growth factors (IGFs) produced by the liver and other tissues.
- Regulation: Regulated by Growth Hormone-Releasing Hormone (GHRH) and Somatostatin (GHIH) from the hypothalamus.
- Disorders of GH Secretion:
- Hyposecretion (too little GH):
- Children: Leads to pituitary dwarfism (or growth hormone deficiency). Affected individuals have normal body proportions but are usually no taller than 4 feet tall.
- Adults: Can lead to Simmond's disease, characterized by atrophy and premature aging, loss of lean body mass, and increased fat mass.
- Hypersecretion (too much GH):
- Children: Leads to pituitary gigantism, where individuals grow to extreme heights (8-9 feet tall) with generally normal body proportions.
- Adults: Leads to acromegaly, a condition characterized by:
- Widened bones and thick fingers/toes.
- Lengthening of the jaw and cheekbones.
- Thickened eyelids, lips, tongue, and nose.
- Enlargement of internal organs.
- Hyposecretion (too little GH):
7. Melanocyte-Stimulating Hormone (MSH):
- Target Tissue: The melanocytes in the epidermis (basal cell layer of the skin).
- Function: Stimulates the melanocytes of the skin, increasing their production of melanin, the pigment responsible for skin and hair color.
- Role in Humans: While very important in the control of skin and hair pigmentation in many animals, its physiological role in healthy adult humans is less clear. It is often released from the same precursor as ACTH. High levels are associated with certain conditions causing hyperpigmentation.
FEEDBACK CONTROL OF THE ANTERIOR PITUITARY
The secretion of anterior pituitary hormones is tightly regulated by negative feedback inhibition, primarily from the hormones secreted by their target glands. This ensures that hormone levels remain within a healthy physiological range.
Two Levels of Negative Feedback:
- Feedback at the Hypothalamus: The hormones secreted by the target glands (e.g., thyroid hormones, cortisol, gonadal steroids) can act directly on the hypothalamus to inhibit the secretion of its releasing hormones. For example, high thyroid hormone levels inhibit TRH release.
- Feedback at the Anterior Pituitary: The target gland hormones can also act directly on the anterior pituitary to inhibit its response to the hypothalamic releasing hormone, thereby reducing the secretion of the trophic hormone. For example, high thyroid hormone levels inhibit the pituitary's response to TRH.
POSTERIOR PITUITARY HORMONES
As discussed, the posterior pituitary does not synthesize hormones but stores and releases two neurohormones produced by the hypothalamus:
1. Antidiuretic Hormone (ADH), or Vasopressin:
- Synthesis Site: Primarily the supraoptic nucleus of the hypothalamus.
- Function:
- Antidiuretic Effect: Its primary role is to conserve water during urine formation by the kidney nephrons. It increases the permeability of the renal collecting ducts to water, allowing more water to be reabsorbed back into the bloodstream, thus reducing urine volume and concentrating the urine.
- Vasopressor Effect: At high concentrations (e.g., during severe hemorrhage), ADH also causes contraction of arteriolar smooth muscle, leading to vasoconstriction and an increase in blood pressure (hence the name vasopressin).
- Regulation: Released in response to increased plasma osmolality (too concentrated blood) or decreased blood volume/pressure.
2. Oxytocin:
- Synthesis Site: Primarily the paraventricular nucleus of the hypothalamus.
- Function:
- Uterine Contraction: Stimulates the powerful contraction of uterine smooth muscle during childbirth, helping to expel the infant. Its release is stimulated by stretching of the cervix (Ferguson reflex).
- Milk Ejection (Let-down Reflex): Promotes the ejection (let-down) of milk from the mammary glands during breast-feeding in response to suckling. It contracts myoepithelial cells surrounding milk ducts.
- Social Bonding: Often referred to as the "love hormone" or "bonding hormone." It plays roles in social recognition, pair bonding, maternal-infant bonding, and other social behaviors.
These two hormones, though produced in the hypothalamus, are indispensable functions carried out by the posterior pituitary.
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Hypothalamus & Pituitary Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Hypothalamus & Pituitary Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Pancreas Physiology
Pancreas Physiology
THE PANCREAS
The pancreas is an elongated, flattened gland located deep in the abdomen, nestled between the stomach and the spine. It is a unique organ because it serves two vital functions:
- Exocrine Function: The vast majority (about 98-99%) of the pancreas is dedicated to its exocrine role. It produces digestive enzymes (e.g., amylase, lipase, proteases) that are secreted into the duodenum via a system of ducts to aid in the digestion of carbohydrates, fats, and proteins in the small intestine.
- Endocrine Function: The remaining 1-2% of the pancreas is composed of specialized clusters of cells called the islets of Langerhans. These islets are hormone-producing factories that secrete critical hormones directly into the bloodstream to regulate blood glucose levels.
I. ENDOCRINE FUNCTION OF THE PANCREAS: THE ISLETS OF LANGERHANS
The endocrine pancreas is composed of approximately 1 to 2 million islets of Langerhans, which collectively constitute only about 1-2% of the total pancreatic mass. Despite their small proportion, these islets are vital for maintaining metabolic homeostasis, especially blood glucose regulation.
Structure of the Islets:
- Each islet is a microscopic cluster of endocrine cells, typically 0.3 mm in diameter.
- They are highly vascularized and organized around small capillaries into which their cells directly secrete their hormones, allowing for rapid systemic distribution.
- The cells within each islet are in close proximity to each other, facilitating paracrine communication (local hormone action between neighboring cells).
Major Cell Types within the Islets:
The islets contain several distinct cell types, each producing specific hormones. These cells are distinguished by their morphological and staining characteristics.
1. Alpha (α) cells
- Constitute about 25% of the islet cells.
- Typically located at the periphery of the islets.
- Their primary secretion is glucagon, often referred to as the "fasting hormone."
2. Beta (β) cells
- Are the most abundant, making up about 60% of the islet cells.
- Predominantly located in the center of the islets.
- Their primary secretions are insulin (the "feasting hormone") and amylin. Amylin is co-secreted with insulin and helps regulate glucose homeostasis by slowing gastric emptying and promoting satiety.
3. Delta (δ) cells
- Comprise about 10% of the islet cells.
- Distributed around the periphery of the islets.
- They secrete somatostatin, a hormone that acts locally (paracrine effect) to inhibit the secretion of both insulin and glucagon, thus modulating their release. It also has systemic effects, inhibiting growth hormone and TSH from the pituitary.
4. PP cells (or Gamma (γ) cells)
- Present in small numbers (less than 1%).
- They secrete pancreatic polypeptide, whose exact physiological role is still not entirely clear but is thought to be involved in regulating pancreatic secretion and satiety.
II. BLOOD GLUCOSE HOMEOSTASIS:
The primary role of the endocrine pancreas is to maintain blood glucose homoeostasis (or homeokinesis), ensuring that blood glucose levels remain within a narrow, healthy range despite variations in food intake and energy expenditure. This delicate balance is achieved through the coordinated actions of insulin and glucagon.
Outline of Glucose Regulation:
- The "Feasting" Hormone - Insulin: Primarily secreted after meals, when blood glucose is high, to promote glucose uptake and storage.
- The "Fasting" Hormone - Glucagon: Primarily secreted during fasting or between meals, when blood glucose is low, to raise blood glucose levels.
- Integrated Hormonal Control of Blood Glucose Concentration: The interplay between insulin, glucagon, and other hormones (like somatostatin, amylin, and gut hormones) orchestrates the precise regulation of glucose.
- Diabetes, Pathophysiological Changes: Disruptions in this hormonal control, particularly insulin function, lead to diabetes mellitus, a chronic metabolic disorder.
III. INSULIN AND ITS METABOLIC EFFECTS
Insulin is the most important anabolic hormone in the body, primarily responsible for lowering blood glucose levels after a meal and promoting energy storage.
Structure and Synthesis:
- Insulin is a small protein hormone (peptide hormone) with a molecular weight of 5808.
- It consists of two peptide chains (A and B chains) linked together by two disulfide bonds. This specific structure is crucial for its biological activity.
- It is secreted by the beta (β) cells of the pancreatic islets via exocytosis, primarily in response to elevated blood glucose levels (a signal of "energy abundance").
Half-Life and Degradation:
- The "half-life" of insulin in circulation is remarkably short, about 6 minutes, meaning half of the secreted insulin is cleared from the bloodstream within this time.
- Consequently, it is cleared completely from the blood in approximately 10-15 minutes. This rapid turnover allows for precise and dynamic control of blood glucose levels.
- Insulin is primarily degraded by the enzyme insulinase, which is found abundantly in the liver and kidneys, and to a lesser extent in muscles and most other tissues.
Role in Blood Glucose Maintenance:
- Insulin maintains blood glucose levels close to 5 mmol/L (90 mg/dL) under fasting conditions.
- After a meal, blood glucose levels may temporarily rise as high as 8 mmol/L (144 mg/dL), but insulin rapidly brings these levels back down to the normal range.
- Overweight and obese individuals often exhibit higher basal levels of insulin (hyperinsulinemia) as a compensatory mechanism to overcome insulin resistance, where their cells do not respond as effectively to insulin.
IV. SYNTHESIS OF INSULIN:
Insulin synthesis is a classic example of protein synthesis for secretion, involving several steps within the β-cells:
- Preproinsulin Formation: Insulin RNA, transcribed from the insulin gene, is translated by ribosomes attached to the endoplasmic reticulum (ER). The initial product is a larger precursor molecule called preproinsulin (molecular weight = 11,500). This molecule contains a signal peptide that directs it into the ER lumen.
- Proinsulin Formation: Inside the ER, the signal peptide is cleaved off, converting preproinsulin into proinsulin (molecular weight = 9,000). Proinsulin consists of the A and B chains linked by a C-peptide.
- Insulin and C-peptide Cleavage: Proinsulin then travels to the Golgi apparatus. Here, specific enzymes (prohormone convertases) cleave the proinsulin molecule, removing the C-peptide and leaving behind the mature, active insulin molecule (A and B chains linked by disulfide bonds) and the peptide fragments (C-peptide).
- Packaging and Secretion: Both insulin and C-peptide are then packaged together into secretory granules. These granules await the appropriate signal (primarily elevated blood glucose) to be released into the bloodstream via exocytosis.
V. INSULIN AND TARGET RECEPTORS: MECHANISM OF ACTION
Insulin exerts its diverse metabolic effects by binding to specific receptors on the surface of target cells.
Insulin Receptor Structure:
- The insulin receptor is a large, complex transmembrane glycoprotein with a molecular weight of approximately 300,000.
- It is composed of four subunits linked by disulfide bonds:
- Two alpha (α) subunits: These are entirely located outside the cell membrane. They are the primary binding sites for insulin.
- Two beta (β) subunits: These penetrate through the cell membrane and extend into the cytoplasm. They contain the intracellular signaling machinery.
Mechanism of Action - Receptor Activation:
- Insulin Binding: When insulin binds to the alpha subunits of the receptor, it induces a conformational change in the entire receptor complex.
- Autophosphorylation: This conformational change leads to the autophosphorylation of tyrosine residues on the beta subunits within the cytoplasm. This autophosphorylation event is crucial, as it activates the intrinsic tyrosine kinase activity of the beta subunits.
- Tyrosine Kinase Activity and IRS Proteins: The activated tyrosine kinase then phosphorylates multiple other intracellular proteins and enzymes, notably a group of molecules called Insulin Receptor Substrates (IRS) proteins.
- Signal Transduction Cascade: Phosphorylated IRS proteins then serve as docking sites for other signaling molecules, initiating a complex intracellular signal transduction cascade involving various kinases and phosphatases (e.g., PI3K/Akt pathway, MAPK pathway).
- Desired Metabolic Effects: Through these downstream signaling pathways, insulin ultimately directs the intracellular metabolic machinery to produce its wide array of desired effects on carbohydrate, fat, and protein metabolism within target cells.
Key Principle: It is important to note that it is the activated receptor, not the insulin itself, that directly causes the subsequent intracellular effects. Insulin acts as the first messenger, initiating a cascade of events inside the cell.
VI. EFFECTS OF INSULIN:
Insulin is the primary anabolic hormone of the body, meaning it promotes the synthesis of complex molecules from simpler ones, thereby facilitating the storage of energy. Its main role is to facilitate the uptake, utilization, and storage of glucose, amino acids, and fatty acids, thereby lowering blood glucose levels and directing nutrient partitioning after a meal. Its effects are visible across several key metabolic pathways:
- Carbohydrate Metabolism
- Fat Metabolism
- Protein Metabolism and Growth
A. EFFECTS OF INSULIN ON CARBOHYDRATE METABOLISM
The most immediate and well-known effect of insulin is its role in managing blood glucose.
Post-Meal Glucose Handling:
After a high-carbohydrate meal, the glucose absorbed into the bloodstream triggers a rapid secretion of insulin from the pancreatic β-cells. Insulin, in turn, causes the rapid uptake, storage, and utilization of this glucose by almost all tissues of the body, most notably by the muscles, adipose tissue, and liver.
Glucose Storage in Muscles:
- Skeletal muscle is a significant storage site for glucose in the form of glycogen.
- Insulin promotes the transport of glucose into muscle cells. Once inside, glucose is polymerized into glycogen by the enzyme glycogen synthase.
- Muscles can store up to 2-3% of their mass as glycogen. This stored glycogen serves as a readily available energy source for muscle contraction, especially during exercise. Unlike liver glycogen, muscle glycogen cannot directly raise blood glucose as muscle cells lack glucose-6-phosphatase.
Glucose Handling in the Liver:
The liver plays a central role in buffering blood glucose levels, and insulin significantly influences its actions. The liver can store up to 60% of the glucose absorbed after a meal.
- Inactivation of Liver Phosphorylase: Insulin acts to inactivate liver phosphorylase, the enzyme responsible for breaking down stored glycogen into glucose (glycogenolysis). By inhibiting this enzyme, insulin prevents the breakdown of the glycogen that has already been stored in the liver cells, thus ensuring that glucose remains stored.
- Enhanced Glucose Uptake by Increased Glucokinase Activity: Insulin causes enhanced uptake of glucose from the blood by liver cells. It does this, in part, by increasing the activity of glucokinase. Glucokinase phosphorylates glucose to glucose-6-phosphate, effectively "trapping" glucose inside the liver cells and maintaining a concentration gradient that favors further glucose entry.
- Increased Glycogen Synthase Activity: Insulin significantly increases the activity of glycogen synthase, the enzyme responsible for polymerization of the monosaccharide units (glucose) into glycogen molecules.
- Result: Through these mechanisms, insulin can lead to the storage of a substantial amount of glycogen in the liver, often up to 5-6% of the liver's mass, translating to approximately 100 grams of stored glycogen in an adult.
- Reversal during Fasting: When blood glucose levels fall (e.g., during fasting), the reverse events happen: insulin levels decrease, liver phosphorylase becomes active, and glycogen is broken down to release glucose into the blood.
Conversion of Excess Glucose to Fatty Acids:
- When the liver has stored its maximum capacity of glycogen, insulin plays a crucial role in preventing excessive hyperglycemia.
- Insulin promotes the conversion of all the excess glucose into fatty acids.
- These fatty acids are then packaged as triglycerides in very low-density lipoproteins (VLDLs), which are subsequently released into the bloodstream and transported to adipose tissue for storage as fat.
Glucose Use by the Brain:
- Insulin has little direct effect on the uptake or use of glucose by the brain.
- Instead, brain cells are highly permeable to glucose and can utilize glucose without the intermediation of insulin. This ensures a constant supply of glucose to the brain, which relies almost exclusively on glucose for energy.
- Vulnerability to Hypoglycemia: However, because the brain is so dependent on glucose, when blood glucose falls too low (e.g., into the range of 20 to 50 mg/100 ml / 1.1-2.8 mmol/L), symptoms of hypoglycemic shock develop. This is characterized by progressive nervous irritability that can lead to fainting, seizures, and even coma, highlighting the brain's sensitivity to glucose deprivation.
B. EFFECT OF INSULIN ON FAT METABOLISM
While less immediately visible than its effects on carbohydrate metabolism, insulin's role in fat metabolism is equally vital for energy storage and overall metabolic health. Insulin is a powerful anti-lipolytic (inhibits fat breakdown) and pro-lipogenic (promotes fat synthesis) hormone.
Insulin Promotes Fat Synthesis and Storage:
- Insulin increases the utilization of glucose by most of the body's tissues, which in turn spares fats from being used as an energy source.
- Mechanisms:
- Increased Glucose Transport into Liver Cells: As discussed, insulin increases glucose uptake by the liver.
- Conversion to Fatty Acids: Excess glucose in the liver is first metabolized through the glycolytic pathway to pyruvate. Pyruvate is subsequently converted into acetyl-CoA, which is the fundamental substrate from which fatty acids are synthesized. Insulin promotes the activity of enzymes involved in fatty acid synthesis.
- Triglyceride Synthesis in Adipose Tissue: Insulin actively promotes the synthesis of triglycerides within adipose (fat) cells. It does this by:
- Increasing the transport of glucose into adipocytes, which provides glycerol-3-phosphate, the backbone for triglyceride synthesis.
- Activating lipoprotein lipase on capillary walls, which breaks down circulating triglycerides (e.g., from VLDLs) into fatty acids and glycerol, allowing their uptake by adipocytes.
- Inhibiting hormone-sensitive lipase, the enzyme responsible for breaking down stored triglycerides.
Insulin Deficiency on Fat Metabolism:
The absence of insulin reverses all these effects that cause storage of fat, leading to significant metabolic derangements.
- Lipolysis of Storage Fat and Release of Free Fatty Acids (FFA):
- In the absence of insulin, the enzyme hormone-sensitive lipase in fat cells becomes strongly activated.
- This enzyme causes the rapid hydrolysis (breakdown) of stored triglycerides, releasing large quantities of fatty acids and glycerol into the circulating blood.
- These fatty acids are then utilized for energy by almost all tissues except the brain. This shift to fat metabolism is a compensatory mechanism during glucose scarcity.
- Increased Plasma Cholesterol and Phospholipid Concentrations:
- The liver, faced with an abundance of fatty acids, converts some of them into phospholipids and cholesterol.
- These lipids are then packaged and released into the blood in the form of lipoproteins.
- Chronically high concentrations of cholesterol and other lipids in the blood promote the development of atherosclerosis (hardening of the arteries), which is a serious complication in poorly controlled diabetes.
- Ketosis and Acidosis (Diabetic Ketoacidosis - DKA):
- When large quantities of fatty acids are mobilized from adipose tissue, the liver's capacity to metabolize them completely can be overwhelmed.
- The carnitine transport mechanism for transporting fatty acids into the mitochondria (where they are oxidized) becomes increasingly activated.
- In the liver, excessive fatty acid oxidation leads to the production of large amounts of acetyl-CoA. When the rate of acetyl-CoA production exceeds the capacity of the citric acid cycle, it is shunted towards the formation of ketone bodies (acetoacetate, β-hydroxybutyrate, and acetone).
- These ketone bodies are acidic. Their excessive production and accumulation in the blood lead to ketosis and, if severe enough, metabolic acidosis, a life-threatening condition known as diabetic ketoacidosis (DKA), particularly common in Type 1 diabetes.
C. EFFECT OF INSULIN ON PROTEIN METABOLISM AND ON GROWTH
Insulin is profoundly anabolic for protein, promoting protein synthesis and storage, which is essential for growth and tissue repair.
Insulin Promotes Synthesis and Storage of Proteins:
- While the full mechanism is complex, several key facts are known:
- Increased Amino Acid Transport: Insulin significantly increases the active transport of many amino acids from the blood into various cells, especially muscle cells. This makes more building blocks available for protein synthesis.
- Increased Translation of mRNA: Insulin increases the rate of translation of messenger RNA (mRNA), leading to the formation of new proteins by ribosomes.
- Increased Transcription of DNA: Insulin also increases the rate of transcription of selected DNA genetic sequences in the cell nuclei, leading to increased synthesis of specific mRNAs and thus specific proteins.
- Inhibition of Protein Catabolism: Insulin inhibits the catabolism (breakdown) of proteins, thereby reducing the rate of amino acid release from cells.
- Depression of Gluconeogenesis in the Liver: In the liver, insulin depresses the rate of gluconeogenesis (the formation of glucose from non-carbohydrate sources like amino acids). By reducing the use of amino acids for glucose production, insulin spares them for protein synthesis.
- Synergistic with Growth Hormone: Insulin works synergistically with growth hormone to promote growth. Both hormones are necessary for normal growth, highlighting insulin's role beyond just glucose regulation.
Deficiency of Insulin on Proteins:
A lack of insulin has devastating effects on protein metabolism.
- Cessation of Protein Storage: All protein storage virtually comes to a halt when insulin is not available.
- Increased Protein Catabolism: The catabolism of proteins dramatically increases, while protein synthesis stops. This leads to the rapid breakdown of muscle and other tissue proteins.
- Elevated Plasma Amino Acids: Large quantities of amino acids are "dumped" into the plasma from the breakdown of tissue proteins, causing plasma amino acid concentration to rise considerably.
- Amino Acids for Energy and Gluconeogenesis: Most of this excess amino acid pool is then either used directly for energy or, more significantly, as substrates for gluconeogenesis in the liver. This contributes to the hyperglycemia seen in insulin deficiency.
- Enhanced Urea Excretion: The degradation of amino acids for energy or glucose production results in the formation of ammonia, which is then converted to urea in the liver and excreted in the urine. This leads to enhanced urea excretion.
- Protein Wasting: The resulting protein wasting is one of the most serious and debilitating effects of severe diabetes mellitus. It can lead to extreme weakness, muscle atrophy, impaired wound healing, and many other deranged functions of organs throughout the body, underscoring the critical importance of insulin for maintaining body mass and function.
VII. GLUCAGON AND ITS FUNCTIONS:
Glucagon is a vital hormone secreted by the alpha (α) cells of the islets of Langerhans. Its primary role is to prevent blood glucose levels from falling too low, especially during periods of fasting or when glucose availability is scarce. Its actions are largely diametrically opposed to those of insulin.
- Secretion Stimulus: Glucagon is secreted primarily in response to a fall in blood glucose concentration (hypoglycemia).
- Structure: Glucagon is a polypeptide hormone with a molecular weight of 3485. It is composed of a single chain of 29 amino acids.
- Effectiveness: Glucagon is an incredibly potent hormone. Even a small amount, such as 1 mg/kg of glucagon, can increase blood glucose concentration by about 20 mg/100 ml (1.1 mmol/L). This potent glucose-raising effect is why glucagon is often referred to as a hyperglycemic hormone.
A. MAJOR EFFECTS OF GLUCAGON: RAISING BLOOD GLUCOSE
Glucagon's main physiological functions are centered on increasing the availability of glucose to the body's cells, particularly those that are highly glucose-dependent (like the brain). It achieves this through two major mechanisms, primarily in the liver:
- Breakdown of liver glycogen (Glycogenolysis): Glucagon rapidly mobilizes stored glucose from the liver.
- Increased gluconeogenesis in the liver: Glucagon stimulates the liver to produce new glucose from non-carbohydrate sources.
Both of these effects greatly enhance the availability of glucose to the other organs of the body, preventing hypoglycemia.
1. Glycogenolysis (Breakdown of Liver Glycogen):
Glucagon initiates a complex cascade of events within the liver cells (hepatocytes) that rapidly leads to the breakdown of glycogen:
- Glucagon first binds to specific G protein-coupled receptors on the surface of the hepatic cell membrane.
- This binding activates adenylyl cyclase (also known as adenylate cyclase), an enzyme located on the inner surface of the cell membrane.
- Adenylyl cyclase then catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP), which acts as a second messenger.
- cAMP then activates a protein kinase regulator protein (specifically, protein kinase A, or PKA).
- Activated PKA then activates another enzyme called phosphorylase b kinase.
- Phosphorylase b kinase then converts the inactive form, phosphorylase b, into its active form, phosphorylase a.
- Phosphorylase a is the enzyme that promotes the degradation of glycogen into glucose-1-phosphate.
- Glucose-1-phosphate is then converted to glucose-6-phosphate, which is subsequently dephosphorylated by glucose-6-phosphatase. This crucial enzyme is present in the liver (and kidneys) but not in muscle, allowing the glucose to be released from the liver cells directly into the bloodstream.
2. Gluconeogenesis (Formation of New Glucose):
Glucagon also significantly increases the liver's capacity to synthesize new glucose from non-carbohydrate precursors.
- Glucagon increases the rate of amino acid uptake by the liver cells.
- It then promotes the conversion of many of these amino acids into glucose.
- This is achieved by activating multiple enzymes that are required for amino acid transport and the gluconeogenic pathway. Notably, it activates the enzyme system for converting pyruvate to phosphoenolpyruvate, which is a critical, rate-limiting step in gluconeogenesis. By enhancing this step, glucagon effectively ramps up the liver's glucose production.
B. OTHER EFFECTS OF GLUCAGON
While its primary role is on glucose metabolism, glucagon can have other effects, though these often require very high concentrations of glucagon, usually above the normal physiological maximum found in blood, and may be observed in pharmacological doses or specific pathological conditions.
- Activates adipose cell lipase: Glucagon directly stimulates hormone-sensitive lipase in adipose tissue, leading to the breakdown of stored triglycerides and making increased quantities of fatty acids available to the energy systems of the body. This provides an alternative fuel source when glucose is scarce.
- Inhibits the storage of triglycerides in the liver: Glucagon counteracts insulin's effect by preventing the liver from synthesizing and storing triglycerides, thus also preventing the liver from removing fatty acids from the blood.
- Enhances the strength of the heart: At high concentrations, glucagon can have a positive inotropic effect, increasing myocardial contractility.
- Increases blood flow in some tissues: This effect is especially noted in the kidneys.
- Enhances bile secretion:
- Inhibits gastric acid secretion: These gastrointestinal effects suggest a broader role in coordinating digestion and metabolism.
C. REGULATION OF GLUCAGON SECRETION
The secretion of glucagon is tightly regulated to ensure appropriate glucose homeostasis.
- Blood Glucose Concentration:
- The most potent regulator: Decreasing blood glucose concentration (hypoglycemia) is the primary stimulus for glucagon release.
- Conversely, increasing blood glucose to hyperglycemic levels decreases plasma glucagon secretion (and stimulates insulin release). This inverse relationship with insulin is central to glucose control.
- Amino Acids:
- Increased blood amino acids stimulate glucagon release. This is particularly important after a protein-rich meal, where glucagon can then promote the rapid conversion of these amino acids to glucose (gluconeogenesis) in the liver, preventing post-meal hypoglycemia that might occur if insulin were to act unopposed.
- Exercise:
- In exhaustive exercise, the blood concentration of glucagon often increases fourfold to fivefold. This helps to mobilize glucose from the liver to provide fuel for working muscles and maintain blood glucose levels during prolonged physical activity.
- Stress: Stress hormones like catecholamines (epinephrine, norepinephrine) can also stimulate glucagon release.
VIII. SOMATOSTATIN: THE MODULATOR
Somatostatin is another important polypeptide hormone secreted by the delta (δ) cells of the islets of Langerhans. While less prominent than insulin and glucagon, it plays a crucial role in regulating their secretion and overall nutrient assimilation.
- Structure: Somatostatin is a polypeptide containing only 14 amino acids.
- Half-Life: It has an extremely short half-life of only 3 minutes in the circulating blood, indicating its role as a local regulator.
- Release Stimulus: Somatostatin release is stimulated by several factors associated with nutrient absorption after a meal:
- Increased blood glucose
- Increased amino acids
- Increased fatty acids
- Increased concentrations of several of the gastrointestinal tract (GIT) hormones released from the upper GIT due to food intake (e.g., gastrin, secretin, CCK).
A. EFFECTS OF SOMATOSTATIN
Somatostatin exerts multiple inhibitory effects:
- Local (Paracrine) Action within the Islets: Somatostatin acts locally within the islets of Langerhans themselves to depress the secretion of both insulin and glucagon. By doing so, it serves as a paracrine regulator, dampening the fluctuations in insulin and glucagon secretion and ensuring a more controlled and prolonged absorption of nutrients.
- Decreases GI Motility: It decreases the motility of the stomach, duodenum, and gallbladder, thereby slowing the rate of food passage through the gastrointestinal tract.
- Decreases GI Secretion and Absorption: It decreases both secretion and absorption in the gastrointestinal tract. This includes inhibition of gastric acid secretion, pancreatic enzyme secretion, and intestinal absorption of nutrients.
By slowing down the rate of nutrient digestion, absorption, and subsequent utilization, somatostatin helps to extend the period over which nutrients are assimilated, preventing rapid surges and drops in blood nutrient levels.
IX. WHY IS GLUCOSE REGULATION IMPORTANT?
A common question is why such elaborate mechanisms exist to regulate glucose, given that the body can use other substrates (proteins and fats) as energy sources. The answer lies in the unique energy requirements of certain critical tissues:
- Glucose is the only nutrient that normally can be used by the brain, retina, and germinal epithelium of the gonads in sufficient quantities to supply them optimally with their required energy.
- The brain is particularly vulnerable. It relies almost exclusively on glucose for its energy needs because fatty acids generally cannot cross the blood-brain barrier efficiently. While ketone bodies can serve as an alternative fuel during prolonged starvation, they do not fully meet the brain's energy demands under normal conditions.
- The retina and germinal epithelium of the gonads also have a high dependency on glucose.
- Most of the glucose formed by gluconeogenesis during the interdigestive period (between meals) is used for metabolism in the brain. This highlights the absolute priority the body places on ensuring a constant supply of glucose to the brain, even at the expense of breaking down proteins and fats.
X. DIABETES MELLITUS:
Diabetes Mellitus is a chronic metabolic syndrome characterized by impaired carbohydrate, fat, and protein metabolism. It fundamentally stems from either:
- A lack of insulin secretion (absolute deficiency)
- Decreased sensitivity of the tissues to insulin (insulin resistance)
Under normal, healthy conditions, the fasting blood glucose concentration in a person (e.g., in the morning before breakfast) lies between 80 and 90 mg/100 ml of blood (4.4 to 5.0 mmol/L). Diabetes disrupts this tightly regulated balance, leading to chronically elevated blood glucose levels (hyperglycemia).
There are two general and major types of diabetes mellitus:
- Type 1 Diabetes Mellitus (T1DM): Formerly known as insulin-dependent diabetes mellitus (IDDM) or "juvenile diabetes." This type is primarily caused by an absolute lack of insulin secretion due to the destruction of pancreatic beta cells.
- Type 2 Diabetes Mellitus (T2DM): Formerly known as non-insulin-dependent diabetes mellitus (NIDDM) or "adult-onset diabetes." This type is characterized by insulin resistance, where target tissues do not respond properly to insulin, often coupled with a progressive decline in insulin secretion.
XI. TYPE 1 DIABETES MELLITUS (T1DM)
Type 1 diabetes is an autoimmune disease resulting in the destruction of insulin-producing beta cells.
- Onset: The onset of Type 1 diabetes typically occurs in childhood or adolescence, often around 14 years of age, hence its historical name "juvenile diabetes mellitus."
- Progression: It can develop very abruptly, often over a period of a few days or weeks, leading to significant metabolic disturbances.
- Principal Sequelae (Consequences): The fundamental issue in T1DM is the absolute absence of insulin, which leads to three principal metabolic sequelae:
- Increased Blood Glucose (Hyperglycemia): Due to the inability of cells to take up glucose and the liver's unchecked production of glucose.
- Increased Utilization of Fats for Energy and for Formation of Cholesterol by the Liver: In the absence of insulin, the body switches to fat metabolism for energy, leading to excessive breakdown of fats.
- Depletion of the Body's Proteins: Without insulin's anabolic effects, proteins are broken down for energy and gluconeogenesis.
- Etiology (Causes): The underlying causes of beta-cell destruction in T1DM are multifactorial:
- Pancreatic Beta Cell Unresponsiveness/Damage: The primary cause is the destruction of the beta cells themselves, rendering them unable to respond to glucose stimuli.
- Autoimmune Response: The most common cause is an autoimmune response where the body's immune system mistakenly attacks and destroys its own beta cells. Genetic predisposition plays a significant role in susceptibility to this autoimmune attack.
- Environmental Insult: Environmental factors, such as viral infections (e.g., coxsackievirus, rubella), are thought to trigger or accelerate the autoimmune process in genetically susceptible individuals.
- Genetic Defect: While not a single gene defect, certain genetic loci (especially HLA genes on chromosome 6) are strongly associated with an increased risk of T1DM.
- Pathophysiology: The lack of insulin drastically decreases the efficiency of peripheral glucose utilization (i.e., glucose uptake by muscle and fat cells) and augments glucose production by the liver (both glycogenolysis and gluconeogenesis proceed unchecked due to lack of insulin and relative excess of glucagon). This combination raises plasma glucose levels significantly, often to 300 to 1200 mg/100 ml (16.7 to 66.7 mmol/L). The increased plasma glucose then has multiple detrimental effects throughout the body.
Effects of Increased Plasma Glucose Concentration (Hyperglycemia) in T1DM:
The sustained elevation of blood glucose causes both acute and chronic complications:
- Cellular Dehydration (Osmotic Effects): Glucose is an osmotically active molecule. When the glucose concentration rises to excessive values in the extracellular fluid (ECF), it exerts a large amount of osmotic pressure. This draws water out of cells into the ECF, leading to cellular dehydration.
- Loss of Glucose in the Urine (Glycosuria): When blood glucose levels exceed the renal threshold (typically around 180-200 mg/100 ml or 10-11.1 mmol/L), the kidney tubules cannot reabsorb all the filtered glucose. This results in glucose appearing in the urine (glycosuria).
- Osmotic Diuresis and Fluid/Electrolyte Depletion: The presence of large amounts of glucose in the renal tubules creates an osmotic gradient, pulling water along with it. This leads to osmotic diuresis, characterized by greatly increased urine volume. This excessive urination (polyuria) can rapidly deplete the body of its fluids and electrolytes (e.g., sodium, potassium), leading to dehydration and electrolyte imbalances. Increased thirst (polydipsia) is a compensatory mechanism.
- Long-Term Tissue Damage (Microvascular and Macrovascular Complications): Chronically increased blood glucose levels are highly detrimental and can cause severe damage to many tissues and organs over time. This includes:
- Microvascular complications: Damage to small blood vessels, leading to retinopathy (eye damage, leading to blindness), nephropathy (kidney damage, leading to renal failure), and neuropathy (nerve damage, causing pain, numbness, and dysfunction in various organs).
- Macrovascular complications: Damage to large blood vessels, accelerating atherosclerosis, which increases the risk of heart attacks, strokes, and peripheral arterial disease.
- Increased Utilization of Fats and Metabolic Acidosis (Diabetic Ketoacidosis - DKA): As discussed previously, in the absence of insulin, the body breaks down fats excessively for energy. This leads to the overproduction of ketone bodies, resulting in metabolic acidosis (diabetic ketoacidosis), a life-threatening emergency.
- Depletion of Body's Proteins: Without insulin, proteins are catabolized, leading to muscle wasting, weakness, and impaired immune function. This constant breakdown of tissue contributes to weight loss despite increased food intake (polyphagia).
XII. TYPE 2 DIABETES MELLITUS (T2DM)
Type 2 diabetes is by far the more common form, accounting for the vast majority of diabetes cases.
- Prevalence: It accounts for about 90% of all cases of diabetes mellitus.
- Onset: The onset of T2DM typically occurs later in life, often after age 30, and frequently between the ages of 50 and 60 years. It develops gradually, often with subtle symptoms that can go unnoticed for years, hence its historical name "adult-onset diabetes." However, with rising obesity rates, T2DM is increasingly diagnosed in adolescents and even children.
- Cause: T2DM primarily results from resistance to the effects of insulin by target cells. This means that even though the pancreas may be producing insulin (sometimes even elevated amounts in the early stages), the body's cells (muscle, fat, liver) do not respond effectively to it. Over time, the pancreatic beta cells become exhausted trying to compensate for this resistance and their insulin secretion declines, leading to both insulin resistance and relative insulin deficiency.
- Obesity as a Major Risk Factor: Obesity is the most important risk factor for Type 2 diabetes in both children and adults. Excess adipose tissue, particularly visceral fat, releases inflammatory cytokines and free fatty acids that contribute to insulin resistance. Genetic predisposition also plays a significant role in T2DM.
Pathophysiology of Type 2 Diabetes:
The progression of T2DM often involves:
- Insulin Resistance: Target cells (muscle, liver, adipose tissue) fail to respond adequately to insulin. Glucose uptake by cells is impaired, and hepatic glucose production remains elevated.
- Compensatory Hyperinsulinemia: In the initial stages, the pancreatic beta cells try to compensate for insulin resistance by producing and secreting more insulin. This can keep blood glucose levels normal for a while, but it places a significant strain on the beta cells.
- Beta Cell Dysfunction and Failure: Over time, the pancreatic beta cells become exhausted and their ability to secrete sufficient insulin declines. This leads to progressive hyperglycemia.
- Glucagon Dysregulation: Often, there is also an inappropriate increase in glucagon secretion, further contributing to hyperglycemia by stimulating hepatic glucose production.
Management of Type 2 Diabetes:
Management typically begins with lifestyle modifications (diet, exercise, weight loss). If these are insufficient, oral medications are used to improve insulin sensitivity, stimulate insulin secretion, or reduce glucose absorption/production. Eventually, many individuals with T2DM may require insulin therapy as beta cell function declines.
XIII. INSULINOMA (HYPERINSULINISM)
While diabetes mellitus is characterized by insufficient insulin action, insulinoma represents the opposite extreme: excessive insulin production (hyperinsulinism).
- Cause: Insulinoma is much rarer than diabetes. It is typically caused by a tumor (adenoma) of the islet of Langerhans, specifically involving the beta cells.
- Malignancy: About 10 to 15 percent of these adenomas are malignant, and occasionally metastatic. These malignant tumors can cause tremendous production of insulin by both the primary and metastatic cancers.
- Consequences: The excessive insulin leads to severe and recurrent hypoglycemia (low blood glucose). This can be so profound that patients may require the administration of more than 1000 grams of glucose every 24 hours to prevent severe hypoglycemia.
- Insulin Shock: The most dangerous consequence of hyperinsulinism is insulin shock. This occurs because the brain is deprived of glucose, which is its primary (and often only) nutrient for energy. Symptoms can include confusion, dizziness, blurred vision, seizures, and ultimately coma, posing a significant risk of permanent brain damage or death if not promptly treated.
| Feature | Type I | Type II |
|---|---|---|
| Age at onset | Usually <20 years | Usually >30 years |
| Body mass | Low (wasted) to normal | Obese |
| Plasma insulin | Low or absent | Normal to high initially |
| Plasma glucagon | High, can be suppressed | High, resistant to suppression |
| Plasma glucose | Increased | Increased |
| Insulin sensitivity | Normal | Reduced |
| Therapy | Insulin | Weight loss, thiazolidinediones, metformin, sulfonylureas, insulin |
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Pancreas Physiology Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Pancreas Physiology Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Adrenal Gland Physiology
Adrenal Gland Physiology
ADRENAL GLAND FUNCTIONS AND REGULATION
The adrenal glands, also known as suprarenal glands, are a pair of small, triangular-shaped endocrine glands located on top of each kidney (ad-renal, meaning "near the kidney"). Despite their small size, these glands are absolutely vital for life, playing a central role in managing stress, regulating metabolism, blood pressure, fluid balance, and even influencing immune function and sexual development.
The adrenal gland is not a single, homogenous organ but rather a composite of two distinct endocrine glands, each with unique origins, structures, and hormonal secretions. These two compartments are:
- The Adrenal Cortex (Outer Layer)
- The Adrenal Medulla (Inner Layer)
These two regions, though anatomically juxtaposed, function almost as separate organs, producing different classes of hormones that contribute synergistically to the body's complex physiological responses.
I. THE ADRENAL GLAND: TWO DISTINCT COMPARTMENTS
A. The Adrenal Cortex (Outer Layer)
The adrenal cortex is the outer, yellowish layer of the adrenal gland, accounting for approximately 80-90% of the gland's total mass. It is derived from embryonic mesoderm and is responsible for synthesizing and secreting a class of steroid hormones known as corticosteroids. These hormones are all synthesized from cholesterol and are lipid-soluble, allowing them to easily pass through cell membranes to exert their effects.
The adrenal cortex is further subdivided into three distinct layers or zones, each characterized by its unique cellular structure, enzymatic machinery, and primary hormonal products. These layers are arranged concentrically, starting from the outermost layer just beneath the capsule, and moving inward towards the medulla:
1. Zona Glomerulosa (15% of the Cortex)
- Location: Outermost layer, directly beneath the adrenal capsule. Cells arranged in small, spherical or arched clusters (glomeruli).
- Primary Hormones: Sole site of mineralocorticoid synthesis, with aldosterone being the most important.
- Function of Aldosterone: Regulates mineral balance (sodium and potassium). Main actions on kidneys:
- Sodium (Na+) reabsorption: Leads to water retention, maintaining blood volume/pressure.
- Potassium (K+) excretion: Essential for maintaining normal potassium levels.
- Hydrogen (H+) excretion: Contributing to acid-base balance.
- Regulation: Primarily regulated by the Renin-Angiotensin-Aldosterone System (RAAS) and plasma potassium concentration. ACTH has a permissive role.
2. Zona Fasciculata (75% of the Cortex)
- Location: Middle and largest layer. Cells arranged in long, straight cords or fascicles. Cells are often called "spongiocytes" due to vacuolated appearance from abundant lipid droplets (cholesterol esters).
- Primary Hormones: Primary site of glucocorticoid synthesis, with cortisol (hydrocortisone) being the most important. Small amounts of adrenal androgens are also produced here.
- Function of Cortisol: Crucial "stress hormone".
- Glucose Metabolism: Promotes gluconeogenesis; decreases peripheral glucose utilization.
- Protein Metabolism: Increases protein catabolism (breakdown).
- Fat Metabolism: Promotes lipolysis.
- Anti-inflammatory/Immunosuppressive: Suppresses immune responses.
- Regulation: Primarily regulated by ACTH (from anterior pituitary), which is regulated by CRH (from hypothalamus). Forms the HPA axis.
3. Zona Reticularis (10% of the Cortex)
- Location: Innermost layer, adjacent to the medulla. Cells arranged in an irregular, anastomosing network (reticular).
- Primary Hormones: Primary site of adrenal androgen synthesis. Includes dehydroepiandrosterone (DHEA) and androstenedione (weak androgens). Some glucocorticoids produced here.
- Function of Androgens: Contribute to secondary sexual characteristics (pubic/axillary hair, libido). In males, masked by testicular androgens. In females, DHEA is a significant estrogen precursor.
- Regulation: Primarily regulated by ACTH.
B. The Adrenal Medulla (Inner Layer)
The adrenal medulla is the central, reddish-brown core of the adrenal gland, completely surrounded by the adrenal cortex. It comprises about 10-20% of the gland's total mass.
- Nature: Embryologically derived from neuroectoderm (neural crest cells), making it essentially a specialized sympathetic ganglion. Cells are called chromaffin cells (modified postganglionic sympathetic neurons).
- Innervation: Directly innervated by preganglionic sympathetic fibers (cholinergic). Allows rapid release of hormones in response to acute stress.
- Primary Hormones: Synthesizes and secretes catecholamines:
- Epinephrine (Adrenaline): ~80% of secretion.
- Norepinephrine (Noradrenaline): ~20% of secretion.
- Dopamine: Smaller amounts.
- Function: Central to the "fight-or-flight" response.
- Cardiovascular: Increased heart rate, contraction force, and blood pressure.
- Metabolic: Increased blood glucose (glycogenolysis/gluconeogenesis), lipolysis.
- Respiratory: Bronchodilation.
- Blood Flow: Shunting blood to muscles and brain.
- Regulation: Regulated by direct neural stimulation from the sympathetic nervous system in response to stress (pain, fear, hypoglycemia, etc.).
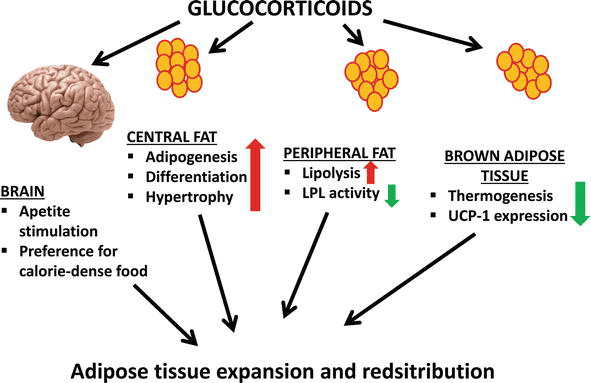
III. GLUCOCORTICOIDS: CORTISOL AND ITS ACTIONS
Glucocorticoids, with cortisol being the prime example, are powerful steroid hormones essential for life. Produced by the zona fasciculata and zona reticularis.
Regulation of Cortisol Secretion (HPA Axis):
- CRH (Corticotropin-Releasing Hormone): Secreted by the hypothalamus. Major regulator of ACTH secretion.
- ACTH (Adrenocorticotropic Hormone): Released from anterior pituitary. Stimulates cortisol synthesis/secretion.
- ADH (Vasopressin): A potent ACTH secretagogue, especially during stress.
- Pulsatile Secretion: Leads to diurnal (24-hour) variations. Peak activity is in the early morning (6-8 AM), diminishing to a nadir around midnight.
- Negative Feedback: Cortisol exerts negative feedback on both the hypothalamus (inhibiting CRH) and anterior pituitary (inhibiting ACTH).
Actions of Glucocorticoids (Cortisol):
1. Metabolic Response to Fasting (Anti-insulin Effects)
Cortisol promotes processes ensuring glucose availability during stress or fasting.
- Gluconeogenesis: Increases enzymes in the liver; mobilizes amino acids from muscle.
- Mobilization of Stored Fat: Promotes lipolysis, releasing free fatty acids (FFA) for energy and glycerol for gluconeogenesis.
- Muscle Cell: Promotes proteolysis (protein breakdown) releasing amino acids. Reduces protein synthesis.
- Fat Cell: Promotes lipolysis (releasing FFA and glycerol). Can promote lipogenesis in central areas.
- Liver Cell: Takes up amino acids/glycerol. Stimulates gluconeogenesis and glycogen synthesis.
- Overall Effect: Increase in circulating glucose (hyperglycemia) and free fatty acids (FFA).
2. Anti-inflammatory and Immunosuppressive Effects
Cortisol suppresses immune responses, inhibits pro-inflammatory cytokines, stabilizes lysosomal membranes, and decreases capillary permeability. Synthetic glucocorticoids are widely used as anti-inflammatory medications.
3. Other Important Actions
- Cardiovascular: Increases cardiac output; enhances vasoconstrictive effects of catecholamines (permissive action).
- Bone Metabolism: Chronic high levels inhibit bone formation, accelerate resorption (osteoporosis).
- Connective Tissue: Decreases collagen synthesis.
- CNS: Affects mood/behavior. High levels: insomnia, irritability, psychosis. Low levels: fatigue, depression.
- Gastrointestinal: Increases gastric acid secretion.
IV. ADRENAL FUNCTION ANOMALIES
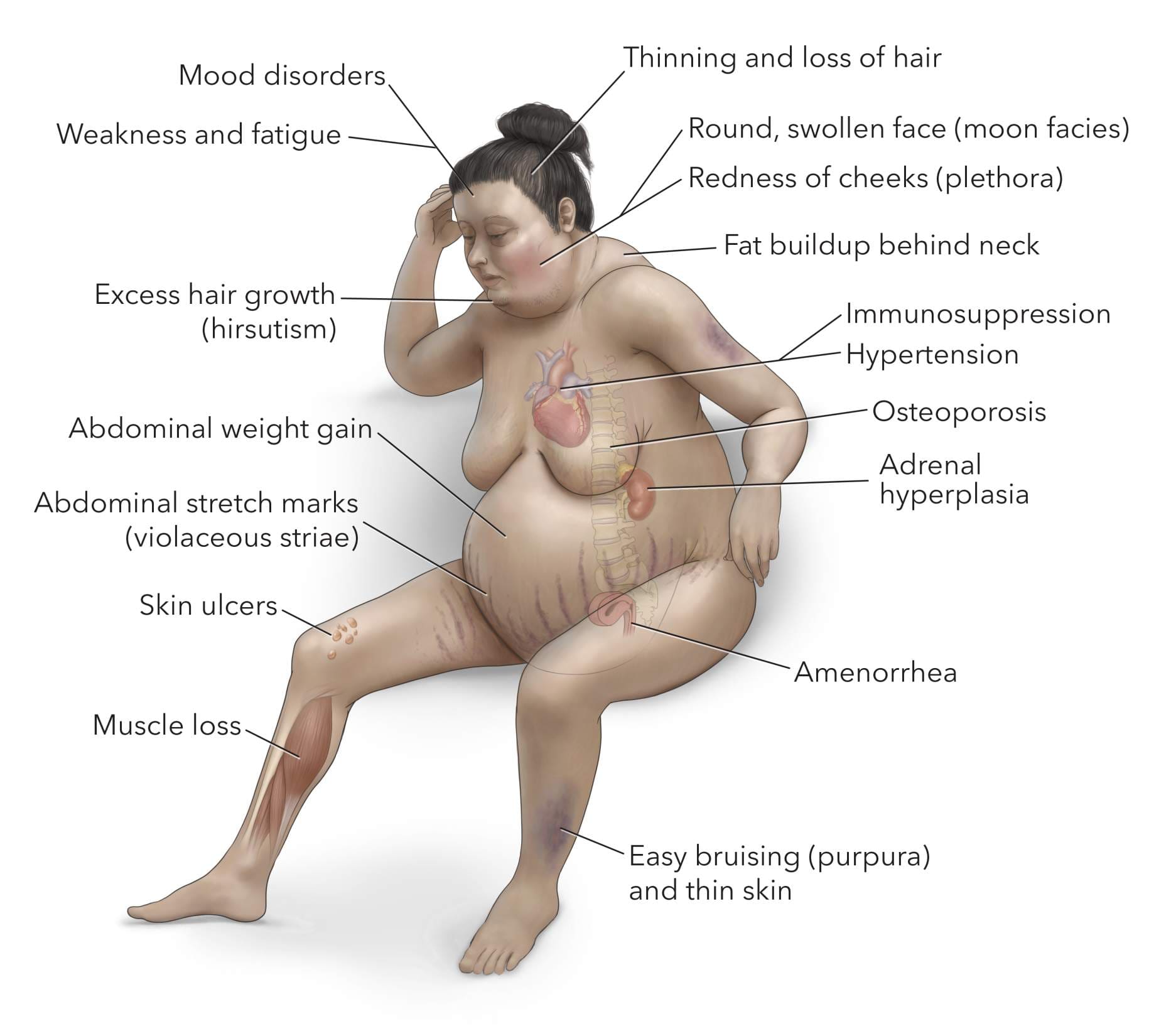
A. CUSHING'S SYNDROME: EXCESSIVE GLUCOCORTICOIDS
Clinical condition resulting from prolonged exposure to excessively high levels of glucocorticoids (cortisol).
Causes of Cushing's Syndrome:
1. Pharmacologic (Exogenous)
- Most common cause. Results from therapeutic administration of exogenous glucocorticoids (e.g., prednisone) for autoimmune/inflammatory diseases.
2. Endogenous (Body's own overproduction)
- a. Cushing's Disease (Pituitary Adenoma): 75-90% of endogenous cases. Caused by a benign pituitary tumor secreting excessive ACTH. Leads to bilateral adrenal hyperplasia.
- Profile: High ACTH, High Cortisol.
- b. Adrenal Adenoma/Carcinoma (Primary Adrenal): Tumor within the adrenal gland autonomously secreting cortisol.
- Profile: Low ACTH (suppressed), High Cortisol.
- c. Ectopic ACTH Production: Non-pituitary tumor (e.g., small cell lung cancer) secreting ACTH.
- Profile: Very High ACTH, Very High Cortisol. Rapid onset.
Clinical Signs and Symptoms:
Body Fat Redistribution
- Central Obesity: Fat in trunk, thin extremities.
- Buffalo Hump: Fat pad between shoulders/neck.
- Moon Facies: Rounded, plethoric face.
Skin Changes
- Purple Striae: Broad stretch marks (abdomen, thighs).
- Thin, Fragile Skin: Easy bruising.
- Hirsutism & Acne: Due to androgenic effects.
Metabolic Derangements
- Hyperglycemia: Adrenal diabetes.
- Muscle Wasting: Proximal muscle weakness (proteolysis).
- Osteoporosis: Increased fracture risk.
Cardiovascular & Other
- Hypertension: Fluid retention, catecholamine sensitivity.
- Psychiatric: Mood swings, psychosis.
- Immune Suppression: Infections.
- Reproductive: Menstrual irregularities, decreased libido.
B. ADRENOCORTICAL INSUFFICIENCY: DEFICIENT GLUCOCORTICOIDS
1. Primary Adrenocortical Insufficiency (Addison's Disease)
Disorder where the adrenal glands themselves are damaged and cannot produce hormones.
- Causes:
- Autoimmune Adrenalitis: Most common cause (80-90%).
- Infections: Tuberculosis, Fungal, HIV.
- Tumors: Metastatic cancer.
- Hemorrhage: Sepsis, trauma (Waterhouse-Friderichsen).
- Drugs (Ketoconazole) or Genetic defects.
- Hormonal Profile: Low Cortisol, Low Aldosterone, Very High ACTH (lack of negative feedback).
Clinical Signs & Symptoms (Addison's)
- Fatigue and Weakness: Profound/chronic.
- Weight Loss & Anorexia.
- Hyperpigmentation: Hallmark sign due to very high ACTH (stimulates melanocytes). Skin darkening in sun-exposed areas, scars, gums.
- Hypotension: Low BP, orthostatic (lack of aldosterone/cortisol).
- Electrolyte Abnormalities: Hyponatremia (low Na+), Hyperkalemia (high K+).
- Salt Craving.
- Hypoglycemia.
Adrenal Crisis (Addisonian Crisis)
A life-threatening acute exacerbation often triggered by stress (infection, trauma, surgery). Characterized by severe hypotension, shock, vomiting, abdominal pain, profound weakness, and altered mental status. Requires immediate IV glucocorticoid replacement.
2. Secondary Adrenocortical Insufficiency
Deficiency of ACTH secretion from the pituitary gland, leading to insufficient stimulation of the adrenal cortex. The adrenal glands are typically healthy.
- Causes:
- Hypopituitarism: Tumors, radiation, Sheehan's syndrome.
- Suppression by Exogenous Steroids: Most common cause. Long-term steroid therapy suppresses HPA axis. Sudden stopping leads to insufficiency. Steroids must be tapered slowly.
- Hormonal Profile: Low Cortisol, Low ACTH. Aldosterone is usually normal (regulated by RAAS).
- Key Distinction from Primary: Absence of hyperpigmentation (due to low ACTH) and less severe electrolyte disturbances (preserved aldosterone).
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Adrenal Gland Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Adrenal Gland Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Thyroid Physiology
Thyroid Gland Physiology
THE THYROID GLAND
The endocrine system is a complex network of ductless glands that play a crucial role in maintaining homeostasis. Unlike exocrine glands that secrete substances through ducts, endocrine glands release their chemical messengers, known as hormones, directly into the surrounding extracellular fluid. These hormones then pass into the bloodstream for general circulation, traveling throughout the body to reach distant target organs or tissues. Upon arrival, hormones excite or inhibit the activity of these target cells, coordinating a vast array of physiological processes, including growth, metabolism, reproduction, and mood.
The thyroid gland is a vital component of this system, holding the distinction of being one of the largest endocrine glands in the human body. Its primary function is to regulate metabolism, energy production, and body temperature.
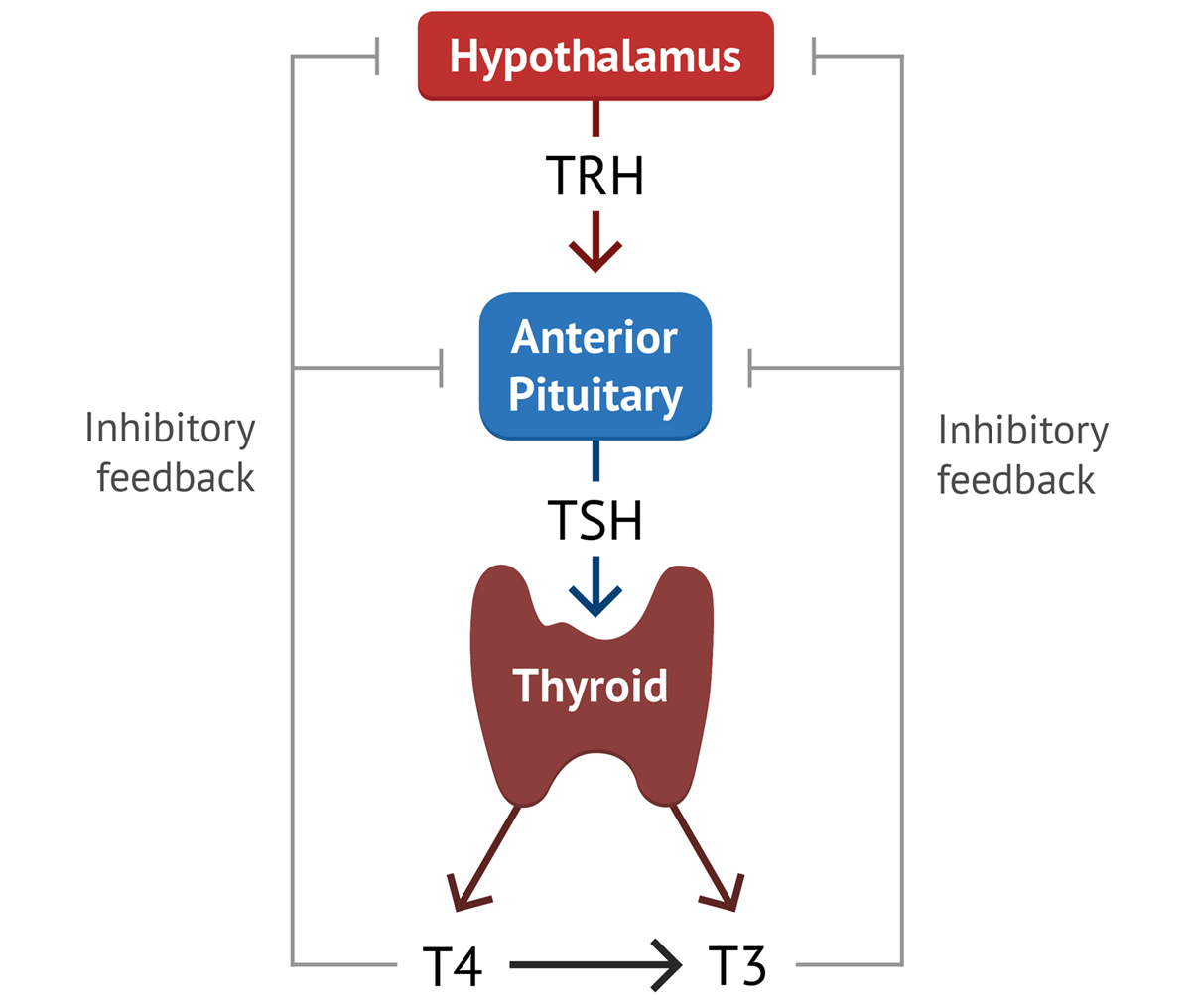
II. HORMONES SECRETED BY THE THYROID GLAND
The thyroid gland produces two main types of hormones:
1. Thyroid Hormones (from Follicular Cells)
- Thyroxine (T4): Also known as tetraiodothyronine. This is the more abundant hormone secreted by the thyroid. While less biologically active at the cellular level compared to T3, it serves as a prohormone and a major reservoir for T3.
- Triiodothyronine (T3): This is the more potent and biologically active form of thyroid hormone. Most of the T3 in the body is generated from the deiodination (removal of an iodine atom) of T4 in peripheral tissues, rather than directly secreted by the thyroid gland.
- Origin: Both T4 and T3 are synthesized and secreted by the follicular cells of the thyroid gland.
2. Calcitonin (from Parafollicular Cells)
- Calcitonin: This hormone plays a role in calcium homeostasis.
- Origin: It is secreted by specialized cells located interspersed between the thyroid follicles, known as parafollicular cells (also called C cells). Calcitonin's primary function is to lower blood calcium levels, acting in opposition to parathyroid hormone.
III. FUNCTIONAL ANATOMY OF THE THYROID GLAND
Understanding the structure of the thyroid gland is key to comprehending its function.
- Location: The thyroid gland is strategically located in the neck, immediately below the larynx (voice box) and anterior (in front of) to the upper part of the trachea (windpipe). Its superficial position makes it palpable during physical examination.
- Weight: In a healthy adult, it typically weighs about 15-20 grams. Its size can fluctuate with age, sex, and physiological states (e.g., pregnancy).
- Gross Structure: The gland consists of two distinct, fleshy lateral lobes, one on each side of the trachea. These lobes are connected by a narrow band of thyroid tissue called the isthmus.
- The isthmus usually overlies the region from the 2nd to the 4th tracheal cartilage, anchoring the gland in place.
- Sometimes, a third lobe, the pyramidal lobe, may extend upwards from the isthmus.
IV. PHYSIOLOGICAL ANATOMY: THE FUNCTIONAL UNIT
The microscopic structure of the thyroid gland reveals its unique functional organization:
- Thyroid Follicles: The lobes of the thyroid gland are composed of numerous, tiny, hollow, spherical structures called follicles. These follicles are the functional units of the thyroid gland. Each follicle is lined by a single layer of cuboidal epithelial cells – the follicular cells.
- Colloid: Each follicle is filled with a thick, sticky, protein-rich substance known as colloid.
- The major constituent of colloid is a large glycoprotein called thyroglobulin (Tg). This thyroglobulin serves as the scaffold for thyroid hormone synthesis and, crucially, as the storage site for thyroid hormones.
- Storage Uniqueness: Unlike most other endocrine glands, which store minimal amounts of pre-formed hormones and secrete them rapidly upon synthesis, the thyroid gland has a remarkable capacity to store considerable amounts of thyroid hormones within the colloid (bound within thyroglobulin). This stored reserve can last for several weeks or even months, providing a buffer against fluctuations in iodine intake or acute demand.
- C Cells (Parafollicular Cells): Interspersed between the follicles are the C cells (or parafollicular cells), which, as mentioned, are responsible for secreting calcitonin.
V. SYNTHESIS, STORAGE, AND SECRETION OF THYROID HORMONES
The production of thyroid hormones is a multi-step process that requires specific raw materials and intricate cellular machinery.
Essential Precursors:
- Tyrosine: An amino acid that forms the backbone of thyroid hormones. It is synthesized by the body and is a crucial component of the thyroglobulin protein.
- Iodine: A trace element that is dietary essential. The body cannot synthesize iodine; it must be consumed through diet (e.g., iodized salt, seafood). Iodine is absolutely critical for thyroid hormone synthesis, as it is incorporated directly into the tyrosine residues of thyroglobulin.
Location of Synthesis: Hormone synthesis primarily occurs on the thyroglobulin molecule within the colloid.
Steps Involved in Synthesis, Storage, and Secretion:
1. Thyroglobulin Production and Release (Into Colloid)
- Process: Follicular cells synthesize the large glycoprotein thyroglobulin (Tg) in their endoplasmic reticulum and Golgi apparatus.
- Delivery: Tg is then packaged into vesicles and released into the follicular lumen (colloid space) by exocytosis.
2. Iodine Uptake (Iodide Trapping)
- Process: Follicular cells actively transport iodide (I-) from the bloodstream into their cytoplasm. This is an energy-dependent process mediated by the Sodium-Iodide Symporter (NIS), which co-transports two Na+ ions with one I- ion.
- Concentration: This mechanism allows the thyroid gland to concentrate iodide to levels 20-40 times higher than in the blood (and even higher under TSH stimulation).
- Transfer to Colloid: Iodide is then transported from the follicular cell cytoplasm into the colloid space via a chloride-iodide counter-transporter called pendrin.
3. Oxidation and Iodination of Tyrosine (Organification)
- Process: Once in the colloid, iodide is rapidly oxidized to iodine (I0 or I+) by the enzyme thyroid peroxidase (TPO).
- Attachment to Tyrosine: This highly reactive iodine then attaches to specific tyrosine residues within the thyroglobulin molecule.
- One iodine atom attached to a tyrosine forms Monoiodotyrosine (MIT).
- Two iodine atoms attached to a tyrosine form Diiodotyrosine (DIT).
4. Coupling Processes (Formation of T4 and T3)
- Process: Still within the thyroglobulin molecule, two iodinated tyrosine molecules are coupled together by TPO:
- DIT + DIT → T4 (Thyroxine): This is the primary coupling reaction, forming the most abundant thyroid hormone.
- MIT + DIT → T3 (Triiodothyronine): This reaction produces T3. Less commonly, DIT + MIT can also form reverse T3 (rT3), which is metabolically inactive.
- Storage: The newly formed T4 and T3 remain covalently bound within the large thyroglobulin molecule, stored as part of the colloid, often for weeks.
Secretion (Upon Stimulation):
- Stimulus: When the body requires thyroid hormones, the follicular cells are stimulated, primarily by Thyroid-Stimulating Hormone (TSH) from the anterior pituitary.
- Endocytosis: Follicular cells engulf small portions of the colloid (containing iodinated thyroglobulin) by endocytosis, forming colloid droplets.
- Lysosomal Digestion: These colloid droplets fuse with lysosomes within the follicular cells. Lysosomal proteases digest the thyroglobulin, releasing free T4, T3, MIT, and DIT.
- Recycling and Diffusion:
- MIT and DIT are deiodinated by the enzyme deiodinase, and the released iodide is recycled back into the synthesis pathway.
- Free T4 and T3 are then released into the bloodstream by diffusing out of the follicular cell.
Quantitative and Potency Differences:
- Approximately 93% of the hormones released from the thyroid gland initially appear in the form of T4.
- However, a majority of the T4 that is secreted from the thyroid gland is subsequently converted to T3 in peripheral tissues (e.g., liver, kidneys, target cells) by enzymes called deiodinases.
- T3 is approximately 4 times more potent in its biologic form than T4 and is considered the major hormone that interacts with the target cells to produce most of the thyroid hormone effects.
- T3 is much smaller in quantities in the circulation and persists for a much shorter time (shorter half-life) than T4, reflecting its higher activity and rapid utilization.
Transport in Blood:
- Both T4 and T3 are highly lipophilic. Once in the blood, they immediately bind to plasma proteins.
- Thyroid hormone-specific binding globulin (TBG) is the primary carrier, transporting 70-80% of thyroid hormones. Other non-specific proteins like albumin and transthyretin also transport a portion.
- Less than 0.1% of T4 and 1% of T3 are in the unbound (free) form.
- These free T4 (fT4) and free T3 (fT3) compounds are biologically active, and it is these components that produce the effects of the thyroid hormones on peripheral tissues and participate in the pituitary feedback mechanism. The protein-bound hormones serve as a circulating reservoir.
VI. REGULATION OF THYROID HORMONE SECRETION (THE HPT AXIS)
Thyroid hormone secretion is under precise control of the hypothalamic-pituitary-thyroid (HPT) axis, a classic example of a negative feedback loop.
- Hypothalamic Control (TRH):
- The secretion of thyroid-stimulating hormone (TSH) from the anterior pituitary is primarily controlled by Thyrotropin-Releasing Hormone (TRH), a tripeptide hormone.
- TRH is secreted by specialized neurosecretory cells in the paraventricular nucleus of the hypothalamus. It is then released into the hypophyseal portal system, which carries it to the anterior pituitary.
- Pituitary Control (TSH):
- Upon reaching the anterior pituitary, TRH stimulates the thyrotroph cells to synthesize and secrete Thyroid-Stimulating Hormone (TSH) (also known as thyrotropin).
- TSH is a glycoprotein hormone that has several effects on the thyroid gland:
- Increases iodide uptake (iodide trapping).
- Increases synthesis of thyroglobulin.
- Increases TPO activity (iodination and coupling).
- Increases endocytosis of colloid and release of T4 and T3.
- Promotes the growth and vascularity of the thyroid gland.
- Thyroid Gland Response (T4 and T3):
- TSH stimulates the thyroid gland to produce and release T4 and T3.
- Negative Feedback (Inhibition of TRH and TSH):
- Elevated levels of free T4 and T3 in the body fluids exert negative feedback on both the hypothalamus and the anterior pituitary.
- They inhibit the secretion of TRH from the hypothalamus.
- They directly inhibit the sensitivity of the thyrotroph cells in the anterior pituitary to TRH, thereby decreasing the secretion of TSH.
- This feedback mechanism ensures that thyroid hormone levels are maintained within a narrow physiological range. For example, if thyroid hormone levels are 1.75 times normal, the rate of TSH secretion can fall essentially to zero as the feedback mechanism shuts down pituitary stimulation.
- Elevated levels of free T4 and T3 in the body fluids exert negative feedback on both the hypothalamus and the anterior pituitary.
- Other Factors Influencing Regulation:
- Cold Exposure: Exposure of an animal (and to some extent, humans, especially infants) to coldness significantly increases the rate of TRH secretion from the hypothalamus. This leads to increased TSH and subsequent thyroid hormone production, which helps to increase metabolic rate and heat production.
- Stress and Excitement: Conditions causing acute stress, excitement, and strong sympathetic nervous system stimulation (e.g., severe anxiety, trauma) can cause an acute decrease in the secretion of TSH, likely by inhibiting TRH release. This is thought to be a protective mechanism to prevent an excessive metabolic response during acute stress.
- Iodine Availability: Chronic iodine deficiency leads to decreased thyroid hormone synthesis, reduced negative feedback, and thus chronically elevated TSH. Prolonged high TSH can lead to thyroid gland enlargement (goiter).
PHYSIOLOGICAL EFFECTS OF THYROID HORMONES
Thyroid hormones (T3 and T4) are essential for normal growth, development, and metabolism in virtually every cell of the body. Their widespread actions underscore their critical role in maintaining overall physiological homeostasis.
I. METABOLIC RATE AND HEAT PRODUCTION
The most fundamental and pervasive effect of thyroid hormones is their impact on the body's metabolic rate.
- A. Increased Metabolic Activities: Thyroid hormones act as a crucial metabolic thermostat. They increase the rate of most metabolic activities in almost all cells of the body. This involves stimulating various cellular processes, including:
- Increased synthesis and degradation of proteins.
- Increased glucose utilization and fat metabolism.
- Enhanced activity of enzymes involved in nutrient oxidation.
- B. Increased Oxygen Consumption: As metabolic processes are upregulated, there is a corresponding increase in oxygen consumption by most metabolically active tissues. This is a direct consequence of enhanced cellular respiration to meet the increased energy demand. This effect is particularly pronounced in tissues like muscle, liver, and kidneys, but excludes the brain, gonads, and spleen, which are less affected by thyroid hormone-induced increases in oxygen consumption.
- C. Elevated Basal Metabolic Rate (BMR): Thyroid hormones are the primary determinants of the Basal Metabolic Rate (BMR), which is the minimum energy expenditure required to maintain essential body functions at rest.
- In conditions of hyperthyroidism (excess thyroid hormone), the BMR can significantly increase by 60-100% above normal. This means the body burns far more calories at rest.
- Conversely, in hypothyroidism (deficient thyroid hormone), the BMR can fall to 40-50% below normal, leading to reduced energy expenditure and weight gain.
- D. Calorigenic Effect: Since increased metabolism results in increased heat production, thyroid hormones have a distinct calorigenic effect.
- The heightened cellular activity and ATP turnover generate more heat as a byproduct.
- This effect is crucial for maintaining body temperature, especially in cold environments. Individuals with hyperthyroidism often complain of heat intolerance due to excessive heat production, while those with hypothyroidism often experience cold intolerance.
II. INTERMEDIARY METABOLISM
Thyroid hormones significantly modulate the rates of many specific reactions involved in the metabolism of carbohydrates, fats, and proteins.
- A. Carbohydrate Metabolism:
- Increased Glucose Uptake: Enhances glucose uptake by cells.
- Increased Glycolysis and Gluconeogenesis: Stimulates both the breakdown of glucose for energy and the synthesis of glucose from non-carbohydrate sources.
- Increased Insulin Secretion: Indirectly increases insulin secretion in response to increased blood glucose levels.
- Increased Glycogenolysis: Promotes the breakdown of glycogen stores (in liver and muscle) to release glucose.
- B. Fat Metabolism:
- Increased Lipolysis: Enhances the breakdown of triglycerides from adipose tissue, releasing fatty acids and glycerol into the blood.
- Reduced Cholesterol and Triglyceride Levels: Thyroid hormones accelerate the removal of cholesterol and triglycerides from the plasma by increasing the number of LDL receptors on liver cells, leading to increased clearance of LDL cholesterol. This is why hypothyroidism is often associated with elevated cholesterol levels.
- C. Protein Metabolism:
- Anabolic and Catabolic Effects: Thyroid hormones are crucial for normal protein synthesis and degradation.
- Physiological vs. Excess Levels: At physiological concentrations, thyroid hormones promote protein synthesis, which is essential for growth and development. However, in excessive amounts (hyperthyroidism), they can cause a net increase in protein catabolism, leading to muscle wasting and negative nitrogen balance.
III. SYMPATHOMIMETIC EFFECT
Thyroid hormones have a remarkable interaction with the sympathetic nervous system.
- A. Definition of Sympathomimetic: A sympathomimetic effect refers to any action that is similar to or mimics those produced by the sympathetic nervous system (the "fight or flight" response), typically by activating adrenergic receptors.
- B. Increased Responsiveness to Catecholamines: Thyroid hormones significantly increase the target cell responsiveness to catecholamines (epinephrine and norepinephrine), which are the neurotransmitters of the sympathetic nervous system and hormones from the adrenal medulla.
- Permissive Action: Thyroid hormones are said to be "permissive" in this context. They do not directly cause sympathetic effects but rather enhance the ability of catecholamines to produce their effects. They achieve this by increasing the production of specific catecholamine target cell receptors, particularly beta-adrenergic receptors, on target cells.
- Clinical Relevance: This explains why many symptoms of hyperthyroidism (e.g., rapid heart rate, tremors, anxiety, sweating) resemble an overactive sympathetic nervous system, even though catecholamine levels themselves might not be significantly elevated.
IV. THE CARDIOVASCULAR SYSTEM
The cardiovascular system is highly sensitive to thyroid hormone levels, exhibiting profound changes in both hyper- and hypothyroidism.
- A. Increased Responsiveness to Catecholamines (Reiteration): The permissive action discussed above is particularly evident in the heart. Thyroid hormones increase the heart's responsiveness to circulating catecholamines, making it more sensitive to their stimulatory effects.
- B. Increased Heart Rate and Force of Contraction: Directly and indirectly (via enhanced catecholamine sensitivity), thyroid hormones:
- Increase heart rate (tachycardia): Even at rest.
- Increase the force of myocardial contraction: Leading to a stronger heartbeat.
- These combined effects result in a significant increase in cardiac output (CO), the volume of blood pumped by the heart per minute. This increased cardiac output is necessary to deliver the additional oxygen required by the metabolically active tissues.
- C. Peripheral Vasodilation: In response to the increased metabolic rate and resulting heat load generated by the calorigenic effect, the body needs to dissipate excess heat. Thyroid hormones promote peripheral vasodilation (widening of blood vessels in the skin).
- This increases blood flow to the skin, allowing for more heat to be transferred from the body's core to the surface, facilitating heat loss and helping to maintain a stable core body temperature.
V. GROWTH AND THE NERVOUS SYSTEM
Thyroid hormones are indispensable for normal growth, maturation, and neurological development.
- A. Effect on Growth (Mainly in Growing Children):
- Stimulates GH Secretion and Action: Thyroid hormone stimulates the secretion of Growth Hormone (GH) from the anterior pituitary and promotes the effects of GH on target tissues. This synergistic action is crucial for skeletal and tissue growth.
- Growth Retardation in Deficiency: A lack of thyroid hormone during childhood (juvenile hypothyroidism or cretinism) leads to severe growth retardation (dwarfism). Importantly, this can often be reversed by thyroid replacement therapy if administered early enough.
- No Excessive Growth in Excess: Unlike excess GH, excess thyroid hormone does not produce excessive growth. In fact, severe hyperthyroidism can sometimes lead to muscle wasting and weight loss, counteracting growth.
- B. Important for Brain Development: Thyroid hormones are critically important in promoting the normal growth and development of the brain during fetal and postnatal life, particularly during the first few years after birth.
- Myelination and Synaptogenesis: They are essential for processes like neuronal migration, myelination (formation of the myelin sheath around nerves), and synaptogenesis (formation of connections between neurons).
- Mental Retardation in Deficiency: Thyroid hormone deficiency during this critical period (e.g., congenital hypothyroidism, historically called cretinism) leads to severe and irreversible mental retardation if thyroid replacement therapy is not administered within days or weeks after birth. The damage to the developing brain is permanent if left untreated.
VI. OTHER EFFECTS OF THYROID HORMONES
Thyroid hormones have diverse effects on many other physiological systems.
- A. Effect on Sleep:
- Hyperthyroidism: Due to the excitable effects of thyroid hormone on the synapses and the general increase in metabolic activity, individuals with hyperthyroidism often experience difficulty sleeping (insomnia), even though they may feel constantly tired. They are in a state of heightened alertness and nervous excitability.
- Hypothyroidism: Conversely, in hypothyroidism, reduced metabolic activity leads to decreased mental alertness and sluggishness. Individuals often experience excessive sleepiness, with sleep sometimes lasting for 12 to 14 hours a day or more.
- B. Effect on Sexual Function:
- Normal Function Requires Normal Levels: For normal sexual function in both men and women, thyroid secretion needs to be within the normal range. Both hypo- and hyperthyroidism can disrupt reproductive health.
- In Men:
- Lack of thyroid hormone (hypothyroidism) can lead to loss of libido (sex drive) and, in severe cases, impotence.
- Great excesses (hyperthyroidism) can also sometimes lead to impotence or decreased libido.
- In Women:
- Lack of thyroid hormone (hypothyroidism) is often associated with menstrual irregularities such as menorrhagia (excessive menstrual bleeding) and polymenorrhea (frequent menstrual bleeding). In severe cases, it can lead to anovulation and infertility.
- Hyperthyroidism can cause oligomenorrhea (infrequent menstruation) or amenorrhea (absence of menstruation), and reduced fertility.
DISORDERS OF THYROID METABOLISM
Thyroid hormones are powerful regulators, and deviations from their precise physiological range can lead to significant and widespread health problems. These disorders are broadly categorized into two main types: hypothyroidism (deficient thyroid hormone) and hyperthyroidism (excessive thyroid hormone).
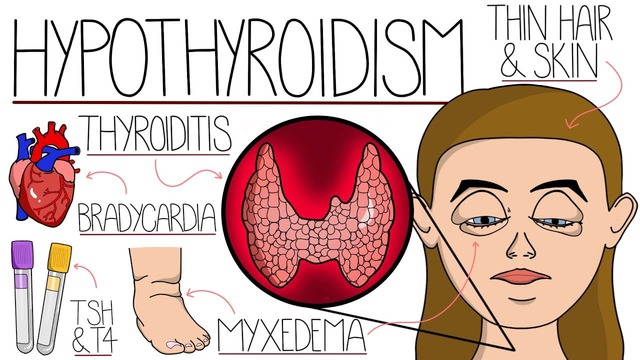
I. HYPOTHYROIDISM: THYROID HORMONE DEFICIENCY
Hypothyroidism refers to the clinical syndrome resulting from a deficiency of thyroid hormones. This underactivity of the thyroid gland can arise from several causes affecting different levels of the hypothalamic-pituitary-thyroid (HPT) axis.
Etiology (Causes):
Hypothyroidism develops if the thyroid gland shows:
- i. Atrophy or Destruction of Secretory Cells: This is the most common cause, often due to autoimmune disease.
- Hashimoto's Thyroiditis: The most prevalent cause of primary hypothyroidism in iodine-sufficient regions. It is an autoimmune disorder where the body's immune system attacks and gradually destroys the thyroid follicular cells.
- Thyroidectomy: Surgical removal of all or part of the thyroid gland (e.g., for thyroid cancer, severe hyperthyroidism).
- Radioactive Iodine (RAI) Therapy: Used to treat hyperthyroidism or thyroid cancer, RAI destroys thyroid tissue.
- External Radiation: Radiation therapy to the neck region for other cancers can damage the thyroid.
- ii. Inadequate Stimulation: This refers to central (secondary or tertiary) hypothyroidism.
- Secondary Hypothyroidism: Due to a deficiency of TSH (Thyroid-Stimulating Hormone) secretion from the anterior pituitary gland. Causes include pituitary tumors, surgery, radiation, or infarction (Sheehan's syndrome).
- Tertiary Hypothyroidism: Due to a deficiency of TRH (Thyrotropin-Releasing Hormone) secretion from the hypothalamus. This is less common and often associated with other hypothalamic-pituitary dysfunctions.
- iii. Iodine Deficiency: A global cause of hypothyroidism, especially in regions with inadequate dietary iodine. Without sufficient iodine, the thyroid cannot synthesize adequate amounts of T4 and T3.
- iv. Genetic Defects: Rare genetic disorders affecting thyroid hormone synthesis (e.g., defects in TPO, thyroglobulin synthesis, or iodine trapping).
- v. Drugs: Certain medications can impair thyroid function (e.g., lithium, amiodarone, interferon-alpha).
Clinical Presentation & Symptoms of Hypothyroidism
The symptoms are varied and reflect a generalized slowing down of metabolic processes.
- Metabolic & General: Decreased BMR (<40-50% normal), Poor Tolerance of Cold, Tendency to Gain Weight, Fatigue and Extreme Somnolence (12-14 hrs/day), Easily Fatigued.
- Cardiovascular: Reduced Heart Rate (Bradycardia), Reduced Blood Pressure, Reduced Cardiac Output.
- Gastrointestinal: Sluggish Gut Movements (Constipation).
- Neurological & Psychiatric: Decreased Alertness, Slow Speech, Poor Memory, Depression, Paresthesias (carpal tunnel).
- Musculoskeletal: Muscle Weakness and Aches.
- Skin, Hair, & Voice: Dry Coarse Skin, Brittle Hair/Nails (Queen Anne's sign), Hoarseness/Slow Husky Voice.
- Reproductive: Menorrhagia/polymenorrhea/infertility (Women), Decreased libido/erectile dysfunction (Men).
Treatment for Hypothyroidism:
- Replacement Therapy: The primary treatment is lifelong replacement therapy with synthetic thyroid hormone, typically levothyroxine (synthetic T4).
- Iodine Supplementation: For iodine deficiency-induced hypothyroidism, iodine supplementation is crucial, often in combination with thyroid hormone if severe deficiency has already occurred.
- Promptness: Timely diagnosis and treatment are essential, particularly in infants and children, to prevent irreversible developmental deficits.
Myxedema (Severe Adult Hypothyroidism)
- Description: Myxedema is a severe form of hypothyroidism that develops in patients with an almost total lack of thyroid hormone function.
- Pathophysiology: Accumulation of hydrophilic mucopolysaccharides (hyaluronic acid and chondroitin sulfate) in interstitial spaces, forming "tissue gel" (mucinous edema).
- Clinical Manifestations: Non-Pitting Edema, Characteristic Swelling (bagginess under eyes, puffy face).
- Myxedema Coma: A rare, life-threatening complication characterized by profound hypothermia, hypoventilation, bradycardia, hypotension, and altered mental status.
Atherosclerosis in Hypothyroidism:
- Increased Blood Cholesterol: Due to altered fat metabolism and diminished liver excretion of cholesterol.
- Increased Atherosclerosis Risk: Strongly associated with hardening/narrowing of arteries.
- Serious Complications: Peripheral Vascular Disease, Deafness, Coronary Artery Disease.
Cretinism (Congenital Hypothyroidism)
- Description: Caused by extreme hypothyroidism during fetal life, infancy, or early childhood.
- Key Characteristics: Failure of body growth (dwarfism) and profound mental retardation.
- Causes: Congenital Lack of Thyroid Gland, Genetic Defects, Iodine Lack (Endemic Cretinism), Maternal Hypothyroidism.
- Irreversible Mental Retardation: If treatment is not administered within days or weeks after birth, the mental retardation becomes permanent.
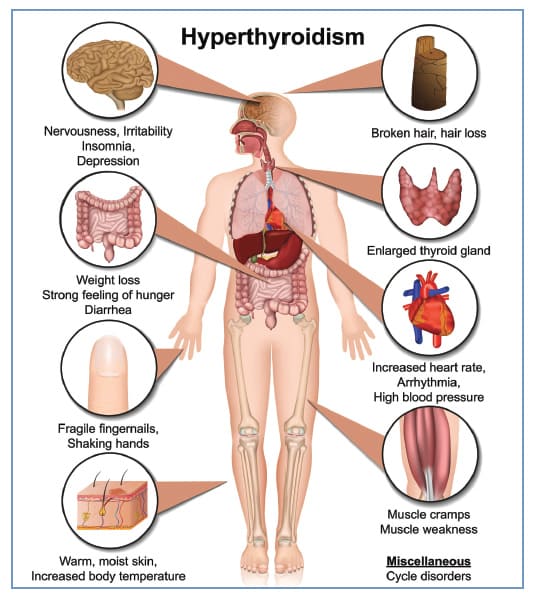
II. HYPERTHYROIDISM: THYROID HORMONE EXCESS
Hyperthyroidism is the clinical syndrome resulting from excessive secretion of thyroid hormones. This leads to a hypermetabolic state with widespread effects.
Etiology (Causes):
- Graves' Disease (Most Common Cause):
- Autoimmune Disease: Body produces antibodies called Thyroid-Stimulating Immunoglobulins (TSI) or TSH receptor antibodies (TRAb).
- TSH Receptor Agonists: TSI bind to TSH receptors and mimic TSH action, continuously stimulating the gland.
- Autonomous Stimulation: Not subject to negative feedback.
- Diffuse Goiter: Uniform enlargement of the gland.
- Toxic Multinodular Goiter (Plummer's Disease): Multiple nodules become autonomous.
- Toxic Adenoma: Single benign tumor autonomously producing hormones.
- Thyroiditis: Inflammation causing temporary release of stored hormones.
- Exogenous Thyroid Hormone Intake: Overdose.
- TSH-Secreting Pituitary Adenoma: Rare tumor.
- Iodine-Induced Hyperthyroidism (Jod-Basedow Phenomenon): Exposure to large iodine amounts in pre-existing abnormality.
Clinical Presentation & Symptoms of Hyperthyroidism
- Metabolic & General: High State of Excitability, Intolerance to Heat/Increased Sweating, Mild to Extreme Weight Loss (despite polyphagia), Extreme Fatigue but Inability to Sleep, Fine Tremor of Hands.
- Cardiovascular: Tachycardia, Palpitations, Increased Cardiac Output, Arrhythmias (Atrial fibrillation), Hypertension.
- Gastrointestinal: Diarrhea.
- Musculoskeletal: Muscle Weakness (Proximal Myopathy), Osteoporosis.
- Neurological & Psychiatric: Nervousness/Anxiety, Psychic Disorders, Hyperreflexia.
F. Eye Changes (Exophthalmos in Graves' Disease)
- Exophthalmos (Proptosis): Prominent feature of Graves' disease only.
- Pathophysiology: Autoimmune inflammatory process behind eyeballs. Deposition of complex water-retaining carbohydrates.
- Mechanism: Fluid retention pushes eyeballs forward.
- Clinical Significance: Eye irritation, dryness, diplopia, optic nerve compression.
Treatment for Hyperthyroidism:
- A. Antithyroid Drugs:
- Propylthiouracil (PTU) and Methimazole (Tapazole): Mechanism: Inhibit thyroid peroxidase (TPO), blocking iodination and coupling. PTU also blocks peripheral conversion of T4 to T3.
- B. Radioactive Iodine (RAI) Therapy: Oral administration of I-131. Selectively destroys follicular cells.
- C. Surgical Removal (Thyroidectomy): Excision of part or most of the gland.
- D. Other Treatments:
- Beta-Blockers (e.g., Propranolol): Symptomatic relief of adrenergic symptoms.
- Inorganic Iodides (e.g., Lugol's solution): In high concentrations (Wolf-Chaikoff Effect), acutely reduce iodide trapping and inhibit hormone release. Used for preparation for surgery or thyroid storm.
III. GOITER: ENLARGEMENT OF THE THYROID GLAND
A goiter simply means an enlargement of the thyroid gland. It can occur in hypothyroidism, hyperthyroidism, or euthyroid states.
A. Toxic Goiter:
- Definition: Enlargement associated with increased secretion of thyroid hormones.
- Causes: Graves' disease (diffuse toxic goiter), toxic multinodular goiter, toxic adenoma.
B. Non-Toxic Goiter (Euthyroid Goiter or Hypothyroid Goiter):
- Definition: Enlargement without an increase in hormone secretion. Often called a "compensatory goiter."
- Causes: Attempt to compensate for decreased hormone production. Stimulated by elevated TSH.
- 1. Endemic Colloid Goiter: Most common cause is iodine deficiency (dietary intake < 50 µg/day). Pituitary secretes more TSH, leading to hypertrophy/hyperplasia and accumulation of poorly iodinated thyroglobulin. Prevalent in mountainous regions.
- 2. Sporadic (Idiopathic) Non-Toxic Goiter: Occurs in regions with sufficient iodine. Causes include mild genetic defects, consumption of goitrogens (cassava, cabbage, turnips), mild thyroiditis, or nodular goiter.
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Thyroid Gland Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Thyroid Gland Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Hormone Regulation, Disorders & Transport
Endocrine System
Regulation of Hormone Secretion
The endocrine system meticulously controls hormone secretion to ensure physiological homeostasis and appropriate responses to environmental cues. This regulation involves complex feedback loops, neural reflexes, and intrinsic biological rhythms.
I. Characteristics of Hormone Secretion and Action
Before delving into the control mechanisms, it's important to understand some general characteristics of hormone dynamics:
Variable Latency and Duration of Action
- Rapid-Acting Hormones: Some hormones, typically those involved in immediate stress responses or neural signaling, are secreted within seconds after stimulation. Examples include epinephrine and norepinephrine from the adrenal medulla. Their full physiological actions can develop within another few seconds to minutes (e.g., increased heart rate, blood pressure, glucose mobilization).
- Slower-Acting Hormones: Others, like thyroxine (thyroid hormone) or growth hormone, may take hours, days, or even months to exert their full physiological effects. Thyroxine, for instance, influences metabolic rate over an extended period, and its full impact on growth and development is gradual. Growth hormone's effects on skeletal growth accumulate over years.
Extremely Low Serum Concentrations
- Hormones are incredibly potent, meaning they can exert significant effects even at very low concentrations. Their serum concentrations are typically measured in picograms to micrograms per 100 ml of blood.
- Consequently, they are secreted in equally small amounts, often in micrograms or milligrams per day. This underscores the efficiency and sensitivity of the endocrine signaling system, where a tiny amount of chemical messenger can orchestrate widespread physiological changes.
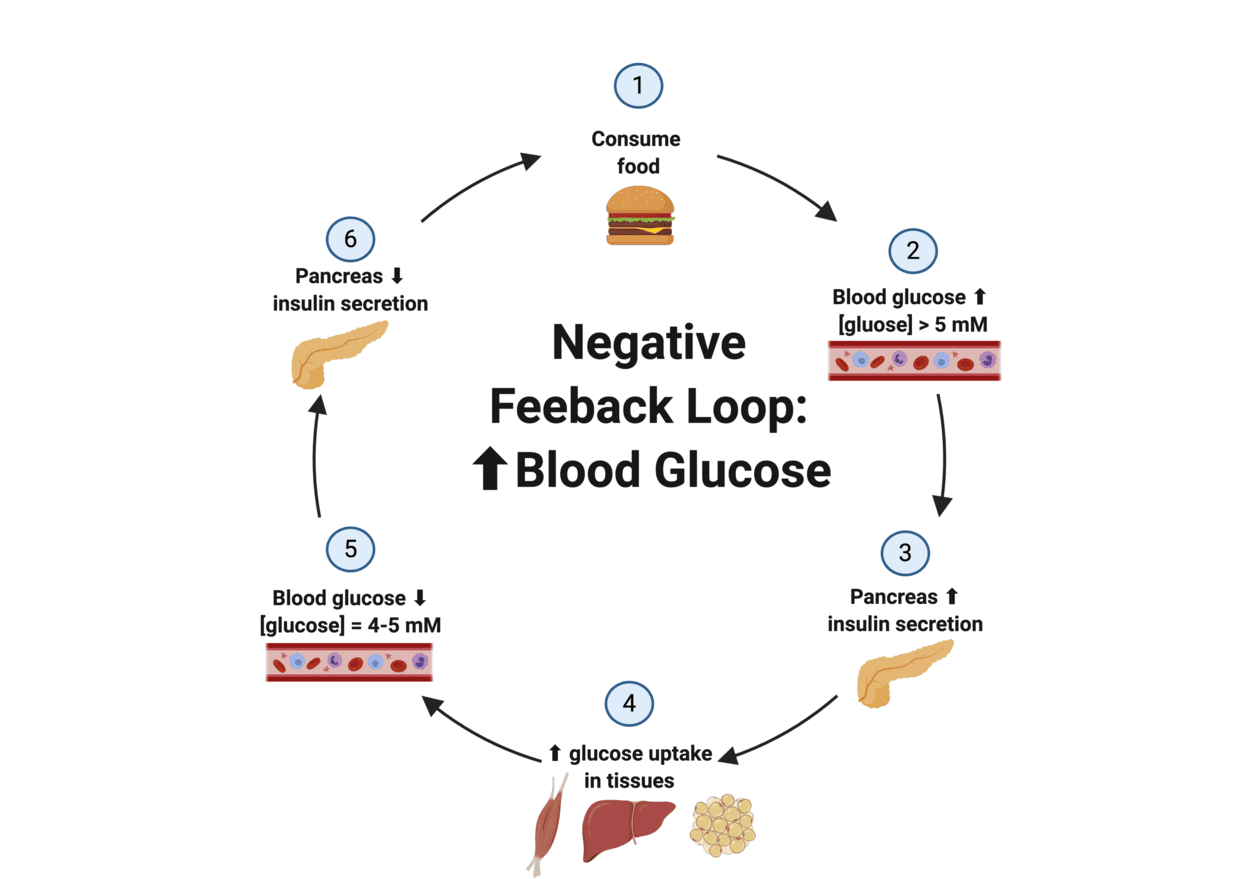
II. Mechanisms of Control of Hormonal Secretion
Hormone secretion is not a constant process but is dynamically regulated by several intricate mechanisms to maintain precise control.
1. Negative-Feedback Control
This is the most common and fundamental regulatory mechanism in the endocrine system, ensuring stability and preventing overactivity.
- Definition: Negative feedback exists when the output of a system counteracts a change in input, thereby maintaining a controlled variable within a narrow, desired range (around a "set point").
- Mechanism:
- A stimulus (e.g., deviation from a set point) triggers an endocrine gland to release a hormone.
- This hormone then acts on target cells to produce a specific physiological response.
- Crucially, the physiological response itself, or the hormone's direct effect on the gland, then inhibits the further release of the initial hormone. This "turns off" the stimulatory signal once the desired level or effect is achieved.
Example: Thyroid Hormone Regulation (HPT Axis)
- Initial Stimulus: If the plasma concentration of free circulating thyroid hormone (T₃ and T₄) falls below a given set point.
- Hypothalamic Response: The hypothalamus senses this drop and secretes Thyrotropin-Releasing Hormone (TRH).
- Anterior Pituitary Response: TRH stimulates the anterior pituitary to secrete Thyroid-Stimulating Hormone (TSH).
- Thyroid Gland Response: TSH stimulates the thyroid gland to increase its secretion of thyroid hormones (T₃ and T₄).
- Negative Feedback Loop: As the levels of T₃ and T₄ in the blood rise back to the set point (or above), they exert negative feedback at two levels:
- On the Anterior Pituitary: T₃ and T₄ directly inhibit the secretion of TSH.
- On the Hypothalamus: T₃ and T₄ also inhibit the secretion of TRH.
- Outcome: This inhibition ensures that once thyroid gland secretion has been "turned on" by TSH, it will not continue unabated. Instead, it will be "turned off" when the appropriate level of free circulating thyroid hormone has been achieved, thus preventing excessive thyroid hormone levels.
Significance:
- Prevents Overactivity: Negative feedback is critical for preventing the overproduction or underproduction of hormones, maintaining stable internal conditions (homeostasis).
- Maintains Set Point: It ensures that hormone levels and their physiological effects remain within a relatively narrow, healthy range.
2. Positive-Feedback Mechanisms
While less common for maintaining ongoing homeostasis, positive feedback plays a vital role in specific, self-amplifying processes.
- Definition: Positive feedback mechanisms control events that are often "out of control" or self-amplifying, and do not require continuous adjustment to a set point; instead, they push the system further in the same direction.
- Nature: Rarely used to maintain homeostasis because they tend to drive physiological variables away from a stable state.
- Mechanism:
- A stimulus triggers an endocrine response.
- This response then amplifies the original stimulus, leading to an even greater response.
- This cycle continues until a specific event or external factor breaks the loop.
Example: Childbirth
- Initial Stimulus: Pressure of the baby's head against the cervix during labor.
- Neural Signal: Nerve impulses from the cervix are sent to the hypothalamus.
- Hormone Release: The hypothalamus stimulates the posterior pituitary to release oxytocin.
- Target Response: Oxytocin stimulates and enhances the contractions of the uterine smooth muscle.
- Positive Feedback Loop: The stronger uterine contractions push the baby further down, increasing the pressure on the cervix. This increased cervical stretching sends more nerve impulses to the hypothalamus, leading to the release of more oxytocin, which in turn causes even stronger contractions.
- Outcome: This intensifies contractions until the baby is delivered from the birth canal. Once the baby is born, the cervical stretching stops, the oxytocin production significantly decreases, and the labor contractions cease, thus breaking the positive feedback loop.
3. Neuroendocrine Reflexes
These mechanisms highlight the intimate connection between the nervous and endocrine systems, often allowing for rapid, precise hormonal responses to external stimuli.
- Definition: Many endocrine control systems involve neuroendocrine reflexes, which include both neural and hormonal components. They typically involve nerve impulses triggering hormone release.
- Mechanism: Neural input to an endocrine gland directly stimulates or inhibits hormone secretion. This often produces a sudden increase in hormone secretion (like "turning up the thermostat setting") in response to a specific stimulus, which is frequently an external one.
Pure Neural Control Example: Adrenal Medulla Secretion
- Mechanism: In some instances, neural input to the endocrine gland is the only factor regulating the secretion of a hormone.
- Example: The secretion of epinephrine and norepinephrine by the adrenal medulla is solely controlled by the sympathetic nervous system. When faced with a perceived threat (fight-or-flight response), sympathetic preganglionic neurons directly stimulate chromaffin cells in the adrenal medulla to release these catecholamines into the bloodstream. There is no hormonal intermediate from a higher endocrine gland.
Combined Feedback and Neuroendocrine Reflexes Example: Cortisol Secretion
- Mechanism: Some endocrine systems incorporate both feedback control (for maintaining a constant basal level of the hormone) and neuroendocrine reflexes (for causing sudden bursts in secretion in response to increased need).
- Example: Cortisol, often called the "stress hormone," is primarily regulated by the HPA axis through negative feedback, which maintains basal cortisol levels. However, during a stress response (e.g., physical injury, psychological stress), sensory input rapidly activates the hypothalamus to release a surge of CRH. This neural-derived signal (via CRH) quickly overrides the negative feedback loop to dramatically increase ACTH and subsequently cortisol secretion. This allows for a rapid and substantial elevation of cortisol to help the body cope with the stressor.
- Significance: This dual control allows for both long-term homeostatic regulation and rapid, adaptive responses to acute challenges.
4. Diurnal (Circadian) Rhythms
Many hormones exhibit predictable fluctuations in their secretion rates over a 24-hour cycle, influenced by the body's internal clock.
- Definition: The secretion rates of many hormones rhythmically fluctuate up and down as a function of time. The most common endocrine rhythm is the diurnal ("day-night") or circadian ("around a day") rhythm.
- Mechanism: These rhythms are controlled by an internal biological clock, the suprachiasmatic nucleus (SCN) in the hypothalamus, which is entrained by external cues, most notably the light-dark cycle (light perceived by the eyes sends signals to the SCN).
- Example: Cortisol Secretion
- Pattern: Cortisol secretion rises during the night, reaches its peak secretion in the morning (typically just before a person wakes up), then gradually falls throughout the day to its lowest level at bedtime.
- Physiological Role: This pattern anticipates the metabolic demands of the active daytime period, ensuring that the body has sufficient glucose and is prepared for activity and potential stressors upon waking.
- Other Examples:
- Growth Hormone: Secreted in pulses, with the largest pulse often occurring shortly after the onset of deep sleep.
- Melatonin: Exhibits a prominent circadian rhythm, with secretion increasing in the evening, peaking during the night, and decreasing in the morning, influencing the sleep-wake cycle.
- Significance: Circadian rhythms allow the endocrine system to anticipate and prepare the body for predictable daily changes in activity, feeding, and sleep, optimizing physiological function over the 24-hour cycle. Disruptions to these rhythms (e.g., jet lag, shift work) can significantly impact health.
Causes of Endocrine Disorders
Endocrine disorders arise when the delicate balance of hormone production, release, and action is disturbed. These disorders fundamentally stem from three main categories: hormone excess (hypersecretion), hormone deficiency (hyposecretion), or decreased target-cell responsiveness to a hormone. Understanding these root causes is crucial for diagnosis and treatment.
I. Hormone Deficiency (Hyposecretion)
Hyposecretion refers to the inadequate production or release of a hormone by an endocrine gland. It can be classified based on the origin of the problem:
A. Primary Hyposecretion
- Definition: Occurs when the abnormality (pathology) directly lies within the hormone-producing gland itself. This gland is failing to secrete sufficient hormone despite appropriate stimulation from its tropic hormone (if applicable).
- Examples:
- Primary Hypothyroidism: The thyroid gland itself is diseased or damaged and cannot produce enough thyroid hormones (T3 and T4), leading to elevated TSH (from the pituitary trying to stimulate it).
- Type 1 Diabetes Mellitus: The pancreatic beta cells (the primary gland) are destroyed and cannot produce insulin.
- Primary Adrenal Insufficiency (Addison's Disease): The adrenal cortex is damaged and cannot produce sufficient cortisol and aldosterone.
B. Secondary Hyposecretion
- Definition: Occurs when the endocrine gland itself is intrinsically normal, but it is secreting too little hormone because of a deficiency of its tropic hormone. The problem originates in a "upstream" gland (e.g., hypothalamus or pituitary) that normally stimulates the target gland.
- Examples:
- Secondary Hypothyroidism: The anterior pituitary fails to produce enough TSH, which then leads to understimulation of a healthy thyroid gland, resulting in reduced thyroid hormone production.
- Secondary Adrenal Insufficiency: The anterior pituitary fails to produce enough ACTH, leading to understimulation of a healthy adrenal cortex.
C. Tertiary Hyposecretion: (Often considered, though not in your provided text, it's a useful distinction)
- Definition: Occurs when the problem is in the hypothalamus, leading to a deficiency of a releasing hormone, which then causes secondary hyposecretion from the pituitary, and subsequently, target gland hyposecretion.
- Example: Insufficient TRH production from the hypothalamus leads to decreased TSH from the pituitary, and ultimately decreased thyroid hormones from the thyroid gland.
D. Causes of Primary Hyposecretion (Detailed)
When the problem resides within the gland itself, the underlying reasons can be diverse:
- Genetic Defects:
- Explanation: Inherited genetic mutations can impair the gland's ability to synthesize a specific hormone, form functional receptors for tropic hormones, or develop correctly.
- Examples:
- Congenital Adrenal Hyperplasia (CAH): A group of genetic disorders affecting enzymes necessary for adrenal steroid synthesis, often leading to cortisol and aldosterone deficiency.
- Genetic defects in insulin production: Some rare forms of diabetes result from genetic errors in beta cell development or insulin synthesis.
- Genetic defects in thyroid hormone synthesis: Can lead to congenital hypothyroidism.
- Dietary Deficiencies:
- Explanation: Lack of essential nutrients required for hormone synthesis.
- Example: Lack of iodine is a classic cause of hypothyroidism. Iodine is an absolute requirement for the synthesis of thyroid hormones (T3 and T4). Chronic iodine deficiency leads to inadequate thyroid hormone production and can cause goiter (enlargement of the thyroid gland) as the pituitary gland tries to compensate by releasing more TSH.
- Chemical or Toxin Exposure:
- Explanation: Certain chemicals or environmental toxins can damage endocrine glands or interfere with hormone synthesis/release.
- Examples:
- Endocrine-Disrupting Chemicals (EDCs): A broad class of chemicals found in pesticides, plastics, and industrial byproducts that can mimic hormones, block their action, or alter their synthesis and metabolism, potentially leading to hyposecretion (or hypersecretion).
- Exposure to certain heavy metals can damage glandular tissue.
- Immunologic (Autoimmune Diseases):
- Explanation: The body's immune system mistakenly attacks and destroys its own hormone-producing cells, leading to a reduction in hormone secretion.
- Examples:
- Autoimmune Thyroiditis (Hashimoto's disease): Autoimmune antibodies attack and progressively destroy the thyroid tissue, leading to hypothyroidism.
- Type 1 Diabetes Mellitus: Autoimmune destruction of the pancreatic beta cells.
- Addison's Disease (Autoimmune Adrenalitis): Autoimmune destruction of the adrenal cortex.
- Other Disease Processes (Non-Autoimmune):
- Explanation: Various pathologies, including infections, infiltrative diseases, or cancerous growth, can destroy or impair endocrine gland tissue.
- Examples:
- Infections: Tuberculosis can destroy adrenal tissue, leading to adrenal insufficiency.
- Infiltrative diseases: Hemochromatosis (iron overload) or amyloidosis can deposit abnormal substances in endocrine glands, impairing their function.
- Cancer: A tumor (even benign) originating within or metastasizing to an endocrine gland can physically destroy healthy hormone-producing cells.
- Iatrogenic Causes:
- Explanation: Physician-induced conditions, often as a consequence of medical treatment.
- Examples:
- Surgical removal of an endocrine gland: For example, a total thyroidectomy (surgical removal of the thyroid gland due to cancer or severe hyperthyroidism) will result in permanent hypothyroidism, requiring lifelong hormone replacement.
- Radiation therapy: Radiation to an area containing an endocrine gland can damage the gland's cells.
- Medications: Certain drugs can suppress hormone synthesis or release (e.g., long-term corticosteroid use can suppress adrenal function, leading to secondary adrenal insufficiency if withdrawn abruptly).
- Idiopathic Causes:
- Explanation: When the cause of the hyposecretion is not known, despite thorough investigation. This is essentially a diagnosis of exclusion.
II. Hormone Excess (Hypersecretion)
Hypersecretion refers to the excessive production or release of a hormone. Like hyposecretion, it can be primary or secondary.
A. Primary Hypersecretion
- Definition: Occurs when the endocrine gland itself is overactive and secretes too much hormone, independent of (or even despite inhibition by) its normal regulatory signals.
- Examples:
- Primary Hyperthyroidism (e.g., Graves' Disease or a hypersecreting thyroid adenoma): The thyroid gland produces excessive thyroid hormones.
- Primary Hyperparathyroidism: A parathyroid adenoma secretes too much PTH.
B. Secondary Hypersecretion
- Definition: Occurs when an endocrine gland is stimulated to produce excessive hormone due to an oversupply of its tropic hormone from an upstream gland.
- Examples:
- Secondary Hyperthyroidism: A pituitary tumor (adenoma) secretes excessive TSH, which overstimulates a healthy thyroid gland.
- Secondary Hyperparathyroidism: Chronic kidney disease leads to low calcium, which then chronically stimulates the parathyroid glands to produce excessive PTH.
C. Tertiary Hypersecretion: (Again, useful to note)
- Definition: Occurs when the hypothalamus produces too much releasing hormone, leading to secondary hypersecretion from the pituitary, and then target gland hypersecretion.
- Example: Excessive CRH production from a hypothalamic tumor leading to increased ACTH and cortisol.
D. Causes of Hypersecretion (Detailed)
1. Tumors (Benign or Malignant):
- Explanation: Endocrine glands are prone to developing tumors (adenomas or carcinomas) that often become autonomous, meaning they ignore normal regulatory input and continuously secrete excess hormone irrespective of the body's needs or negative feedback signals.
- Examples:
- Pituitary Adenomas: Can cause hypersecretion of specific pituitary hormones (e.g., prolactinoma leading to hyperprolactinemia; somatotropinoma leading to excess GH and acromegaly/gigantism; corticotropinoma leading to excess ACTH and Cushing's disease).
- Adrenal Adenomas: Can cause excess cortisol (Cushing's syndrome) or aldosterone (Conn's syndrome).
- Thyroid Adenomas: Can cause primary hyperthyroidism.
- Ectopic Hormone Production: Some non-endocrine tumors (e.g., lung cancer) can secrete hormones (e.g., ACTH, ADH) that they don't normally produce, leading to paraneoplastic syndromes of hormone excess.
2. Immunologic Causes (Autoimmune Stimulation):
- Explanation: In certain autoimmune disorders, the immune system produces abnormal antibodies that mimic the action of a tropic hormone, constantly stimulating the target gland to overproduce its hormones.
- Example: Graves' Disease (the most common cause of hyperthyroidism): The body produces an autoantibody called Thyroid-Stimulating Immunoglobulin (TSI). TSI binds to the TSH receptors on the thyroid gland cells and mimics the action of TSH, but unlike TSH, its activity is not regulated by negative feedback. This leads to excessive stimulation of the thyroid gland, resulting in continuous overproduction and secretion of thyroid hormones.
III. Abnormal Target-Cell Responsiveness
Even if hormone secretion is normal, an endocrine disorder can arise if the target cells fail to respond appropriately to the hormone. This is often due to problems with receptors or post-receptor signaling pathways.
- A. Definition: This occurs when the hormone is present in adequate amounts, but the target cells do not respond effectively to its presence. The problem can be due to:
- Lack of Functional Receptors: The cells may not produce enough receptors, or the receptors produced may be structurally abnormal and unable to bind the hormone effectively.
- Defective Intracellular Signaling Pathways: Even if the hormone binds to its receptor, the subsequent steps within the cell that normally translate the hormone-receptor binding into a cellular response may be impaired.
- B. Examples:
- Type 2 Diabetes Mellitus (Insulin Resistance): In the early stages, the pancreas often produces normal or even elevated levels of insulin. However, the target cells (especially muscle, liver, and adipose tissue) become resistant to insulin's effects. This resistance means that despite adequate insulin, glucose uptake into cells is impaired, leading to high blood glucose. This is a classic example of decreased target-cell responsiveness.
- Testicular Feminization Syndrome (Androgen Insensitivity Syndrome, AIS):
- Explanation: In this condition, individuals are genetically male (XY chromosomes) and have testes that produce normal amounts of testosterone (a masculinizing hormone).
- Problem: However, due to a specific genetic defect, the target cells throughout the body (e.g., those involved in the development of male secondary sexual characteristics and external genitalia) either do not produce functional androgen receptors or produce receptors that are unresponsive to testosterone.
- Outcome: Although adequate testosterone is available, masculinization does not take place. The body develops female external characteristics and a female gender identity, just as if no testosterone were present, because the cells cannot "hear" the testosterone signal.
- Nephrogenic Diabetes Insipidus: The kidneys (target cells for ADH) fail to respond to antidiuretic hormone (ADH), even when ADH levels are normal or high, leading to excessive water excretion.
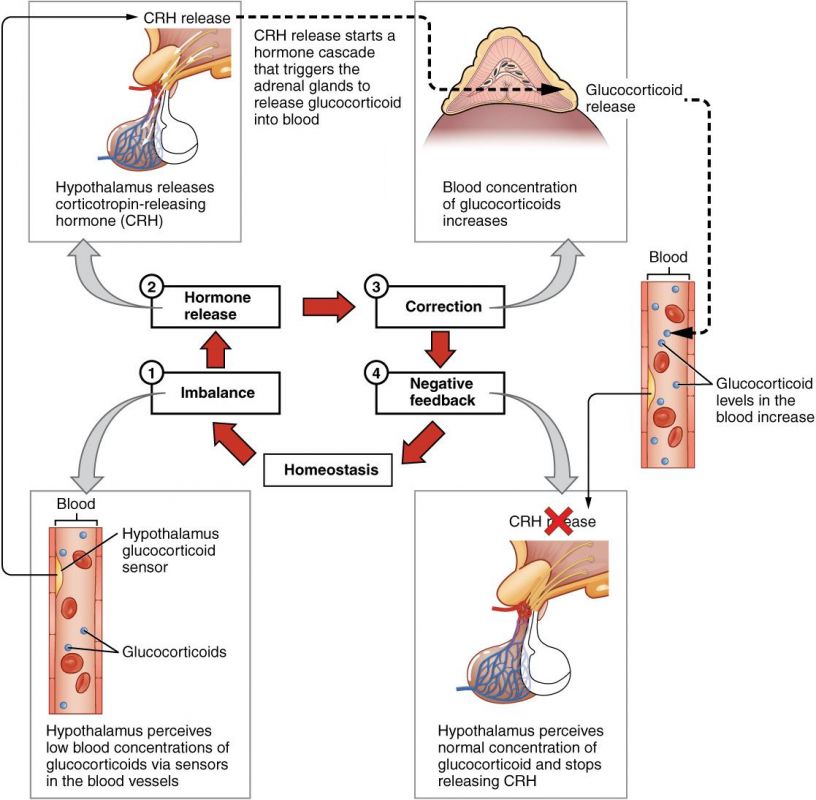
Transport, Clearance, and Mechanisms of Action of Hormones
For hormones to exert their effects, they must first be transported from their site of synthesis to target cells, then cleared from the body once their job is done. Their action at the cellular level is precise, mediated by specific receptors and intracellular signaling pathways.
I. Transport of Hormones
The method of hormone transport in the blood plasma depends primarily on their chemical nature (water-soluble vs. lipid-soluble).
A. Water-Soluble Hormones:
- Examples: Catecholamines (epinephrine, norepinephrine), peptide hormones (e.g., insulin, growth hormone, TSH, ACTH, ADH, oxytocin, gastrin, secretin).
- Transport Mechanism: These hormones are hydrophilic (water-loving) and can therefore be transported dissolved directly in the plasma. The plasma, being largely water, provides an ideal medium for their circulation.
- Implications:
- Rapid Action: They can quickly diffuse out of capillaries to reach their target cells.
- Short Half-Life: They are typically more susceptible to enzymatic degradation and renal excretion, leading to a shorter half-life in the bloodstream.
B. Lipid-Soluble Hormones (Bound Transport):
- Examples: Steroid hormones (e.g., cortisol, aldosterone, estrogen, testosterone, progesterone) and thyroid hormones (T₃ and T₄).
- Transport Mechanism: These hormones are lipophilic (lipid-loving) and hydrophobic (water-fearing). They are poorly soluble in plasma and would quickly diffuse out or be degraded if not protected. Therefore, they are predominantly transported bound to specific plasma proteins.
- Specific Binding Proteins: For instance, thyroid hormones bind to Thyroxine-Binding Globulin (TBG), Transthyretin, and Albumin. Cortisol binds to Corticosteroid-Binding Globulin (CBG) and Albumin. Sex hormones bind to Sex Hormone-Binding Globulin (SHBG) and Albumin.
- "Free" Hormone: Only a small fraction, typically less than 10% (often 0.03% to 0.1% for thyroid hormones, 1-10% for steroids), exists in the unbound or "free" form. It is this free fraction that is biologically active, capable of diffusing across capillary walls and interacting with target cell receptors. The bound portion acts as a circulating reservoir.
- Implications:
- Protection from Degradation: Binding to plasma proteins protects the hormones from rapid enzymatic degradation and filtration by the kidneys.
- Prolonged Half-Life: This significantly slows their clearance from plasma, giving them a much longer half-life (e.g., hours to days) compared to water-soluble hormones.
- Buffer System: The bound form acts as a buffer, ensuring a relatively stable concentration of free hormone available to tissues, even if secretion rates fluctuate.
- Delivery Mechanism: When free hormone levels drop (e.g., due to tissue uptake), some bound hormone dissociates from the binding proteins to maintain equilibrium, thus replenishing the free, active pool.
II. Clearance of Hormones from Plasma
Hormones, once released and having exerted their effects, must be inactivated and removed from the circulation to prevent prolonged action and allow for dynamic regulation. While the secretion of hormones is tightly controlled, their inactivation and excretion are generally not regulated in the same dynamic, feedback-loop manner.
Hormones are "cleared" from the plasma through several mechanisms:
- Metabolic Destruction by the Tissues:
- Explanation: Enzymes within target cells, liver, kidneys, and other tissues break down hormones into inactive metabolites. This is the primary route for many peptide and protein hormones, which are degraded by peptidases.
- Example: Insulin is rapidly metabolized by insulinase in the liver and kidneys. Catecholamines are broken down by Monoamine Oxidase (MAO) and Catechol-O-Methyltransferase (COMT).
- Binding with the Tissues (Cellular Uptake):
- Explanation: Hormones, particularly those that are lipid-soluble, are taken up by target cells where they bind to their receptors and are then internalized or degraded intracellularly. Even non-target cells can take up and metabolize hormones.
- Example: Thyroid hormones are taken up by cells throughout the body, where they bind to nuclear receptors and are eventually metabolized.
- Excretion by the Liver into the Bile:
- Explanation: Many lipid-soluble hormones (especially steroid hormones) and their metabolites are conjugated in the liver (e.g., with glucuronic acid or sulfate) to make them more water-soluble. These conjugated forms can then be excreted by the liver into the bile, which passes into the intestine and is eventually eliminated in the feces.
- Example: Estrogen and testosterone metabolites are often excreted via the bile.
- Excretion by the Kidneys into the Urine:
- Explanation: Water-soluble hormones (like most peptide hormones) and the water-soluble metabolites of lipid-soluble hormones are readily filtered by the glomeruli in the kidneys and excreted in the urine.
- Example: Many small peptide hormones and the conjugated forms of steroid and thyroid hormones are eliminated renally. Renal failure can significantly prolong the half-life of many hormones.
III. Mechanisms of Action of Hormones & Role of Receptors
Hormones do not act directly on target cells in a generalized way. Instead, their actions are highly specific and mediated by the interaction with specialized proteins called receptors.
A. Hormone Receptors:
- Definition: Hormone receptors are large protein molecules present in target cells. They are the molecular "locks" that specific hormones (the "keys") fit into.
- Specificity: Each receptor is highly specific for a particular hormone or a class of hormones. This means a hormone can act on a target cell only if that target cell possesses the specific receptor for that hormone. Non-target cells, lacking the receptor, will not respond to the hormone.
- Abundance: Target cells typically have a large number of receptors, ranging from 2,000 to 100,000 or more per cell. The number and sensitivity of these receptors can be dynamically regulated (up-regulation or down-regulation) in response to varying hormone concentrations or physiological states.
- Hormone-Receptor Complex: When a hormone binds to its receptor, it forms a hormone-receptor complex. This binding is reversible and initiates a cascade of intracellular events that ultimately lead to the characteristic cellular response.
B. Location of Hormone Receptors (Determined by Hormone Solubility):
The chemical nature of the hormone dictates where its receptor will be located within the target cell.
1. Cell Membrane (Surface Receptors)
- Hormones: Receptors for protein/peptide hormones (e.g., insulin, growth hormone, TSH, ADH) and adrenal medullary hormones (catecholamines) (epinephrine, norepinephrine). These hormones are water-soluble and cannot easily cross the lipid bilayer of the cell membrane.
- Mechanism: The hormone binds to its receptor on the outer surface of the cell membrane. This binding then triggers a conformational change in the receptor, activating intracellular signaling pathways.
2. Cytoplasm (Intracellular Receptors)
- Hormones: Receptors for steroid hormones (e.g., cortisol, aldosterone, estrogen, testosterone, progesterone). These hormones are lipid-soluble and can readily diffuse across the cell membrane into the cytoplasm.
- Mechanism: Once inside the cytoplasm, the steroid hormone binds to its specific receptor. The hormone-receptor complex then typically translocates into the nucleus.
3. Nucleus (Intracellular Receptors)
- Hormones: Receptors for thyroid hormones (T₃ and T₄). Although lipid-soluble and able to cross the cell membrane, thyroid hormone receptors are predominantly located within the nucleus, often bound to DNA even in the absence of hormone.
- Mechanism: Thyroid hormones enter the cell and then the nucleus, where they bind to their receptors.
C. Mechanisms of Action (How the Hormone-Receptor Complex Triggers a Response)
The binding of a hormone to its receptor initiates a series of events that alter cellular function. These mechanisms vary depending on the receptor location.
1. By Altering Permeability of Cell Membrane (Ligand-Gated Ion Channels):
- Explanation: Some hormones (or neurotransmitters) bind to receptors that are directly coupled to ion channels. When the hormone binds, it causes a conformational change that opens or closes the ion channel, altering the permeability of the cell membrane to specific ions (e.g., Na⁺, K⁺, Ca²⁺). This change in ion flow can alter the cell's electrical potential or trigger downstream signaling.
- Example: While more characteristic of neurotransmitter action at synapses (e.g., acetylcholine at the neuromuscular junction opening Na⁺ channels), some hormones can influence ion channels. For instance, insulin can indirectly affect ion transport, and some growth factors can modulate ion channel activity.
2. By Activating Intracellular Enzyme - Action Through Second Messenger Systems (for Membrane-Bound Receptors):
- Explanation: This is the most common mechanism for water-soluble hormones. Since they cannot enter the cell, they act as "first messengers." Their binding to a surface receptor activates an enzyme or a G-protein within the membrane. This activation then leads to the production of intracellular "second messengers" that amplify the signal and relay it to various effector proteins within the cell.
- Key Second Messenger Systems:
- cAMP (Cyclic Adenosine Monophosphate): Hormone binds to receptor → activates G-protein → activates adenylyl cyclase → converts ATP to cAMP → cAMP activates protein kinase A → phosphorylation of various proteins → cellular response. (e.g., Glucagon, ACTH, TSH, LH, FSH, ADH [V2 receptor], Epinephrine [β-adrenergic receptor]).
- cGMP (Cyclic Guanosine Monophosphate): Similar to cAMP, but uses guanylyl cyclase. (e.g., ANP, nitric oxide).
- IP₃ (Inositol Trisphosphate) and DAG (Diacylglycerol): Hormone binds to receptor → activates G-protein → activates phospholipase C → cleaves PIP₂ into IP₃ and DAG. IP₃ mobilizes intracellular Ca²⁺, and DAG activates protein kinase C. (e.g., TRH, GnRH, ADH [V1 receptor], Angiotensin II, Epinephrine [α₁-adrenergic receptor]).
- Ca²⁺-Calmodulin Complex: Increased intracellular Ca²⁺ (from IP₃ or other sources) binds to the protein calmodulin, forming a complex that then activates various enzymes to produce a cellular response. Ca²⁺ itself is often considered a second messenger.
- Signal Amplification: Second messenger systems allow for significant signal amplification: one hormone molecule can lead to the production of many second messenger molecules, which in turn activate many downstream targets, resulting in a large cellular response.
3. By Acting on Genes (for Intracellular Receptors):
- Explanation: This is the primary mechanism for lipid-soluble hormones (steroids and thyroid hormones) that bind to intracellular receptors (cytoplasmic or nuclear). The hormone-receptor complex directly interacts with the cell's genetic material.
- Mechanism:
- The hormone diffuses into the cell and binds to its receptor, often causing the receptor to change shape.
- The hormone-receptor complex then binds to specific DNA sequences called Hormone Response Elements (HREs) located in the promoter regions of target genes.
- This binding can either activate or repress the transcription of specific genes, leading to changes in the production of mRNA.
- The altered mRNA levels then lead to changes in the synthesis of specific proteins (enzymes, structural proteins, regulatory proteins), which ultimately mediate the long-term physiological effects of the hormone.
- Examples: All steroid hormones (cortisol, estrogen, testosterone, aldosterone) and thyroid hormones act primarily by altering gene expression, which explains their relatively slower but more sustained effects on target cells.
https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Hormone Regulation, Disorders & Transport
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Hormone Regulation, Disorders & Transport
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Endocrine System Physiology
Endocrine System
Introduction to the Endocrine System
The endocrine system is a crucial communication network within the body, working in conjunction with the nervous system to maintain homeostasis and regulate various physiological processes. Unlike the nervous system, which primarily uses electrical signals for rapid, short-duration responses, the endocrine system employs chemical messengers called hormones that travel through the bloodstream, eliciting slower, more sustained responses.
Overall Functions of the Endocrine System
The endocrine system is responsible for a wide array of vital functions, ensuring the body's proper growth, development, and adaptation. Its key roles include:
- Regulating Nutrient Metabolism and Water & Electrolyte Balance:
- This is fundamental for maintaining a constant internal environment (homeostasis). Hormones control how the body processes carbohydrates, fats, and proteins for energy, storage, or conversion. They also play a critical role in balancing water and crucial ions like sodium, potassium, and calcium, essential for cell function and overall fluid balance.
- Inducing Adaptive Changes to Help the Body Cope with Stressful Situations:
- The endocrine system orchestrates the body's "fight or flight" response and longer-term stress adaptation. Hormones like cortisol and adrenaline prepare the body for perceived threats by mobilizing energy stores and altering physiological functions.
- Promoting Smooth, Sequential Growth and Development:
- From fetal development through childhood and adolescence, hormones meticulously control the growth of tissues, organs, and the entire body. They also dictate the timing and progression of puberty and the maturation of reproductive organs.
- Controlling Reproduction:
- Hormones are central to all aspects of reproduction, including the development of sex characteristics, the regulation of menstrual cycles in females, sperm production in males, pregnancy, childbirth, and lactation.
- Regulating Red Blood Cell Production:
- Erythropoietin, a hormone primarily produced by the kidneys, stimulates the bone marrow to produce red blood cells, which are vital for oxygen transport.
- Controlling and Integrating Activities of the Circulatory and Digestive Systems (along with the Autonomic Nervous System):
- Hormones influence heart rate, blood pressure, blood vessel tone, and blood volume (circulatory system). In the digestive system, they regulate appetite, enzyme secretion, motility, and nutrient absorption. This integration ensures that these systems function efficiently and are coordinated with the body's overall needs.
Definitions
- Hormone: A hormone is a chemical messenger synthesized by specialized cells, typically within endocrine glands. These messengers are transported by the blood to target organs or tissues (their "site of action"), where they bind to specific receptors and cause an alteration in the function of the cell. An example is Growth Hormone, which promotes overall body growth.
- Endocrine Glands: Endocrine glands are specialized organs or tissues that synthesize and release classical hormones directly into the blood. They are often referred to as ductless glands because, unlike exocrine glands (which release secretions through ducts), their hormones are secreted directly into the bloodstream without the need for ducts. Examples include the thyroid gland, adrenal glands, pituitary gland, and pancreas (for its endocrine function).
Tropic Hormones
- Definition: Tropic hormones are a special class of hormones that have as their primary function the regulation of hormone secretion by another endocrine gland. The term "tropic" means "nourishing," which reflects their role in stimulating and maintaining their endocrine target tissues.
- Function: Tropic hormones not only stimulate the target gland to produce and secrete its own hormones but also maintain the structural integrity (trophic effect) of that gland. Without their tropic hormone, target glands can atrophy (shrink) and become underactive.
- Example: Thyroid-Stimulating Hormone (TSH): TSH is a tropic hormone secreted by the anterior pituitary gland. It stimulates the thyroid gland to secrete thyroid hormones (T3 and T4). Additionally, TSH maintains the normal size and health of the thyroid gland. In the absence of TSH, the thyroid gland would atrophy (shrink) and produce very low levels of its hormones, leading to hypothyroidism.
Complexity of Endocrine Function
The endocrine system, while seemingly straightforward in its definition, exhibits remarkable complexity in its operations:
- A Single Endocrine Gland May Produce Multiple Hormones: Many glands are multi-functional. For instance, the anterior pituitary gland is a master gland that secretes at least six different hormones, each with distinct target tissues and functions (e.g., TSH, Growth Hormone, ACTH, FSH, LH, Prolactin).
- A Single Hormone May Be Secreted by More Than One Endocrine Gland: Some hormones are produced by different endocrine sources, sometimes serving different roles depending on their origin. For example, somatostatin is secreted by both the hypothalamus (where it inhibits Growth Hormone release) and the pancreas (where it acts as a paracrine to inhibit insulin and glucagon secretion). In the stomach, somatostatin also acts as a paracrine to inhibit gastric acid secretion.
- A Single Hormone May Have More Than One Type of Target Cell and Induce Multiple Effects: Hormones often exert diverse effects by binding to different subtypes of receptors on various target cells.
- Example: Vasopressin (Antidiuretic Hormone - ADH):
- Water Reabsorption: Vasopressin promotes water reabsorption by the kidney tubules by binding with V2 receptors on the distal and collecting tubular cells. This helps conserve body water.
- Vasoconstriction: It also causes vasoconstriction of arterioles throughout the body by binding with V1 receptors on arteriolar smooth muscle. This helps increase blood pressure.
- Example: Vasopressin (Antidiuretic Hormone - ADH):
- Hormones with Multiple Target-Cell Types Can Coordinate Activities Toward a Common End: Sometimes, a single hormone's actions on different tissues converge to achieve a unified physiological goal.
- Example: Insulin: After a meal, insulin acts on muscle cells, liver cells, and fat cells. In muscle and fat, it promotes glucose uptake and storage. In the liver, it promotes glucose conversion to glycogen and fat synthesis. All these actions work in concert to store nutrients after absorption of a meal, thus lowering blood glucose.
- The Rate of Secretion of Some Hormones Varies Considerably in a Cyclic Pattern (Temporal Coordination): Endocrine systems often provide temporal (time) coordination of function, with hormone levels fluctuating in predictable cycles. This is particularly evident in:
- Reproductive cycles: Such as the menstrual cycle, where highly specific patterns of hormone secretion (e.g., estrogens, progesterone, LH, FSH) are required for normal ovarian and uterine function.
- Circadian rhythms: Many hormones follow a 24-hour cycle (e.g., cortisol, melatonin).
- A Single Target Cell May Be Influenced by More Than One Hormone: Target cells often possess receptors for multiple hormones, allowing for complex regulation and fine-tuning of their responses.
- Example: Liver Cells:
- Insulin: Promotes the conversion of glucose into glycogen within liver cells by stimulating specific hepatic enzymes.
- Glucagon: By activating different hepatic enzymes, glucagon enhances the degradation of glycogen back into glucose within liver cells. This antagonistic interaction allows precise control of blood glucose levels.
- Example: Liver Cells:
- The Same Chemical Messenger May Be Either a Hormone or a Neurotransmitter: The distinction between chemical messengers can sometimes be blurred, depending on their source and mode of delivery.
- Example: Norepinephrine:
- Hormone: Secreted by the adrenal medulla (an endocrine gland) into the bloodstream, acting on distant target cells.
- Neurotransmitter: Released by sympathetic postganglionic nerve fibers directly onto target cells at a synapse.
- Example: Norepinephrine:
- Some Organs are Solely Endocrine in Function, While Others Perform Non-Endocrine Functions as Well:
- Solely Endocrine: Organs like the anterior pituitary specialize exclusively in hormone secretion.
- Mixed Function: Other organs have both endocrine and non-endocrine roles. For example, the testes produce sperm (non-endocrine, reproductive function) and also secrete the male sex hormone testosterone (endocrine function). Similarly, the pancreas has both exocrine (digestive enzyme secretion) and endocrine (insulin, glucagon secretion) functions.
Glands and Hormones
I. Central Endocrine Glands
These glands, primarily located in the brain, serve as crucial regulators, often controlling the activity of other endocrine glands throughout the body.
A. Hypothalamus
The hypothalamus is a neuroendocrine organ, acting as the interface between the nervous and endocrine systems. It produces a variety of releasing and inhibiting hormones that primarily regulate the anterior pituitary gland.
- Hormones:
- Releasing Hormones:
- Thyrotropin-Releasing Hormone (TRH): Stimulates the release of TSH and prolactin from the anterior pituitary.
- Corticotropin-Releasing Hormone (CRH): Stimulates the release of ACTH from the anterior pituitary.
- Gonadotropin-Releasing Hormone (GnRH): Stimulates the release of FSH and LH from the anterior pituitary.
- Growth Hormone-Releasing Hormone (GHRH): Stimulates the release of GH from the anterior pituitary.
- Prolactin-Releasing Hormone (PRH): Stimulates the release of prolactin from the anterior pituitary (its existence as a distinct peptide is debated; TRH can also stimulate prolactin).
- Inhibiting Hormones:
- Somatostatin (Growth Hormone-Inhibiting Hormone, GHIH): Inhibits the release of GH and TSH from the anterior pituitary.
- Dopamine (Prolactin-Inhibiting Hormone, PIH): Inhibits the release of prolactin from the anterior pituitary.
- Releasing Hormones:
- Target Cells: Primarily the anterior pituitary gland.
- Major Functions: Control Release of Anterior Pituitary Hormones: The hypothalamus integrates neural input and secretes specific releasing or inhibiting hormones into the hypophyseal portal system, which then travel to the anterior pituitary, either stimulating or inhibiting the secretion of its own hormones. This forms the basis of many endocrine axes (e.g., HPA axis, HPT axis, HPG axis).

B. Posterior Pituitary (Neurohypophysis)
The posterior pituitary does not synthesize hormones; it stores and releases two hormones produced by neurosecretory cells in the hypothalamus (specifically the supraoptic and paraventricular nuclei). These hormones are transported down axons to the posterior pituitary for storage and release.
1. Vasopressin (Antidiuretic Hormone, ADH)
- Synthesis Site: Primarily supraoptic nucleus of the hypothalamus.
- Target Cells:
- Kidney tubules (collecting ducts and distal tubules): Primary target.
- Arterioles: Smooth muscle cells.
- Major Functions:
- Increases H₂O Reabsorption: ADH increases the permeability of the collecting ducts and distal tubules to water, leading to increased water reabsorption from the filtrate back into the blood. This concentrates the urine and conserves body water. It achieves this by inserting aquaporin-2 water channels into the luminal membrane of these cells.
- Produces Vasoconstriction: At high concentrations, ADH can cause generalized constriction of arterioles, leading to an increase in total peripheral resistance and arterial blood pressure. This pressor effect is particularly important in cases of severe hemorrhage.
2. Oxytocin
- Synthesis Site: Primarily paraventricular nucleus of the hypothalamus.
- Target Cells:
- Uterus: Smooth muscle cells.
- Mammary Glands (Breasts): Myoepithelial cells surrounding milk-producing alveoli.
- Major Functions:
- Increases Contractility (Uterus): Stimulates uterine smooth muscle contraction, particularly important during childbirth (positive feedback loop during labor).
- Causes Milk Ejection (Let-down Reflex): Stimulates the contraction of myoepithelial cells in the mammary glands, forcing milk from the alveoli into the ducts, making it available for suckling. This is a neuroendocrine reflex triggered by suckling.
C. Anterior Pituitary (Adenohypophysis)
The anterior pituitary synthesizes and secretes six major peptide hormones, often under the control of hypothalamic releasing and inhibiting hormones. Many of these are tropic hormones, regulating other endocrine glands.
1. Thyroid-Stimulating Hormone (TSH, Thyrotropin)
- Target Cells: Thyroid follicular cells.
- Major Functions:
- Stimulates T₃ and T₄ Secretion: TSH binds to receptors on thyroid follicular cells, stimulating all steps of thyroid hormone synthesis and release (iodide trapping, oxidation, organification, coupling, endocytosis, proteolysis, and release of T₃ and T₄).
- Maintains Thyroid Gland Size: TSH has a trophic effect on the thyroid gland, maintaining its growth and vascularity.
2. Adrenocorticotropic Hormone (ACTH, Corticotropin)
- Target Cells: Primarily the zona fasciculata and zona reticularis of the adrenal cortex.
- Major Functions:
- Stimulates Cortisol Secretion: ACTH stimulates these layers of the adrenal cortex to synthesize and secrete glucocorticoids (e.g., cortisol) and adrenal androgens.
- Maintains Adrenal Cortex Integrity: It also has a trophic effect on these layers, maintaining their size and function.
3. Growth Hormone (GH, Somatotropin)
- Target Cells:
- Directly: Bone and soft tissues (skeletal muscle, adipose tissue).
- Indirectly (via IGF-I): Liver (primarily produces IGF-I), bone and soft tissues.
- Major Functions:
- Promotes Overall Growth: GH is essential for normal physical growth throughout childhood and adolescence. Many of its growth-promoting effects are mediated indirectly by stimulating the liver (and other tissues) to produce Insulin-like Growth Factor-I (IGF-I). IGF-I then acts on target cells to promote protein anabolism (increased amino acid uptake and protein synthesis), cell proliferation, and tissue growth, particularly in bone (epiphyseal plate growth) and soft tissues.
- Direct Metabolic Effects: GH also has direct metabolic effects that include:
- Mobilizing fat stores: Increases lipolysis in adipose tissue, leading to increased fatty acid use for energy.
- Conserving glucose: Decreases glucose uptake by tissues and increases glucose output by the liver, thus increasing blood glucose levels (diabetogenic effect).
4. Follicle-Stimulating Hormone (FSH)
- Target Cells (Females): Ovarian follicles.
- Major Functions (Females):
- Promotes Follicular Growth and Development: Stimulates the growth of ovarian follicles in the ovary.
- Stimulates Estrogen Secretion: Acts on granulosa cells within the follicles to promote estrogen synthesis and secretion.
- Target Cells (Males): Seminiferous tubules in testes (specifically Sertoli cells).
- Major Functions (Males):
- Stimulates Sperm Production (Spermatogenesis): Essential for supporting the development of sperm.
5. Luteinizing Hormone (LH)
- Target Cells (Females): Ovarian follicle and corpus luteum.
- Major Functions (Females):
- Stimulates Ovulation: Triggers the rupture of the mature follicle and the release of the egg.
- Corpus Luteum Development: Promotes the formation of the corpus luteum from the ruptured follicle.
- Estrogen and Progesterone Secretion: Stimulates the corpus luteum to secrete progesterone and estrogen after ovulation.
- Target Cells (Males): Interstitial cells of Leydig in testes.
- Major Functions (Males):
- Stimulates Testosterone Secretion: Acts on Leydig cells to promote the synthesis and secretion of testosterone.
6. Prolactin (PRL)
- Target Cells (Females): Mammary glands.
- Major Functions (Females):
- Promotes Breast Development: Contributes to the development of glandular tissue in the breasts during puberty and pregnancy.
- Stimulates Milk Secretion (Lactogenesis): Initiates and maintains milk production after childbirth.
- Target Cells (Males): Unclear, but may play a role in testicular function.
- Major Functions (Males): Uncertain, but high levels can inhibit GnRH secretion, leading to reduced testosterone and infertility.
D. Pineal Gland
The pineal gland is a small endocrine gland located deep in the brain. Its primary hormone is melatonin, which plays a role in regulating circadian rhythms.
- Melatonin
- Target Cells: Brain (anterior pituitary, suprachiasmatic nucleus), reproductive organs, immune system, and possibly others.
- Major Functions:
- Entrains Body's Biological Rhythm (Circadian Rhythm): Melatonin secretion is high during darkness and low during light, providing the body with a "darkness signal." This helps synchronize the body's internal clock (circadian rhythm) with the external light-dark cycle, influencing sleep-wake cycles.
- Inhibits Gonadotropins: Its reduction (as light exposure increases with age) is thought to be involved in the initiation of puberty. High melatonin levels in childhood may inhibit the release of GnRH.
- Antioxidant & Immune Enhancement: Functions as a potent antioxidant, protecting cells from oxidative stress. It also influences immune function.
II. Peripheral Endocrine Glands
These glands are distributed throughout the body and have diverse, specialized functions.
A. Thyroid Gland
Located in the neck, the thyroid gland produces thyroid hormones that regulate metabolism and calcitonin that influences calcium balance.
- Tetraiodothyronine (T₄, Thyroxine) & Triiodothyronine (T₃)
- Synthesized by: Thyroid follicular cells.
- Target Cells: Most cells in the body.
- Major Functions:
- Increases Metabolic Rate: Increases oxygen consumption and heat production, boosting the basal metabolic rate. This affects the metabolism of carbohydrates, fats, and proteins.
- Essential for Normal Growth and Nerve Development: Critical for proper nervous system development in children and for maintaining normal neurological function in adults. It also plays a permissive role in the actions of Growth Hormone.
- Calcitonin
- Synthesized by: Thyroid C cells (parafollicular cells).
- Target Cells: Bone (osteoclasts primarily).
- Major Functions:
- Decreases Plasma Ca²⁺ Concentration: When blood calcium levels are high, calcitonin is released. It inhibits osteoclast activity (which break down bone and release calcium) and stimulates osteoblast activity (which build bone and store calcium), thereby promoting calcium deposition into bone and lowering blood calcium levels. Its physiological role in humans is generally considered less significant than PTH in normal calcium homeostasis.
B. Adrenal Glands
Two glands located atop the kidneys, each composed of an outer cortex and an inner medulla, producing different sets of hormones.
1. Adrenal Cortex
The outer part of the adrenal gland, divided into three zones, each producing distinct steroid hormones.
Zona Glomerulosa
- Hormone: Aldosterone (a mineralocorticoid).
- Target Cells: Kidney tubules (distal and collecting tubules).
- Major Functions:
- Increases Na⁺ Reabsorption: Stimulates the reabsorption of sodium ions from the kidney tubules into the blood.
- Increases K⁺ Secretion: Promotes the secretion of potassium ions from the blood into the kidney tubules for excretion. These actions contribute to maintaining blood volume, blood pressure, and electrolyte balance.
Zona Fasciculata and Zona Reticularis
- Hormone: Cortisol (a glucocorticoid).
- Target Cells: Most cells in the body.
- Major Functions:
- Increases Blood Glucose: Increases blood glucose levels by promoting gluconeogenesis (glucose formation from non-carbohydrate sources) in the liver and reducing glucose uptake by peripheral tissues (at the expense of protein and fat stores).
- Contributes to Stress Adaptation: Plays a critical role in the body's response to stress by mobilizing energy stores and suppressing immune and inflammatory responses.
- Immunosuppressive and Anti-inflammatory: Pharmacological doses are used to suppress the immune system and reduce inflammation.
- Hormone: Androgens (Dehydroepiandrosterone, DHEA).
- Target Cells: Primarily bone and brain (in females, other tissues convert DHEA into more potent androgens or estrogens).
- Major Functions:
- Pubertal Growth Spurt and Sex Drive: In females, adrenal androgens contribute to the pubertal growth spurt and are the primary source of sex drive (libido). In males, their role is less significant compared to testicular androgens.
2. Adrenal Medulla
The inner part of the adrenal gland, derived from neural tissue, secreting catecholamines.
- Hormones: Epinephrine (Adrenaline) and Norepinephrine (Noradrenaline) (catecholamines).
- Target Cells: Sympathetic receptor sites throughout the body (adrenergic receptors on various organs and tissues).
- Major Functions:
- Reinforce Sympathetic Nervous System: Act as hormones to augment and prolong the effects of sympathetic nervous system activation, preparing the body for "fight or flight."
- Contribute to Stress Adaptation: Mobilize energy stores (increase glucose and fatty acids in blood), increase heart rate and contractility, vasoconstrict/vasodilate specific blood vessels to redistribute blood flow, and dilate airways.
- Blood Pressure Regulation: Contribute to the acute regulation of blood pressure.
C. Endocrine Pancreas (Islets of Langerhans)
Clusters of endocrine cells within the pancreas that regulate blood glucose levels.
1. Insulin (from β cells)
- Target Cells: Most cells in the body (especially skeletal muscle, adipose tissue, and liver).
- Major Functions:
- Promotes Cellular Uptake, Use, and Storage of Absorbed Nutrients: Primarily lowers blood glucose by:
- Increasing glucose uptake by insulin-sensitive cells (muscle, adipose tissue) through the insertion of GLUT4 transporters.
- Promoting glycogenesis (glucose to glycogen) in liver and muscle.
- Promoting fat synthesis (lipogenesis) and inhibiting lipolysis in adipose tissue.
- Promoting protein synthesis.
- Overall Anabolic Hormone: Dominant during the absorptive (fed) state.
- Promotes Cellular Uptake, Use, and Storage of Absorbed Nutrients: Primarily lowers blood glucose by:
2. Glucagon (from α cells)
- Target Cells: Most cells in the body (primarily liver, but also adipose tissue).
- Major Functions:
- Maintains Nutrient Levels in Blood During the Postabsorptive State: Primarily raises blood glucose by:
- Promoting glycogenolysis (glycogen breakdown to glucose) in the liver.
- Promoting gluconeogenesis in the liver.
- Increasing lipolysis in adipose tissue to provide fatty acids for energy and glycerol for gluconeogenesis.
- Overall Catabolic Hormone: Dominant during the post-absorptive (fasting) state.
- Maintains Nutrient Levels in Blood During the Postabsorptive State: Primarily raises blood glucose by:
3. Somatostatin (from δ cells)
- Target Cells: Digestive system (pancreatic α and β cells, GI tract cells).
- Major Functions:
- Inhibits Digestion and Absorption of Nutrients: Acts as a local paracrine to inhibit the secretion of insulin and glucagon from the pancreas.
- It also suppresses the motility and secretion of the GI tract, slowing down the rate of nutrient absorption.
D. Parathyroid Gland
Typically four small glands located on the posterior surface of the thyroid gland, crucial for calcium homeostasis.
- Parathyroid Hormone (PTH)
- Target Cells: Bone, kidneys, and intestine (indirectly via vitamin D activation).
- Major Functions:
- Increases Plasma Ca²⁺ Concentration: This is its primary role. It achieves this by:
- Bone: Stimulating osteoclast activity, leading to bone resorption and release of calcium (and phosphate) into the blood.
- Kidneys: Increasing renal reabsorption of calcium (reducing urinary calcium excretion) and inhibiting renal reabsorption of phosphate (increasing urinary phosphate excretion).
- Intestine: Stimulating the kidneys to convert vitamin D into its active form (calcitriol), which then increases intestinal absorption of calcium.
- Decreases Plasma PO₄²⁻ Concentration: By increasing phosphate excretion by the kidneys.
- Increases Plasma Ca²⁺ Concentration: This is its primary role. It achieves this by:
E. Female Gonads (Ovaries)
Paired reproductive organs in females, producing female sex hormones.
- Estrogen (e.g., Estradiol)
- Target Cells: Female sex organs and body as a whole.
- Major Functions:
- Promotes Follicular Development: Essential for the growth and maturation of ovarian follicles.
- Governs Development of Female Secondary Sexual Characteristics: Responsible for breast development, widening of hips, and characteristic fat distribution.
- Stimulates Uterine and Breast Growth: Promotes endometrial proliferation in the uterus and breast glandular tissue development.
- Progesterone
- Target Cells: Bone, Uterus.
- Major Functions:
- Prepares for Pregnancy: Induces secretory changes in the endometrium of the uterus to prepare it for implantation and supports pregnancy.
- Promotes Closure of Epiphyseal Plate: Contributes to the cessation of linear bone growth during puberty.
- Inhibin
- Target Cells: Anterior pituitary.
- Major Functions:
- Inhibits Secretion of FSH: Provides negative feedback to the anterior pituitary, helping to regulate FSH levels.
F. Male Gonads (Testes)
Paired reproductive organs in males, producing male sex hormones.
- Testosterone
- Target Cells: Male sex organs and body as a whole.
- Major Functions:
- Stimulates Sperm Production (Spermatogenesis): Essential for the maturation of sperm.
- Governs Development of Male Secondary Sexual Characteristics: Responsible for features like facial hair growth, deepening of voice, and increased muscle mass.
- Promotes Sex Drive (Libido): Influences sexual desire.
- Target Cells: Bone.
- Major Functions:
- Enhances Pubertal Growth Spurt; Promotes Closure of the Epiphyseal Plate: Contributes to rapid growth during puberty and eventually stops linear bone growth.
- Inhibin
- Target Cells: Anterior pituitary.
- Major Functions:
- Inhibits Secretion of FSH: Provides negative feedback to the anterior pituitary, helping to regulate FSH levels.
G. Placenta (during pregnancy)
A temporary organ that connects the mother to the developing fetus, also acting as an endocrine gland.
- Estrogen (e.g., Estriol) and Progesterone
- Target Cells: Female sex organs (uterus, breasts).
- Major Functions:
- Help Maintain Pregnancy: Essential for maintaining the uterine lining and preventing premature contractions.
- Prepare Breasts for Lactation: Contribute to mammary gland development for milk production.
- Human Chorionic Gonadotropin (hCG)
- Target Cells: Ovarian corpus luteum.
- Major Functions:
- Maintains Corpus Luteum of Pregnancy: Prevents the degeneration of the corpus luteum, ensuring continued progesterone (and estrogen) production in early pregnancy until the placenta can take over hormone synthesis. This is the hormone detected in pregnancy tests.
III. Organs With Secondary Endocrine Functions
Many organs primarily known for non-endocrine roles also produce and secrete hormones that have significant systemic effects.
A. Kidneys
Beyond their role in filtration and waste excretion, the kidneys produce hormones vital for blood pressure and red blood cell production.
- Renin (by activating angiotensinogen)
- Produced by: Juxtaglomerular cells.
- Target Cells: Acts on angiotensinogen (a plasma protein from the liver) to produce angiotensin I, which is then converted to angiotensin II. Angiotensin II then targets the zona glomerulosa of the adrenal cortex.
- Major Functions:
- Stimulates Aldosterone Secretion: Angiotensin II is a powerful stimulus for aldosterone release from the adrenal cortex.
- Potent Vasoconstrictor: Angiotensin II directly constricts blood vessels throughout the body, increasing total peripheral resistance and blood pressure.
- Stimulates Thirst: Contributes to fluid intake regulation.
- (Part of the Renin-Angiotensin-Aldosterone System - RAAS, which regulates blood pressure and fluid balance).
- Erythropoietin
- Produced by: Peritubular cells in the renal cortex.
- Target Cells: Bone marrow (specifically erythrocyte progenitor cells).
- Major Functions:
- Stimulates Erythrocyte Production: Acts on the bone marrow to stimulate the proliferation and differentiation of red blood cell precursors, leading to increased red blood cell mass. This is a crucial response to hypoxia.
B. Stomach
The stomach is involved in digestion but also produces hormones that regulate appetite and digestive processes.
- Ghrelin
- Produced by: P/D1 cells in the fundus of the stomach.
- Target Cells: Hypothalamus (arcuate nucleus).
- Major Functions:
- Signals Hunger, Stimulates Appetite: Often called the "hunger hormone," its levels rise before meals and fall after, signaling the brain to initiate food intake.
- Gastrin
- Produced by: G cells in the pyloric antrum.
- Target Cells: Digestive tract exocrine glands (e.g., parietal cells, chief cells) and smooth muscles of the stomach, pancreas, liver, gallbladder.
- Major Functions:
- Controls Motility and Secretion: Primarily stimulates the secretion of gastric acid (HCl) by parietal cells and promotes gastric motility, facilitating digestion and absorptive processes. It also has trophic effects on the gastric mucosa.
C. Small Intestine
The small intestine produces several hormones that coordinate digestion and nutrient absorption.
- Secretin and Cholecystokinin (CCK)
- Secretin produced by: S cells in the duodenum.
- CCK produced by: I cells in the duodenum and jejunum.
- Target Cells: Primarily endocrine pancreas (for insulin secretion), but also exocrine pancreas, gallbladder, stomach.
- Major Functions:
- Stimulates Insulin Secretion: While their primary roles are related to exocrine secretions (e.g., secretin stimulates bicarbonate-rich fluid from the pancreas; CCK stimulates enzyme-rich fluid from the pancreas and gallbladder contraction), they also have an "incretin effect" indirectly stimulating insulin secretion in response to nutrient presence.
- Glucose-Dependent Insulinotropic Peptide (GIP, Gastric Inhibitory Peptide)
- Produced by: K cells in the duodenum and jejunum.
- Target Cells: Endocrine pancreas (β cells).
- Major Functions:
- Stimulates Insulin Secretion: A major incretin hormone, GIP significantly enhances insulin release from pancreatic β-cells in response to oral glucose intake, anticipating the rise in blood glucose.
- Peptide YY₃-₃₆ (PYY)
- Produced by: L cells in the ileum and colon.
- Target Cells: Hypothalamus (arcuate nucleus).
- Major Functions:
- Signals Satiety, Suppresses Appetite: Released post-prandially, PYY helps to signal fullness and reduce food intake, contributing to short-term satiety.
D. Liver
The liver is a metabolic powerhouse and also an endocrine organ.
- Insulin-like Growth Factor-I (IGF-I, Somatomedin C)
- Produced by: Liver (primarily) in response to Growth Hormone.
- Target Cells: Bone and soft tissues.
- Major Functions:
- Promotes Growth: Mediates many of the growth-promoting effects of Growth Hormone, stimulating cell proliferation, protein synthesis, and linear growth, particularly in cartilage and bone.
- Thrombopoietin
- Produced by: Liver (primary site) and kidneys.
- Target Cells: Bone marrow (megakaryocytes).
- Major Functions:
- Stimulates Platelet Production: Regulates the production of platelets by stimulating the growth and differentiation of megakaryocytes in the bone marrow.
- Hepcidin
- Produced by: Liver.
- Target Cells: Intestine (duodenal enterocytes), macrophages.
- Major Functions:
- Inhibits Iron Absorption into Blood: The master regulator of iron homeostasis. It controls the release of iron from enterocytes, macrophages, and hepatocytes into the plasma, thereby reducing intestinal iron absorption and preventing iron overload.
E. Skin
The skin plays a crucial role in vitamin D synthesis, a hormone precursor.
- Vitamin D (active form, Calcitriol)
- Precursor produced in: Skin (via UV light exposure from 7-dehydrocholesterol). Activated in liver and kidneys.
- Target Cells: Intestine (enterocytes), bone, kidneys.
- Major Functions:
- Increases Absorption of Ingested Ca²⁺ and PO₄²⁻: The active form of vitamin D (1,25-dihydroxyvitamin D₃ or calcitriol) acts as a hormone, primarily stimulating the intestinal absorption of calcium and phosphate, contributing to bone mineralization and maintaining plasma calcium levels.
F. Thymus
A lymphatic organ located in the chest, primarily active in childhood and crucial for immune system development.
- Thymosin
- Produced by: Thymus.
- Target Cells: T lymphocytes (thymocytes).
- Major Functions:
- Enhances T Lymphocyte Proliferation and Function: Involved in the maturation and differentiation of T lymphocytes, essential for cell-mediated immunity.
G. Heart
The heart, primarily a circulatory organ, also produces hormones involved in fluid and electrolyte balance.
- Atrial and Brain Natriuretic Peptides (ANP, BNP)
- ANP produced by: Atrial muscle cells (primarily).
- BNP produced by: Ventricular muscle cells (primarily).
- Target Cells: Kidney tubules, adrenal cortex, blood vessels.
- Major Functions:
- Inhibit Na⁺ Reabsorption: Promote the excretion of sodium and water by the kidneys (natriuresis and diuresis), thereby reducing blood volume and blood pressure.
- Vasodilation: Cause vasodilation, further lowering blood pressure.
- Inhibit Renin and Aldosterone Secretion: Suppress the RAAS.
H. Adipose Tissue
Far from being just a storage depot, adipose tissue is an active endocrine organ.
- Leptin
- Produced by: Adipocytes (fat cells).
- Target Cells: Hypothalamus (arcuate nucleus).
- Major Functions:
- Suppresses Appetite, Important in Long-Term Control of Body Weight: Signals satiety to the brain, informing it about the body's energy stores. Higher leptin levels (indicating more fat) typically lead to decreased appetite and increased energy expenditure.
- Other Adipokines (e.g., Adiponectin, Resistin, inflammatory cytokines)
- Produced by: Adipocytes.
- Target Cells: Multiple sites (liver, muscle, immune cells).
- Major Functions:
- Play a Role in Metabolism and Inflammation: These hormones influence insulin sensitivity, glucose and lipid metabolism, and immune responses, linking obesity to metabolic disorders and chronic inflammation.
Classification of hormones depending upon chemical nature
| Steroids | Proteins | Derivatives of tyrosine |
|---|---|---|
|
Aldosterone 11-deoxycorticosterone Cortisol Corticosterone Testosterone Dihydrotestosterone Dehydroepiandrosterone Androstenedione Estrogen Progesterone |
Growth hormone (GH) Thyroid-stimulating hormone (TSH) Adrenocorticotropic hormone (ACTH) Follicle-stimulating hormone (FSH) Luteinizing hormone (LH) Prolactin Antidiuretic hormone (ADH) Oxytocin Parathormone Calcitonin Insulin Glucagon Somatostatin Pancreatic polypeptide Human chorionic gonadotropin (HCG) Human chorionic somatomammotropin. |
Thyroxine (T₄) Triiodothyronine (T₃) Adrenaline (Epinephrine) Noradrenaline (Norepinephrine) Dopamine. |
Endocrine System
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Endocrine System
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.
Urinary System
Urinary System
The Urinary System
The urinary system, also known as the renal system, is a vital organ system responsible for filtering waste products from the blood, maintaining fluid and electrolyte balance, regulating blood pressure, and producing hormones.
It is composed of the following main organs:
- Kidneys: The primary organs of filtration and urine formation.
- Ureters: Tubes that transport urine from the kidneys to the urinary bladder.
- Urinary Bladder: A muscular sac that stores urine.
- Urethra: A tube that excretes urine from the bladder to the outside of the body.
Main Functions of the Urinary System
- Maintenance of Water and Electrolyte Balance: Regulates the body's total water volume and the concentration of various electrolytes (e.g., sodium, potassium, calcium, phosphate).
- Regulation of Osmotic Pressure: By controlling water and solute excretion, the kidneys help maintain the appropriate osmolarity of body fluids.
- Excretion of Metabolic Wastes: Removes nitrogenous wastes such as urea (from protein metabolism), creatinine (from muscle metabolism), and uric acid (from nucleic acid metabolism). Also excretes drugs and toxins.
- Regulation of Blood pH: By adjusting the excretion of hydrogen ions (H+) and bicarbonate ions (HCO3-), the kidneys play a crucial role in maintaining acid-base balance.
- Regulation of Blood Pressure: Produces the enzyme renin, which initiates the renin-angiotensin-aldosterone system (RAAS), a key mechanism for regulating blood pressure.
- Production of Erythropoietin: Secretes the hormone erythropoietin (EPO), which stimulates red blood cell production in the bone marrow.
- Activation of Vitamin D: Converts inactive vitamin D to its active form (calcitriol), which is essential for calcium absorption and bone health.
- Gluconeogenesis: During prolonged fasting, the kidneys can synthesize glucose from non-carbohydrate precursors.
Parts of the Urinary System: Upper and Lower Tracts
For anatomical and clinical convenience, the urinary system is often divided into two main parts:
1. Upper Urinary Tract
- Location: Primarily located in the abdominal cavity.
- Components:
- Two Kidneys
- Upper two-thirds of the Ureters
2. Lower Urinary Tract
- Location: Primarily located in the pelvis and perineum.
- Components:
- Lower one-third of the Ureters
- Urinary Bladder
- Urethra
Upper Urinary Tract Details
Kidneys
- Description: Two reddish-brown, bean-shaped organs, each about the size of a fist. They are the primary blood-filtering organs of the urinary system.
- Location: Located on the posterior abdominal wall, one on each side of the vertebral column, retroperitoneally (behind the peritoneum).
- Specific Positioning:
- Typically extend from the level of the T12 vertebra to the L3 vertebra.
- The right kidney is usually positioned slightly lower than the left kidney (approximately half an inch) due to the large size and position of the liver on the right side of the abdominal cavity.
- Mobility: Kidneys are not rigidly fixed. They exhibit some mobility and move vertically with respiration (descending during inspiration and ascending during expiration) and with changes in body position.
- Hilum: The medial concave border of each kidney features a vertical cleft called the renal hilum. This is the entry and exit point for the renal artery and vein, nerves, and the renal pelvis (which narrows to become the ureter). The hilum is typically located around the level of the L1 vertebra.
- Function:
- Urine Formation: Filters blood to produce urine, removing metabolic wastes and excess water/electrolytes.
- Erythropoietin Production: Produces the hormone erythropoietin, essential for red blood cell synthesis.
- Renin Production: Produces renin, vital for blood pressure regulation.
- Vitamin D Activation: Activates vitamin D.
Coverings of the Kidney (from innermost to outermost)
The kidneys are protected and held in place by four layers of tissue:
- Renal Capsule (Fibrous Capsule):
- Description: The innermost layer. A thin, tough, transparent, fibrous connective tissue capsule that directly surrounds the kidney parenchyma.
- Attachment: It is closely and firmly attached to the kidney surface.
- Function: Provides a barrier against infection and trauma.
- Perirenal Fat (Adipose Capsule):
- Description: A layer of adipose tissue (fat) that surrounds the renal capsule.
- Function: Cushions the kidney against physical trauma and helps hold it in position. Its presence provides insulation.
- Renal Fascia (Gerota's Fascia):
- Description: A condensation of connective tissue that encloses both the kidney and the adrenal gland (which sits atop the kidney). It has an anterior and a posterior layer.
- Function: Anchors the kidney to the posterior abdominal wall and surrounding structures, helping to maintain its position. It also separates the kidney from the surrounding pararenal fat.
- Pararenal Fat (Paranephric Fat):
- Description: The outermost layer, consisting of a variable amount of fat located external to the renal fascia. It is part of the broader retroperitoneal fat.
- Function: Provides additional cushioning and support for the kidney.
Internal Structure of Kidneys
When sectioned, the kidney reveals two distinct regions: an outer cortex and an inner medulla.
Renal Cortex
- Description: The outer, lighter reddish-brown region of the kidney. It is granular in appearance.
- Extension into Medulla: The cortex extends inwards, forming structures called renal columns (of Bertin). These columns separate the renal pyramids within the medulla.
- Contents: Contains the renal corpuscles (glomeruli and Bowman's capsules), convoluted tubules (proximal and distal), and cortical collecting ducts. It's the primary site of blood filtration.
Renal Medulla
- Description: The inner, darker reddish-brown region, appearing striated.
- Renal Pyramids: The medulla is organized into 8-18 cone-shaped masses called renal pyramids.
- Base: The broad base of each pyramid faces the renal cortex.
- Apex: The pointed apex of each pyramid, known as the renal papilla, projects medially into a minor calyx.
- Contents: Primarily contains the loops of Henle, collecting ducts, and vasa recta (blood vessels that parallel the loops of Henle). It's crucial for establishing the osmotic gradient necessary for concentrating urine.
Renal Lobes: Each renal pyramid, along with its overlying cap of cortical tissue and half of each adjacent renal column, constitutes a renal lobe. There are typically 8-18 renal lobes per kidney.
Collecting System within the Kidney
- Hilum: As mentioned, the hilum is the indentation on the medial aspect of the kidney. It serves as the point of entry for the renal artery, nerves, and lymphatic vessels, and the point of exit for the renal vein and the ureter. The arrangement of these structures from anterior to posterior at the hilum is typically Renal Vein (V), Renal Artery (A), Ureter (U), and sometimes a second artery (A), often remembered by the mnemonic "V.A.U.A.".
- Renal Sinus: This is the fat-filled space within the hilum, extending into the kidney. It contains the renal pelvis, calyces, renal vessels, nerves, and fat.
- Renal Pelvis: The upper, expanded, funnel-shaped part of the ureter, located within the renal sinus. It collects urine from the major calyces.
- Major Calyces: The renal pelvis typically divides into 2-3 large collecting tubes called major calyces.
- Minor Calyces: Each major calyx, in turn, divides into several smaller collecting cups called minor calyces. Each minor calyx receives urine directly from one or more renal papillae.
- Connection to Collecting Tubules: The renal papillae are the tips of the renal pyramids, where the collecting ducts open, allowing urine to drain into the minor calyces. The collecting tubules (part of the nephron unit) ultimately drain into these collecting ducts.
Relations of the Kidneys
The kidneys are retroperitoneal organs, meaning they lie behind the peritoneum that lines the abdominal cavity. Their relations differ slightly between the right and left sides due to the asymmetry of abdominal organs.
Anteriorly
Right Kidney:
- Right Adrenal Gland: Sits superiorly on its superomedial aspect.
- Liver: The large right lobe of the liver is anterior to the superior pole.
- Second Part of Duodenum: Part of the small intestine, crosses the hilum.
- Right Colic Flexure (Hepatic Flexure): The bend between the ascending and transverse colon, lies anterior to the inferior pole.
- Small Intestine (Ileum): Coils of small intestine may be anterior.
Left Kidney:
- Left Adrenal Gland: Sits superiorly on its superomedial aspect.
- Spleen: Lies superiorly and laterally.
- Stomach: The posterior wall of the stomach can be anterior to the upper part.
- Pancreas: Crosses the middle part of the kidney, typically at the hilum.
- Left Colic Flexure (Splenic Flexure): The bend between the transverse and descending colon, lies anterior to the inferior pole.
- Jejunum: Coils of small intestine may be anterior.
Posteriorly
Both kidneys are related posteriorly to the diaphragm superiorly, and the following muscles of the posterior abdominal wall (from medial to lateral):
- Psoas Major Muscle: Medial to the kidney.
- Quadratus Lumborum Muscle: Lateral to the psoas major.
- Transversus Abdominis Muscle: Lateral to the quadratus lumborum.
Additionally:
- Right Kidney: Diaphragm (separates from pleura/right lung), Subcostal Nerve, Iliohypogastric Nerve, Ilioinguinal Nerve, and the 12th Rib.
- Left Kidney: Diaphragm (separates from pleura/left lung), Subcostal Nerve, Iliohypogastric Nerve, Ilioinguinal Nerve, and both the 11th and 12th Ribs (reflecting its slightly higher position).
Blood Supply of Kidneys
The kidneys receive a rich blood supply, vital for their filtration function.
Arterial Supply:
- Renal Artery: Each kidney receives blood from a large renal artery, which is a direct branch of the abdominal aorta. These branches typically arise at the level of the L1 vertebra.
- Branching Pattern: The renal artery undergoes extensive branching within the kidney:
- Segmental Arteries: Within the renal sinus, the renal artery typically divides into 5 segmental arteries, supplying different vascular segments of the kidney.
- Lobar Arteries: Each segmental artery branches into lobar arteries, one for each renal lobe.
- Interlobar Arteries: Lobar arteries pass between the renal pyramids as interlobar arteries.
- Arcuate Arteries: At the junction of the cortex and medulla (base of the pyramids), interlobar arteries arch over the bases of the pyramids to become arcuate arteries.
- Cortical Radiate Arteries (Interlobular Arteries): Arcuate arteries give off numerous cortical radiate arteries that extend into the cortex.
- Afferent Glomerular Arterioles: These arise from the cortical radiate arteries and supply blood to the glomeruli, where filtration begins.
- Efferent Glomerular Arterioles: Blood leaves the glomerulus via efferent arterioles, which then form the peritubular capillaries (around convoluted tubules) and vasa recta (around loops of Henle in the medulla).
Venous Drainage:
- Renal Vein: Blood drains from the kidney via veins that generally follow the arterial pathway in reverse (e.g., cortical radiate veins, arcuate veins, interlobar veins). These converge to form the renal vein.
- Superior Vena Cava Connection: The right and left renal veins drain directly into the inferior vena cava (IVC). The left renal vein is typically longer than the right, as it crosses the aorta anteriorly to reach the IVC.
Lymphatic Drainage:
- The lymphatic vessels from the kidneys generally follow the renal arteries and veins.
- They drain into the paraaortic lymph nodes (or lumbar lymph nodes), which are located along the abdominal aorta.
Ureters
- Description: The ureters are two slender, muscular tubes that transport urine from the renal pelvis of each kidney to the urinary bladder.
- Length: Each ureter is approximately 25-30 cm (10-12 inches) long in adults.
- Diameter: The diameter is about 3 mm, but it can vary and has several physiological narrowings.
- Propulsion of Urine: Urine is not transported by gravity alone. The muscular walls of the ureters undergo rhythmic, wave-like contractions called peristalsis, which actively propel urine downwards, regardless of body position. This peristaltic action typically occurs 1 to 5 times per minute.
Narrowings of the Ureters (Sites of Potential Obstruction)
These three anatomical narrowings are clinically important as they are common sites for the lodgement of urinary calculi (kidney stones), which can cause significant pain and obstruction.
- Ureteropelvic Junction (UPJ): At the beginning of the ureter, where the renal pelvis narrows to become the ureter.
- Pelvic Brim (Iliac Vessels Crossing): Where the ureter crosses the brim of the lesser pelvis, anterior to the bifurcation of the common iliac artery (or just distal to it, where it crosses the external iliac artery).
- Ureterovesical Junction (UVJ): At the end of the ureter, where it passes obliquely through the muscular wall of the urinary bladder. This oblique entry acts as a physiological valve, preventing reflux of urine from the bladder back into the ureter.
Relations of the Ureter
The relations of the ureters are extensive due to their long course through the retroperitoneal space.
- POSTERIORLY:
- Throughout its course in the abdomen, the ureter descends anterior to the psoas major muscle. This muscle separates the ureter from the lumbar transverse processes.
- It crosses anterior to the common iliac vessels or their bifurcations at the pelvic brim.
- In the pelvis, it runs anterior to the internal iliac artery branches.
- ANTERIORLY:
- Right Ureter: Crossed anteriorly by the duodenum (descending part), right colic vessels, ileocolic vessels, right testicular/ovarian vessels, and may be crossed by coils of ileum.
- Left Ureter: Crossed by the sigmoid colon and its mesocolon, left colic vessels, left testicular/ovarian vessels, and may be crossed by coils of jejunum.
- In Females (Pelvic Part): The ureter passes inferior to the uterine artery ("water under the bridge"). This relationship is critically important during hysterectomy (surgical removal of the uterus) to avoid accidental ligation or injury to the ureter.
Blood Supply of the Ureters
The ureters have an extensive and variable arterial supply from several sources along their length, forming an anastomotic network.
- Upper Part: Supplied by branches from the renal artery.
- Middle Part: Supplied by branches from the gonadal arteries (testicular artery in males, ovarian artery in females), and also sometimes from the aorta, common iliac, or internal iliac arteries.
- Lower Part: Supplied by branches from the superior vesical artery (a branch of the internal iliac artery), middle rectal artery, and uterine artery (in females).
- Narrowings and Stone Lodgement: As mentioned, the three physiological narrowings (UPJ, pelvic brim, UVJ) are prime locations for kidney stones (ureterolithiasis) to become lodged, leading to obstruction and severe pain.
- Cancer of Ureters: Though less common than kidney or bladder cancer, transitional cell carcinoma can develop in the ureters. Obstruction from a tumor can lead to similar symptoms as a stone.
- Referred Pain (Renal Colic): Obstruction or irritation of the ureter (most commonly by a stone) causes intense, spasmodic pain known as renal colic.
- Nature of Pain: It is typically a severe, wave-like, excruciating pain that radiates from the flank (costovertebral angle) anteriorly and inferiorly towards the groin, labia (in females), or scrotum (in males).
- Mechanism of Referred Pain: The sensory innervation of the ureter is primarily from T11-L2 spinal cord segments. Pain signals are perceived by the brain as originating from the dermatomes supplied by these same spinal cord segments, which include the flank, lower abdomen, and groin region. The colicky nature is due to the ureter's attempt to contract and expel the obstruction.
- Associated Symptoms: Nausea, vomiting, hematuria (blood in urine), and dysuria (painful urination) may accompany renal colic.
Lower Urinary Tract
The lower urinary tract is responsible for the storage and elimination of urine. It is composed of the lower one-third of the ureters, the urinary bladder, and the urethra.
Urinary Bladder
- Description: A muscular, distensible reservoir located in the pelvic cavity that temporarily stores urine until it is excreted from the body. Its shape and position vary depending on the amount of urine it contains.
- Shape: When empty, it is roughly pyramidal in shape. As it fills, it becomes more ovoid or spherical.
- Main Function: Storage of urine.
- Average Capacity: The average functional capacity is around 300-500 ml (0.5 liters), though it can distend to hold up to 1 liter or more before reflex emptying occurs or discomfort becomes significant.
- Urge to Urinate: The first urge to urinate typically occurs when the bladder contains about 150-200 ml of urine.
Position and Relations (Varies with Filling)
- Empty Bladder:
- It is a pelvic organ, lying mostly within the lesser pelvis.
- It is located directly posterior to the symphysis pubis.
- In males, it is superior to the prostate gland.
- In females, it is anterior to the vagina and uterus.
- Filled Bladder:
- As the bladder fills, it expands superiorly and anteriorly, lifting out of the pelvis and becoming an abdominal organ. This expansion occurs between the peritoneum and the anterior abdominal wall.
- Clinical Significance: This superior expansion into the abdominal cavity, especially when distended, makes it more susceptible to injury from blunt abdominal trauma. It is cited as the second most commonly injured organ in blunt abdominal trauma (after the spleen).
Anatomical Parts and Surfaces
The pyramidal shape of an empty bladder helps describe its surfaces and relations. It has:
- Apex: The anterior-superior part, pointing towards the symphysis pubis. It is connected to the umbilicus by the median umbilical ligament (a remnant of the urachus).
- Neck: The most inferior and fixed part of the bladder, funnel-shaped, continuous with the urethra. It is supported by ligaments and, in males, is surrounded by the prostate gland.
- Posterior Surface (Base or Fundus): Faces posteriorly and slightly inferiorly.
- In Males: Related to the rectum, seminal vesicles, and vasa deferentia.
- In Females: Related to the vagina (anterior wall) and the uterus (cervix and lower body).
- Superior Surface: The uppermost surface.
- In Males: Covered by peritoneum and related to coils of small intestine and the sigmoid colon.
- In Females: Covered by peritoneum and related to the body of the uterus.
- Two Inferolateral Surfaces: These are the largest surfaces when the bladder is empty, facing anterolaterally. They are separated from the anterior abdominal wall by the retropubic space (of Retzius), which contains loose areolar tissue and fat (extraperitoneal fat).
Internal Structure
- Detrusor Muscle: The muscular wall of the bladder is composed of three layers of smooth muscle collectively known as the detrusor muscle. This muscle contracts during micturition (urination) to expel urine.
- Trigone: A smooth, triangular area on the internal posterior floor of the bladder. It is bounded by the openings of the two ureters (superiorly) and the internal urethral orifice (inferiorly). The mucosa of the trigone is smooth and firmly attached, unlike the rest of the bladder which forms folds (rugae) when empty. The trigone is sensitive to stretch, signaling the urge to urinate.
- Internal Urethral Sphincter: Involuntary smooth muscle fibers around the neck of the bladder, particularly well-developed in males, preventing semen reflux during ejaculation. Its role in urinary continence is debated in females.
Blood Supply of the Urinary Bladder
- Arterial Supply: Primarily supplied by branches of the internal iliac arteries:
- Superior Vesical Arteries: Usually arise from the umbilical artery (a remnant of the fetal umbilical artery).
- Inferior Vesical Arteries: In males, these typically supply the fundus of the bladder, prostate, and seminal vesicles.
- Vaginal Arteries: In females, these branches from the internal iliac artery supply the inferior part of the bladder and the vagina.
- (Additional supply from obturator and gluteal arteries may occur).
- Venous Drainage: Venous plexuses (vesical venous plexus) drain into the internal iliac veins.
Nerve Supply of the Urinary Bladder (Micturition Reflex)
The innervation of the bladder is complex, involving both sympathetic and parasympathetic divisions of the autonomic nervous system, as well as somatic innervation for the external urethral sphincter.
- Sympathetic Innervation (Hypogastric Nerves, T11-L2):
- Effect on Detrusor Muscle: Causes relaxation of the detrusor muscle, allowing the bladder to fill and store urine.
- Effect on Internal Urethral Sphincter (in males): Causes contraction of the internal urethral sphincter, preventing urine leakage.
- Overall Role: Promotes urine storage.
- Parasympathetic Innervation (Pelvic Splanchnic Nerves, S2-S4):
- Effect on Detrusor Muscle: Causes contraction of the detrusor muscle, leading to expulsion of urine.
- Effect on Internal Urethral Sphincter (in males): Causes relaxation of the internal urethral sphincter.
- Overall Role: Mediates the emptying (micturition) reflex.
- Somatic Innervation (Pudendal Nerve, S2-S4):
- Innervates the external urethral sphincter, which is composed of skeletal muscle and is under voluntary control. This allows for conscious control over urination.
The Urethra
The urethra is a tube that carries urine from the urinary bladder to the outside of the body. There are significant anatomical differences between the male and female urethra.
Male Urethra
- Length: Considerably longer than the female urethra, approximately 20 cm (8 inches) long.
- Function: Serves a dual role: it is a passageway for both urine (during micturition) and semen (during ejaculation).
- Course and Parts: The male urethra is divided into four main parts:
- Preprostatic Urethra: Shortest part (0.5-1.5 cm), extending from the internal urethral orifice to the prostate gland. Surrounded by the internal urethral sphincter.
- Prostatic Urethra: Passes through the prostate gland (3-4 cm). Receives the ejaculatory ducts and prostatic ducts.
- Membranous Urethra: The shortest and narrowest part (1-1.5 cm), extending from the apex of the prostate to the bulb of the penis. It passes through the urogenital diaphragm and is surrounded by the external urethral sphincter (voluntary skeletal muscle). This is the least protected part and most vulnerable to injury.
- Spongy (Penile) Urethra: The longest part (about 15 cm), passing through the corpus spongiosum of the penis. It ends at the external urethral orifice (meatus) at the tip of the glans penis. It receives the ducts of the bulbourethral glands.
- External Urethral Orifice: Opens at the end of the penis.
Female Urethra
- Length: Much shorter than the male urethra, approximately 3-4 cm (1.5 inches) long.
- Function: Serves only as a passageway for urine.
- Course: Extends from the internal urethral orifice of the bladder, passing through the deep perineal pouch, and opening at the external urethral orifice (meatus) into the vestibule, anterior to the vaginal opening.
- External Urethral Sphincter: Surrounds the middle part of the urethra, providing voluntary control over urination.
- Clinical Significance of Short Length: The short length and close proximity of the female urethra to the anus make females more susceptible to urinary tract infections (UTIs), as bacteria can more easily ascend into the bladder.
Urinary System Quiz
Systems Anatomy
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Urinary System Quiz
Systems Anatomy
Preparing questions...
Exam Completed!
See your performance breakdown below.
Genitourinary System: Female Reproductive System
Genitourinary System Female Reproductive System
Female External Genitalia (Vulva)
The female external genitalia, collectively known as the vulva (or pudendum), encompass all the visible structures that extend from the pubic region to the perineum. These structures protect the internal reproductive organs, play a role in sexual arousal and function, and contain the openings of the urethra and vagina.
The vulva is composed of the following main structures:
- Mons Pubis (Mons Veneris)
- Labia Majora
- Labia Minora
- Clitoris
- Vestibule
- Urethral Orifice
- Vaginal Orifice
- Greater Vestibular Glands (Bartholin's Glands) (and Lesser Vestibular Glands)

1. Mons Pubis (Mons Veneris)
- Description: A rounded, fleshy prominence composed of adipose tissue (fat pad) overlying the pubic symphysis (the joint between the two pubic bones).
- Location: Anterior to the labia majora and superior to the clitoris.
- Covering: After puberty, it becomes covered with pubic hair.
- Hair Pattern:
- Female Pattern: Typically forms an inverted triangle, with a horizontal upper border (concave or convex downwards).
- Male Pattern: Tends to be diamond-shaped, extending upwards towards the umbilicus.
- Clinical Relevance: A change in female hair pattern towards a masculine distribution (hirsutism) can indicate underlying hormonal imbalances (e.g., polycystic ovary syndrome, adrenal disorders) and may be associated with infertility or other endocrine issues.
- Function: Acts as a protective cushion over the pubic bone during sexual intercourse.
2. Labia Majora
- Description: Two prominent, longitudinal, fleshy folds of skin that extend downwards and backwards from the mons pubis. They are homologous to the scrotum in males.
- Composition: Primarily composed of fat, elastic tissue, and some smooth muscle. Their outer surface is covered with pubic hair after puberty, while the inner surface is hairless and smooth.
- Extension: They unite anteriorly at the mons pubis and posteriorly, they tend to merge into the perineum, often forming a posterior commissure anterior to the anal canal.
- Erectile Tissue: The tissue within the labia majora is non-erectile.
- Function: Act as a protective barrier, enclosing and safeguarding the more delicate internal structures of the vulva (labia minora, clitoris, and vestibule).
3. Labia Minora (Nymphae)
- Description: Two smaller, hairless folds of highly vascularized, reddish-pink skin located medial to the labia majora. They are homologous to the ventral aspect of the penis in males.
- Composition: Rich in erectile tissue, blood vessels, nerve endings, and sebaceous glands, giving them a rich sensory innervation.
- Anatomical Configuration:
- Anteriorly: Each labium minus splits into two folds:
- Medial Fold: Passes over the clitoris to form the frenulum of the clitoris on its undersurface.
- Lateral Fold: Passes over the clitoris to form the prepuce (hood) of the clitoris on its dorsal surface.
- Posteriorly: In nulliparous (never given birth) women, the labia minora may fuse to form a transverse fold called the fourchette anterior to the vaginal opening. This often disappears after childbirth.
- Anteriorly: Each labium minus splits into two folds:
- Erectile Tissue: The labia minora contain erectile tissue, and they engorge with blood during sexual arousal.
- Function:
- Sexual Arousal: Highly sensitive due to rich nerve supply, contributing significantly to sexual pleasure. They swell and darken during arousal.
- Protection: Further protect the urethral and vaginal orifices.
- Lubrication: Contain numerous sebaceous glands for lubrication.
- Surgical Use: The tissue's elasticity and rich blood supply make it suitable for grafting, particularly in reconstructive surgery (e.g., for facial wounds, though this is a specialized application and not its primary function).
4. Clitoris
- Description: A highly sensitive organ composed of erectile tissue, homologous to the penis in males. It is the primary organ for sexual pleasure in females.
- Location: Located anteriorly, where the labia minora converge.
- Structure:
- Fixed Root: Attached to the pubic arch. Composed of three masses of erectile tissue:
- Crura (Corpora Cavernosa): Two crura, continuous with the corpora cavernosa of the body of the clitoris, attach to the pubic rami and are covered by the ischiocavernosus muscles.
- Bulbs of the Vestibule (Corpus Spongiosum): Two masses of erectile tissue, located on either side of the vaginal orifice (split in females due to the presence of the vagina), covered by the bulbospongiosus muscles. These merge anteriorly to form the body of the clitoris.
- Free Body: Protrudes externally, composed of two corpora cavernosa and a small amount of corpus spongiosum (which forms the glans).
- Glans Clitoris: The most external, highly sensitive, pea-sized tip of the clitoris.
- Coverings: Covered by the prepuce (hood) and frenulum, formed by the labia minora.
- Fixed Root: Attached to the pubic arch. Composed of three masses of erectile tissue:
- Erection Mechanism (Similar to penis):
- Psychic and Tactile Stimulation: Pleasurable sights, sounds, smells, and touch (nipples, inner thighs, clitoris itself) trigger a parasympathetic discharge from the sacral spinal cord segments (S2, S3, S4).
- Vasodilation: This leads to the release of nitric oxide, causing vasodilation of arteries supplying the erectile tissue (corpora cavernosa and bulbs of vestibule).
- Engorgement: Increased blood flow rapidly fills the vascular spaces within the erectile tissue.
- Venous Occlusion: The engorged erectile tissue compresses the veins draining the clitoris against the surrounding tough fibrous capsule (tunica albuginea) and pelvic bones, retarding venous outflow.
- Erection: The combination of increased arterial inflow and decreased venous outflow leads to engorgement and rigidity of the clitoris.
- Function: Solely dedicated to sexual sensation and arousal.
5. Vestibule
- Description: The almond-shaped space or cleft enclosed by the labia minora.
- Boundaries:
- Laterally: Labia minora.
- Anteriorly: Clitoris.
- Posteriorly: Fourchette (if present) or posterior commissure.
- Contents/Openings: It contains the openings of several important structures:
- Urethral Orifice
- Vaginal Orifice
- Ducts of the Greater Vestibular Glands (Bartholin's Glands)
- Ducts of the Lesser Vestibular Glands (Skene's Glands/Paraurethral Glands): These small mucus-secreting glands are located around the urethral orifice.
6. Urethral Orifice (External Urethral Meatus)
- Description: The opening of the female urethra.
- Location: Located posterior to the clitoris and anterior to the vaginal orifice, typically about 2.5 cm below the clitoris. It often appears as a small, slit-like or star-shaped opening.
- Function: Serves as the sole passage for urine in females.
- Clinical Relevance:
- Infections: Its short length (about 4 cm, much shorter than in males) and close proximity to the anal canal and vaginal opening make females more susceptible to recurrent urinary tract infections (UTIs), as bacteria can easily ascend into the bladder.
- Trauma: Can be traumatized during sexual intercourse or childbirth.
- Obstruction: Infections like Herpes Simplex Virus (HSV) can cause severe pain and swelling, potentially leading to dysuria or acute urinary retention if the orifice is obstructed.
7. Vaginal Orifice (Introitus)
- Description: The external opening of the vagina, located posterior to the urethral orifice and anterior to the anus.
- Covering: In virgins, it is typically partially covered by the hymen.
- Hymen:
- Description: A thin, vascularized mucous membrane that partially occludes the vaginal opening. Its shape and thickness vary widely among individuals.
- Breakage: It often ruptures or stretches during first sexual intercourse or due to other activities (e.g., tampon insertion, vigorous exercise). Remnants after rupture are called hymenal tags.
- Clinical Significance: The absence of an intact hymen is NOT a reliable indicator of non-virginity, as it can be broken by non-coital activities or may be naturally absent/very thin.
- Imperforate Hymen: A congenital condition where the hymen completely blocks the vaginal opening.
- Clinical Relevance: Can lead to menstrual blood retention (hematocolpos) at menarche, causing severe lower abdominal pain, absence of menstruation (primary amenorrhea), and a bulging bluish mass visible at the introitus. Requires surgical incision (hymenotomy) to allow drainage.
8. Greater Vestibular Glands (Bartholin's Glands)
- Description: Two small, pea-sized glands, homologous to the bulbourethral glands in males.
- Location: Located on each side of the vaginal orifice, embedded within the posterior aspect of the labia majora.
- Ducts: Their ducts open into the vestibule, specifically between the labia minora and the hymen (or its remnants), usually at the 4 o'clock and 8 o'clock positions relative to the vaginal opening.
- Function: Produce a clear, mucus-like secretion during sexual arousal to lubricate the vestibule and vaginal opening, facilitating intercourse.
- Clinical Relevance: Can become blocked, leading to a Bartholin's cyst (painless swelling) or, if infected, a Bartholin's abscess (painful, inflamed, pus-filled swelling).
Lesser Vestibular Glands (Skene's Glands / Paraurethral Glands)
- Description: Numerous small mucous glands that open into the vestibule, mainly around the urethral orifice. Homologous to the prostate gland in males.
- Function: Produce mucus that lubricates the urethral opening and vestibule. Some researchers believe they contribute to "female ejaculation."
- Clinical Relevance: Can become infected, leading to Skene's gland cysts or abscesses.
Blood Supply, Venous Drainage, and Lymphatics of the Vulva
- Arterial Supply: Primarily from branches of the internal pudendal artery and external pudendal artery.
- Clitoris: Specific branches include the deep arteries of the clitoris, artery of the bulb of the vestibule, and dorsal artery of the clitoris (all branches of the internal pudendal artery).
- Rest of the Vulva: Supplied by labial branches of both internal and external pudendal arteries.
- Venous Drainage: Venous plexuses drain into the internal pudendal veins and external pudendal veins, which then drain into the internal iliac veins and femoral veins, respectively.
- Lymphatic Drainage:
- Lymph from the skin and superficial structures of the vulva (mons pubis, labia majora, labia minora, clitoris, vestibule) drains predominantly to the superficial inguinal lymph nodes (specifically the medial group).
- Lymph from the deeper parts of the clitoris may also drain to the deep inguinal and even internal iliac lymph nodes.
- Innervation:
- Pudendal Nerve (S2-S4): Provides most of the somatic (sensory and motor) innervation to the vulva. Its branches include the dorsal nerve of the clitoris, labial nerves, and perineal nerves.
- Ilioinguinal Nerve (L1): Provides sensory innervation to the anterior labia majora and mons pubis.
- Genital Branch of Genitofemoral Nerve (L1-L2): Provides sensory innervation to the anterior labia majora.
Common Clinical Conditions of the Vulva
The vulva, being external and exposed, is susceptible to a variety of conditions, including infections, inflammatory disorders, structural anomalies, and neoplastic changes. These conditions can cause discomfort, pain, itching, and impact sexual health and quality of life.
1. Pubic Lice (Pthirus Pubis, "Crabs")
- Description: An infestation of the pubic hair and surrounding coarse body hair (e.g., perineum, thighs, eyelashes, axilla) by the parasitic insect Pthirus pubis.
- Transmission: Primarily through close physical contact, most commonly sexual contact. Can also be spread through infested clothing, bedding, or towels.
- Clinical Features:
- Intense pruritus (itching): Especially at night, caused by an allergic reaction to the louse's saliva.
- Maculae caeruleae ("blue spots"): Small, bluish-gray spots on the skin where lice have fed, caused by anticoagulant in the louse's saliva.
- Visible nits (eggs): Small, oval, whitish-yellow eggs firmly attached to the hair shafts, close to the skin.
- Adult lice: Tiny (2-3 mm), crab-shaped insects that are difficult to see with the naked eye but may be spotted clinging to hairs.
- Excoriations: Skin damage from scratching, which can lead to secondary bacterial infections.
- Diagnosis: Visual identification of nits, nymphs, or adult lice.
- Treatment:
- Topical Insecticides: Permethrin (1% cream rinse), pyrethrins with piperonyl butoxide. Apply to affected areas, leave for recommended time, then wash off. Repeat in 7-10 days to kill newly hatched lice.
- Hygiene: Wash all clothing, bedding, and towels in hot water and dry on high heat, or dry clean. Items that cannot be washed should be sealed in plastic bags for two weeks.
- Treatment of Sexual Partners: All recent sexual contacts should be treated simultaneously.
- Combing: Fine-toothed combs can help remove nits.
2. Bartholin's Cyst
- Description: A fluid-filled sac that forms when the duct of a Bartholin's gland becomes obstructed.
- Etiology: Obstruction of the duct can be due to inflammation, infection, trauma, or congenital narrowing. When the duct is blocked, the mucus produced by the gland accumulates, forming a cyst.
- Clinical Features:
- Painless swelling: Typically located unilaterally at the posterior aspect of the labia majora, near the vaginal opening (4 or 8 o'clock position).
- Size: Can range from small to several centimeters, potentially causing discomfort during walking, sitting, or intercourse.
- Discomfort: Larger cysts can cause pressure or a sensation of fullness. The statement "swelling of labia majora during sexual excitement" might refer to the cyst becoming more noticeable or slightly larger due to increased blood flow, but the cyst itself is not erectile tissue.
- Diagnosis: Clinical examination.
- Treatment:
- Asymptomatic cysts: Often require no treatment.
- Symptomatic cysts:
- Sitz baths: Warm compresses can sometimes encourage drainage.
- Word Catheter Insertion: A small catheter is inserted into the cyst, inflated, and left in place for several weeks to create a new drainage pathway (marsupialization).
- Marsupialization: A surgical procedure where the cyst is incised, drained, and the edges of the cyst wall are sutured open to the skin, creating a permanent opening.
- Excision of the gland: Rarely performed due to potential for scarring and disfigurement, and risk of recurrence if not completely removed. Reserved for recurrent cases or suspicion of malignancy.
3. Bartholin's Abscess
- Description: An infection of a Bartholin's gland or cyst, leading to the formation of a pus-filled collection.
- Etiology: Most commonly results from bacterial infection of an existing Bartholin's cyst. Common causative organisms include E. coli, staphylococci, streptococci, and sexually transmitted bacteria (e.g., Neisseria gonorrhoeae, Chlamydia trachomatis).
- Clinical Features:
- Severe, throbbing pain: Unilateral, often making walking, sitting, and intercourse excruciatingly painful.
- Erythema (redness), swelling, and tenderness: Of the affected labium majora.
- Hotness (increased local temperature).
- Fever and chills: May be present if the infection is significant.
- Fluctuant mass: A pus-filled lesion that may spontaneously rupture, providing temporary relief.
- Diagnosis: Clinical examination.
- Treatment:
- Incision and Drainage (I&D): The primary treatment. A small incision is made to drain the pus.
- Word Catheter: Can be inserted after drainage to keep the site open and promote healing.
- Antibiotics: Often prescribed, especially if there is significant cellulitis, systemic symptoms (fever), or risk factors for STIs.
- Sitz baths: Can aid in comfort and healing post-drainage.
4. Herpes Simplex (Genital Herpes)
- Description: A sexually transmitted infection (STI) caused by the Herpes Simplex Virus (HSV), typically HSV-2, but HSV-1 (oral herpes) can also cause genital lesions. It is characterized by recurrent outbreaks of painful sores.
- Transmission: Direct skin-to-skin contact, typically during sexual activity, even when lesions are not visible (asymptomatic shedding).
- Clinical Features:
- Primary Infection: Often the most severe. Symptoms include:
- Multiple, painful vesicles (blisters): Small, fluid-filled, which quickly rupture to form shallow, exquisitely painful ulcers on the vulva, perineum, vagina, and cervix.
- Flu-like symptoms: Fever, headache, malaise, muscle aches.
- Lymphadenopathy: Tender, swollen inguinal lymph nodes.
- Dysuria: Painful urination, sometimes leading to urinary retention due to severe pain upon voiding.
- Recurrent Episodes: Less severe and shorter in duration than primary outbreaks. Often preceded by prodromal symptoms like tingling, itching, or burning at the site of future lesions. Lesions tend to be fewer and localized.
- Factors Triggering Recurrence: Stress, illness, fever, sunlight, menstruation, trauma, sexual intercourse. The statement "oral, vaginal and haematogenous" for transmission is partly misleading; haematogenous spread is rare in typical genital herpes. Oral transmission usually refers to oral-genital contact.
- Primary Infection: Often the most severe. Symptoms include:
- Diagnosis: Viral culture, PCR, or antigen detection from fluid in vesicles or ulcers. Serology for HSV antibodies can determine past exposure.
- Treatment:
- Antiviral Medications: Acyclovir, valacyclovir, famciclovir. These do not cure herpes but can reduce the severity and frequency of outbreaks, promote healing, and suppress viral shedding.
- Episodic Therapy: Taken at the onset of an outbreak or prodromal symptoms.
- Suppressive Therapy: Taken daily to prevent outbreaks, especially for frequent or severe recurrences.
- Symptomatic Relief: Pain relievers (NSAIDs), topical anesthetics (e.g., lidocaine cream), sitz baths, loose clothing.
- Antiviral Medications: Acyclovir, valacyclovir, famciclovir. These do not cure herpes but can reduce the severity and frequency of outbreaks, promote healing, and suppress viral shedding.
5. Genital Warts (Condylomata Acuminata)
- Description: Benign epithelial growths on the vulva, perineum, vagina, cervix, or anus, caused by infection with the Human Papillomavirus (HPV), particularly low-risk types (e.g., HPV 6 and 11).
- Transmission: Primarily through sexual contact.
- Clinical Features:
- Warty growths: Can be flesh-colored, whitish, or reddish-brown. They can be small or large, solitary or clustered, and may have a cauliflower-like appearance.
- Location: Commonly found on the labia, perineum, perianal area, vagina, and cervix.
- Symptoms: Often asymptomatic, but can cause itching, burning, discomfort, bleeding, or pain during intercourse.
- Diagnosis: Clinical appearance. Biopsy may be performed for atypical lesions or if there's uncertainty.
- Treatment: Aims to remove visible warts and alleviate symptoms, but does not eradicate the underlying HPV infection.
- Patient-applied treatments: Imiquimod cream (immune response modifier), podofilox solution/gel (cytotoxic agent).
- Clinician-applied treatments:
- Trichloroacetic Acid (TCA): Chemical cautery.
- Cryotherapy: Freezing with liquid nitrogen.
- Surgical Excision: Cutting out the warts.
- Electrocautery: Burning off the warts.
- Laser Therapy: Vaporizing the warts.
- Prevention: HPV vaccination (Gardasil 9) is highly effective in preventing infection with the most common high-risk HPV types (associated with cancer) and low-risk types (associated with genital warts).
6. Herpes Zoster (Shingles)
- Description: A painful viral rash caused by the reactivation of the Varicella-Zoster Virus (VZV), the same virus that causes chickenpox.
- Etiology: After a primary chickenpox infection, VZV remains latent in the dorsal root ganglia. Reactivation occurs when the immune system is weakened (e.g., by age, stress, illness, immunosuppression).
- Clinical Features:
- Prodromal Phase: Often involves localized pain, tingling, itching, or numbness in the affected dermatome for several days before the rash appears.
- Painful rash: Characterized by a band-like (dermatomal) distribution of vesicles on an erythematous base. The lesions do not cross the midline, as they are confined to the sensory distribution of a single or contiguous nerve.
- Location: While it can affect any dermatome, if it affects the sacral dermatomes (S2-S4), it can appear on the vulva or perineum, causing intense pain.
- Resolution: Vesicles progress to pustules, crust over, and heal within 2-4 weeks.
- Complications: Post-herpetic neuralgia (PHN): Persistent pain in the affected dermatome even after the rash has healed, especially common in older individuals.
- Diagnosis: Clinical appearance. Viral culture or PCR can confirm.
- Treatment:
- Antiviral Medications: Acyclovir, valacyclovir, famciclovir. These are most effective if started within 72 hours of rash onset, reducing pain, shortening the duration of the rash, and decreasing the risk of PHN.
- Pain Management: NSAIDs, neuropathic pain medications (gabapentin, pregabalin) for PHN.
- Vaccination: The shingles vaccine (Shingrix) is highly effective in preventing herpes zoster and PHN in adults over 50.
7. Imperforate Hymen
- Description: A congenital anomaly where the hymen completely obstructs the vaginal opening, without any natural perforations.
- Etiology: Failure of the central portion of the hymen to canalize during fetal development.
- Clinical Features (typically present at puberty):
- Primary amenorrhea: Absence of menstruation by age 15-16, despite normal secondary sexual characteristics.
- Cyclic abdominal or pelvic pain: Due to the accumulation of menstrual blood within the vagina (hematocolpos), uterus (hematometra), and potentially fallopian tubes (hematosalpinx).
- Bulging bluish mass: Visible at the introitus, especially if hematocolpos is significant.
- Urinary symptoms: Pressure on the bladder can lead to urinary frequency or retention.
- Back pain.
- Diagnosis: Clinical examination. Ultrasound can confirm the presence of accumulated blood.
- Treatment: Surgical incision (hymenotomy or hymenectomy): A crucial procedure to create an opening and allow drainage of accumulated menstrual blood. This typically provides immediate relief of symptoms.
8. Disorders of Sexual Development (DSD) / Ambiguous Genitalia (formerly Hermaphroditism)
- Description: As mentioned in the male external genitalia section, these are congenital conditions where there is a discrepancy between the external genitalia and the internal reproductive organs or chromosomal sex. The term "hermaphroditism" is outdated and replaced by DSD.
- Clinical Features in the Vulva: This can manifest as ambiguous genitalia at birth, where the external genitalia are not clearly male or female. For example, in a female (XX karyotype) with congenital adrenal hyperplasia, there might be significant virilization leading to clitoromegaly (enlarged clitoris resembling a small penis) and fusion of the labia, making the gender assignment difficult.
- Diagnosis and Management: Requires a multidisciplinary team involving geneticists, endocrinologists, neonatologists, and surgeons to determine chromosomal sex, gonadal sex, and internal anatomy, followed by appropriate medical, surgical, and psychological management, with careful consideration of gender identity.
Female Internal Genitalia
The female internal genitalia are located within the pelvic cavity and are essential for reproduction. They include the organs responsible for producing ova (eggs), facilitating fertilization, nurturing a developing fetus, and enabling childbirth.
The internal genitalia are composed of:
- Ovaries (Gonads)
- Uterine Tubes (Fallopian Tubes or Oviducts)
- Uterus
- Vagina
1. Ovaries (Female Gonads)
- Description: Paired, almond-shaped organs, typically whitish or grayish in color, located on either side of the uterus, within the pelvic cavity. They are homologous to the testes in males.
- Size: In young, reproductive-aged women, each ovary measures approximately 3-5 cm (1.5-2 inches) in length, 2-3 cm (1 inch) in width, and 1-1.5 cm (1/3 inch) in thickness. After menopause, due to hormonal changes, they tend to shrink and become less active.
- Function: The ovaries have two primary functions:
- Oogenesis: Production and release of female gametes (ova or eggs). Females are born with a finite, lifelong supply of primordial follicles, each containing an immature oocyte. This process begins before birth and continues until menopause.
- Hormone Production: Secrete crucial female sex hormones, primarily:
- Estrogen: Responsible for the development of female secondary sexual characteristics, growth of the uterine lining, and regulation of the menstrual cycle.
- Progesterone: Primarily involved in preparing the uterus for pregnancy, maintaining pregnancy, and regulating the menstrual cycle.
- (Also produce small amounts of androgens).
- Location and Support: Suspended in the pelvic cavity by several ligaments:
- Suspensory Ligament of the Ovary: Connects the ovary to the lateral pelvic wall, containing the ovarian artery and vein.
- Ovarian Ligament: Connects the ovary to the lateral aspect of the uterus.
- Mesovarium: A fold of peritoneum that attaches the ovary to the posterior surface of the broad ligament.
- Microscopic Structure: Consists of an outer cortex (containing ovarian follicles at various stages of development) and an inner medulla (containing blood vessels, nerves, and lymphatic vessels).
2. Uterine Tubes (Fallopian Tubes or Oviducts)
- Description: Paired, muscular tubes that extend from the superior lateral aspects of the uterus towards the ovaries, but do not directly attach to them. They provide a passageway for the ovum from the ovary to the uterus and are the typical site of fertilization.
- Length and Width: Measure approximately 10-13 cm (4-5 inches) in length. Their width varies, being widest at the ovarian end (infundibulum, about 1 inch) and narrowest where they enter the uterine wall (isthmus, diameter of a thin spaghetti strand).
- Segments (from lateral to medial):
- Infundibulum: The funnel-shaped, most lateral part, opening into the peritoneal cavity. It is fringed with finger-like projections called fimbriae.
- Fimbriae: Ciliated projections that sweep over the surface of the ovary. During ovulation, they become more active and swell, helping to "catch" the released ovum.
- Ampulla: The longest and widest part of the tube, where fertilization typically occurs.
- Isthmus: A narrower, thick-walled segment connecting the ampulla to the uterus.
- Interstitial (or Intramural) Part: The segment that passes through the muscular wall of the uterus.
- Infundibulum: The funnel-shaped, most lateral part, opening into the peritoneal cavity. It is fringed with finger-like projections called fimbriae.
- Wall Structure: Composed of three layers:
- Mucosa: Lined with ciliated columnar epithelial cells and secretory (peg) cells. The cilia beat in waves towards the uterus, moving the ovum, while peg cells provide nourishment for the ovum and sperm.
- Muscularis: Smooth muscle layers that contract rhythmically (peristalsis) to help propel the ovum towards the uterus.
- Serosa: Outer peritoneal covering.
- Function:
- Ovum Transport: Fimbriae capture the ovum after ovulation, and the coordinated action of cilia and muscular peristalsis transports it towards the uterus.
- Site of Fertilization: The ampulla is the most common site where sperm meets and fertilizes the ovum.
- Sperm Capacitation: The tubal environment contributes to the capacitation of sperm, enabling them to fertilize the ovum.
- Clinical Relevance: Blockage of the fallopian tubes (e.g., due to pelvic inflammatory disease, endometriosis) can lead to infertility. Ectopic pregnancy (implantation of a fertilized egg outside the uterus, most commonly in the fallopian tube) is a significant clinical concern.
3. Uterus
- Description: A hollow, thick-walled, pear-shaped muscular organ located in the pelvic cavity, between the bladder (anteriorly) and the rectum (posteriorly). It is where a fertilized egg implants and develops during pregnancy.
- Size (non-gravid): Approximately 7.5 cm (3 inches) long, 5 cm (2 inches) wide, and 2.5 cm (1 inch) thick.
- Support: Held in place by various ligaments, including the broad ligament, round ligament, uterosacral ligaments, and cardinal ligaments.
- Parts of the Uterus:
- Fundus: The dome-shaped, superior-most portion of the uterus, above the openings of the fallopian tubes. This is the primary site of implantation for a normal pregnancy.
- Body (Corpus): The main, largest part of the uterus, extending from the fundus to the isthmus.
- Isthmus: A constricted, narrow region between the body and the cervix.
- Cervix: The lowermost, cylindrical portion of the uterus that projects into the vagina.
- Wall Structure (Three Layers):
- Perimetrium: The outermost serous layer, part of the peritoneum.
- Myometrium: The thick, muscular middle layer, composed of interlacing bundles of smooth muscle. It is responsible for uterine contractions during labor and menstruation. It expands significantly during pregnancy (10-20 times its normal size) to accommodate the growing fetus.
- Endometrium: The inner lining of the uterine cavity, a vascular and glandular mucous membrane. It undergoes cyclical changes in response to ovarian hormones (estrogen and progesterone).
- Function: Thickens during the menstrual cycle to prepare for the implantation of a fertilized egg. If fertilization and implantation do not occur, the superficial layer of the endometrium (stratum functionalis) is shed during menstruation. If implantation occurs, the endometrium forms the maternal part of the placenta.
- Function:
- Implantation and Gestation: Provides a suitable environment for the implantation of a fertilized egg and supports fetal development throughout pregnancy.
- Childbirth: Its strong muscular contractions (myometrium) are crucial for expelling the fetus during labor.
- Menstruation: Sheds its inner lining if pregnancy does not occur.
4. The Cervix
- Description: The narrow, cylindrical lower portion of the uterus that connects the uterus to the vagina. It acts as a gateway between the uterine cavity and the vaginal canal.
- Parts:
- Ectocervix: The portion that protrudes into the vagina, covered by stratified squamous epithelium.
- Endocervix: The canal leading into the uterus, lined by columnar glandular epithelium.
- Transformation Zone: The area where the squamous epithelium of the ectocervix meets the columnar epithelium of the endocervix. This area is highly susceptible to HPV infection and cervical cancer.
- Cervical Mucus: The endocervical glands produce cervical mucus, which changes in quality and quantity throughout the menstrual cycle, primarily influenced by estrogen and progesterone levels:
- High Estrogen (around ovulation): Mucus becomes thin, watery, clear, and "stretchy" (spinnbarkeit). This creates a "friendly environment" for sperm, facilitating their passage into the uterus.
- High Progesterone (after ovulation, during luteal phase): Mucus becomes thick, viscous, and opaque, forming a "mucus plug" that hinders sperm entry and protects the uterine cavity from infection.
- Function:
- Sperm Transport and Barrier: Regulates sperm entry into the uterus.
- Protection: Acts as a physical and chemical barrier against infection ascending from the vagina into the uterus.
- Childbirth: During labor, the cervix effaces (thins) and dilates (opens) significantly (up to 10 cm or roughly 50 times its normal width), allowing the baby to exit the uterus into the vagina.
5. The Vagina
- Description: A muscular, elastic tube (sheath-like, from Latin "vagin") that extends from the cervix to the vulva (external genitalia).
- Length: Typically 7-10 cm (3-4 inches) long.
- Structure:
- Muscular Wall: Composed of smooth muscle with an inner layer of stratified squamous epithelium, arranged in transverse folds called rugae, which allow for significant distension during childbirth and intercourse.
- No Glands: The vaginal wall itself does not contain glands. Lubrication primarily comes from cervical mucus, transudation across the vaginal walls during arousal, and secretions from the greater and lesser vestibular glands.
- Function:
- Sexual Intercourse: Receives the penis and sperm during coitus. Its elasticity and rugae accommodate the penis.
- Birth Canal: Serves as the passageway for the baby during vaginal delivery.
- Menstrual Flow: Provides an exit route for menstrual blood from the uterus.
- Protection: Its acidic environment (due to lactic acid produced by normal bacterial flora, primarily Lactobacillus) helps protect against pathogenic infections.
Female Reproductive Quiz
Systems Anatomy
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Female Reproductive Quiz
Systems Anatomy
Preparing questions...
Exam Completed!
See your performance breakdown below.