Thyroid Gland Physiology
THE THYROID GLAND
The endocrine system is a complex network of ductless glands that play a crucial role in maintaining homeostasis. Unlike exocrine glands that secrete substances through ducts, endocrine glands release their chemical messengers, known as hormones, directly into the surrounding extracellular fluid. These hormones then pass into the bloodstream for general circulation, traveling throughout the body to reach distant target organs or tissues. Upon arrival, hormones excite or inhibit the activity of these target cells, coordinating a vast array of physiological processes, including growth, metabolism, reproduction, and mood.
The thyroid gland is a vital component of this system, holding the distinction of being one of the largest endocrine glands in the human body. Its primary function is to regulate metabolism, energy production, and body temperature.
II. HORMONES SECRETED BY THE THYROID GLAND
The thyroid gland produces two main types of hormones:
1. Thyroid Hormones (from Follicular Cells)
- Thyroxine (T4): Also known as tetraiodothyronine. This is the more abundant hormone secreted by the thyroid. While less biologically active at the cellular level compared to T3, it serves as a prohormone and a major reservoir for T3.
- Triiodothyronine (T3): This is the more potent and biologically active form of thyroid hormone. Most of the T3 in the body is generated from the deiodination (removal of an iodine atom) of T4 in peripheral tissues, rather than directly secreted by the thyroid gland.
- Origin: Both T4 and T3 are synthesized and secreted by the follicular cells of the thyroid gland.
2. Calcitonin (from Parafollicular Cells)
- Calcitonin: This hormone plays a role in calcium homeostasis.
- Origin: It is secreted by specialized cells located interspersed between the thyroid follicles, known as parafollicular cells (also called C cells). Calcitonin's primary function is to lower blood calcium levels, acting in opposition to parathyroid hormone.
III. FUNCTIONAL ANATOMY OF THE THYROID GLAND
Understanding the structure of the thyroid gland is key to comprehending its function.
- Location: The thyroid gland is strategically located in the neck, immediately below the larynx (voice box) and anterior (in front of) to the upper part of the trachea (windpipe). Its superficial position makes it palpable during physical examination.
- Weight: In a healthy adult, it typically weighs about 15-20 grams. Its size can fluctuate with age, sex, and physiological states (e.g., pregnancy).
- Gross Structure: The gland consists of two distinct, fleshy lateral lobes, one on each side of the trachea. These lobes are connected by a narrow band of thyroid tissue called the isthmus.
- The isthmus usually overlies the region from the 2nd to the 4th tracheal cartilage, anchoring the gland in place.
- Sometimes, a third lobe, the pyramidal lobe, may extend upwards from the isthmus.
IV. PHYSIOLOGICAL ANATOMY: THE FUNCTIONAL UNIT
The microscopic structure of the thyroid gland reveals its unique functional organization:
- Thyroid Follicles: The lobes of the thyroid gland are composed of numerous, tiny, hollow, spherical structures called follicles. These follicles are the functional units of the thyroid gland. Each follicle is lined by a single layer of cuboidal epithelial cells – the follicular cells.
- Colloid: Each follicle is filled with a thick, sticky, protein-rich substance known as colloid.
- The major constituent of colloid is a large glycoprotein called thyroglobulin (Tg). This thyroglobulin serves as the scaffold for thyroid hormone synthesis and, crucially, as the storage site for thyroid hormones.
- Storage Uniqueness: Unlike most other endocrine glands, which store minimal amounts of pre-formed hormones and secrete them rapidly upon synthesis, the thyroid gland has a remarkable capacity to store considerable amounts of thyroid hormones within the colloid (bound within thyroglobulin). This stored reserve can last for several weeks or even months, providing a buffer against fluctuations in iodine intake or acute demand.
- C Cells (Parafollicular Cells): Interspersed between the follicles are the C cells (or parafollicular cells), which, as mentioned, are responsible for secreting calcitonin.
V. SYNTHESIS, STORAGE, AND SECRETION OF THYROID HORMONES
The production of thyroid hormones is a multi-step process that requires specific raw materials and intricate cellular machinery.
Essential Precursors:
- Tyrosine: An amino acid that forms the backbone of thyroid hormones. It is synthesized by the body and is a crucial component of the thyroglobulin protein.
- Iodine: A trace element that is dietary essential. The body cannot synthesize iodine; it must be consumed through diet (e.g., iodized salt, seafood). Iodine is absolutely critical for thyroid hormone synthesis, as it is incorporated directly into the tyrosine residues of thyroglobulin.
Location of Synthesis: Hormone synthesis primarily occurs on the thyroglobulin molecule within the colloid.
Steps Involved in Synthesis, Storage, and Secretion:
1. Thyroglobulin Production and Release (Into Colloid)
- Process: Follicular cells synthesize the large glycoprotein thyroglobulin (Tg) in their endoplasmic reticulum and Golgi apparatus.
- Delivery: Tg is then packaged into vesicles and released into the follicular lumen (colloid space) by exocytosis.
2. Iodine Uptake (Iodide Trapping)
- Process: Follicular cells actively transport iodide (I-) from the bloodstream into their cytoplasm. This is an energy-dependent process mediated by the Sodium-Iodide Symporter (NIS), which co-transports two Na+ ions with one I- ion.
- Concentration: This mechanism allows the thyroid gland to concentrate iodide to levels 20-40 times higher than in the blood (and even higher under TSH stimulation).
- Transfer to Colloid: Iodide is then transported from the follicular cell cytoplasm into the colloid space via a chloride-iodide counter-transporter called pendrin.
3. Oxidation and Iodination of Tyrosine (Organification)
- Process: Once in the colloid, iodide is rapidly oxidized to iodine (I0 or I+) by the enzyme thyroid peroxidase (TPO).
- Attachment to Tyrosine: This highly reactive iodine then attaches to specific tyrosine residues within the thyroglobulin molecule.
- One iodine atom attached to a tyrosine forms Monoiodotyrosine (MIT).
- Two iodine atoms attached to a tyrosine form Diiodotyrosine (DIT).
4. Coupling Processes (Formation of T4 and T3)
- Process: Still within the thyroglobulin molecule, two iodinated tyrosine molecules are coupled together by TPO:
- DIT + DIT → T4 (Thyroxine): This is the primary coupling reaction, forming the most abundant thyroid hormone.
- MIT + DIT → T3 (Triiodothyronine): This reaction produces T3. Less commonly, DIT + MIT can also form reverse T3 (rT3), which is metabolically inactive.
- Storage: The newly formed T4 and T3 remain covalently bound within the large thyroglobulin molecule, stored as part of the colloid, often for weeks.
Secretion (Upon Stimulation):
- Stimulus: When the body requires thyroid hormones, the follicular cells are stimulated, primarily by Thyroid-Stimulating Hormone (TSH) from the anterior pituitary.
- Endocytosis: Follicular cells engulf small portions of the colloid (containing iodinated thyroglobulin) by endocytosis, forming colloid droplets.
- Lysosomal Digestion: These colloid droplets fuse with lysosomes within the follicular cells. Lysosomal proteases digest the thyroglobulin, releasing free T4, T3, MIT, and DIT.
- Recycling and Diffusion:
- MIT and DIT are deiodinated by the enzyme deiodinase, and the released iodide is recycled back into the synthesis pathway.
- Free T4 and T3 are then released into the bloodstream by diffusing out of the follicular cell.
Quantitative and Potency Differences:
- Approximately 93% of the hormones released from the thyroid gland initially appear in the form of T4.
- However, a majority of the T4 that is secreted from the thyroid gland is subsequently converted to T3 in peripheral tissues (e.g., liver, kidneys, target cells) by enzymes called deiodinases.
- T3 is approximately 4 times more potent in its biologic form than T4 and is considered the major hormone that interacts with the target cells to produce most of the thyroid hormone effects.
- T3 is much smaller in quantities in the circulation and persists for a much shorter time (shorter half-life) than T4, reflecting its higher activity and rapid utilization.
Transport in Blood:
- Both T4 and T3 are highly lipophilic. Once in the blood, they immediately bind to plasma proteins.
- Thyroid hormone-specific binding globulin (TBG) is the primary carrier, transporting 70-80% of thyroid hormones. Other non-specific proteins like albumin and transthyretin also transport a portion.
- Less than 0.1% of T4 and 1% of T3 are in the unbound (free) form.
- These free T4 (fT4) and free T3 (fT3) compounds are biologically active, and it is these components that produce the effects of the thyroid hormones on peripheral tissues and participate in the pituitary feedback mechanism. The protein-bound hormones serve as a circulating reservoir.
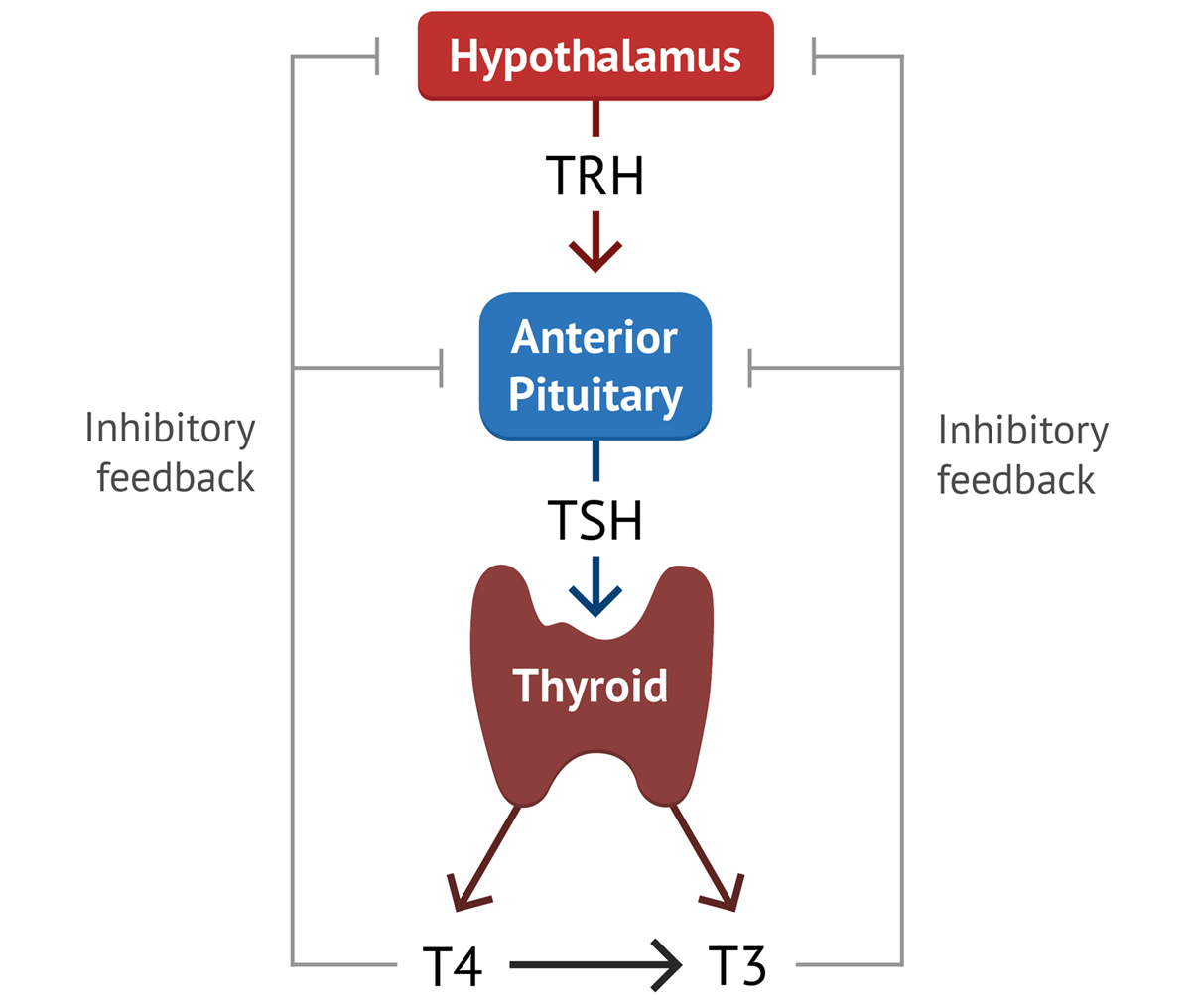
VI. REGULATION OF THYROID HORMONE SECRETION (THE HPT AXIS)
Thyroid hormone secretion is under precise control of the hypothalamic-pituitary-thyroid (HPT) axis, a classic example of a negative feedback loop.
- Hypothalamic Control (TRH):
- The secretion of thyroid-stimulating hormone (TSH) from the anterior pituitary is primarily controlled by Thyrotropin-Releasing Hormone (TRH), a tripeptide hormone.
- TRH is secreted by specialized neurosecretory cells in the paraventricular nucleus of the hypothalamus. It is then released into the hypophyseal portal system, which carries it to the anterior pituitary.
- Pituitary Control (TSH):
- Upon reaching the anterior pituitary, TRH stimulates the thyrotroph cells to synthesize and secrete Thyroid-Stimulating Hormone (TSH) (also known as thyrotropin).
- TSH is a glycoprotein hormone that has several effects on the thyroid gland:
- Increases iodide uptake (iodide trapping).
- Increases synthesis of thyroglobulin.
- Increases TPO activity (iodination and coupling).
- Increases endocytosis of colloid and release of T4 and T3.
- Promotes the growth and vascularity of the thyroid gland.
- Thyroid Gland Response (T4 and T3):
- TSH stimulates the thyroid gland to produce and release T4 and T3.
- Negative Feedback (Inhibition of TRH and TSH):
- Elevated levels of free T4 and T3 in the body fluids exert negative feedback on both the hypothalamus and the anterior pituitary.
- They inhibit the secretion of TRH from the hypothalamus.
- They directly inhibit the sensitivity of the thyrotroph cells in the anterior pituitary to TRH, thereby decreasing the secretion of TSH.
- This feedback mechanism ensures that thyroid hormone levels are maintained within a narrow physiological range. For example, if thyroid hormone levels are 1.75 times normal, the rate of TSH secretion can fall essentially to zero as the feedback mechanism shuts down pituitary stimulation.
- Elevated levels of free T4 and T3 in the body fluids exert negative feedback on both the hypothalamus and the anterior pituitary.
- Other Factors Influencing Regulation:
- Cold Exposure: Exposure of an animal (and to some extent, humans, especially infants) to coldness significantly increases the rate of TRH secretion from the hypothalamus. This leads to increased TSH and subsequent thyroid hormone production, which helps to increase metabolic rate and heat production.
- Stress and Excitement: Conditions causing acute stress, excitement, and strong sympathetic nervous system stimulation (e.g., severe anxiety, trauma) can cause an acute decrease in the secretion of TSH, likely by inhibiting TRH release. This is thought to be a protective mechanism to prevent an excessive metabolic response during acute stress.
- Iodine Availability: Chronic iodine deficiency leads to decreased thyroid hormone synthesis, reduced negative feedback, and thus chronically elevated TSH. Prolonged high TSH can lead to thyroid gland enlargement (goiter).
PHYSIOLOGICAL EFFECTS OF THYROID HORMONES
Thyroid hormones (T3 and T4) are essential for normal growth, development, and metabolism in virtually every cell of the body. Their widespread actions underscore their critical role in maintaining overall physiological homeostasis.
I. METABOLIC RATE AND HEAT PRODUCTION
The most fundamental and pervasive effect of thyroid hormones is their impact on the body's metabolic rate.
- A. Increased Metabolic Activities: Thyroid hormones act as a crucial metabolic thermostat. They increase the rate of most metabolic activities in almost all cells of the body. This involves stimulating various cellular processes, including:
- Increased synthesis and degradation of proteins.
- Increased glucose utilization and fat metabolism.
- Enhanced activity of enzymes involved in nutrient oxidation.
- B. Increased Oxygen Consumption: As metabolic processes are upregulated, there is a corresponding increase in oxygen consumption by most metabolically active tissues. This is a direct consequence of enhanced cellular respiration to meet the increased energy demand. This effect is particularly pronounced in tissues like muscle, liver, and kidneys, but excludes the brain, gonads, and spleen, which are less affected by thyroid hormone-induced increases in oxygen consumption.
- C. Elevated Basal Metabolic Rate (BMR): Thyroid hormones are the primary determinants of the Basal Metabolic Rate (BMR), which is the minimum energy expenditure required to maintain essential body functions at rest.
- In conditions of hyperthyroidism (excess thyroid hormone), the BMR can significantly increase by 60-100% above normal. This means the body burns far more calories at rest.
- Conversely, in hypothyroidism (deficient thyroid hormone), the BMR can fall to 40-50% below normal, leading to reduced energy expenditure and weight gain.
- D. Calorigenic Effect: Since increased metabolism results in increased heat production, thyroid hormones have a distinct calorigenic effect.
- The heightened cellular activity and ATP turnover generate more heat as a byproduct.
- This effect is crucial for maintaining body temperature, especially in cold environments. Individuals with hyperthyroidism often complain of heat intolerance due to excessive heat production, while those with hypothyroidism often experience cold intolerance.
II. INTERMEDIARY METABOLISM
Thyroid hormones significantly modulate the rates of many specific reactions involved in the metabolism of carbohydrates, fats, and proteins.
- A. Carbohydrate Metabolism:
- Increased Glucose Uptake: Enhances glucose uptake by cells.
- Increased Glycolysis and Gluconeogenesis: Stimulates both the breakdown of glucose for energy and the synthesis of glucose from non-carbohydrate sources.
- Increased Insulin Secretion: Indirectly increases insulin secretion in response to increased blood glucose levels.
- Increased Glycogenolysis: Promotes the breakdown of glycogen stores (in liver and muscle) to release glucose.
- B. Fat Metabolism:
- Increased Lipolysis: Enhances the breakdown of triglycerides from adipose tissue, releasing fatty acids and glycerol into the blood.
- Reduced Cholesterol and Triglyceride Levels: Thyroid hormones accelerate the removal of cholesterol and triglycerides from the plasma by increasing the number of LDL receptors on liver cells, leading to increased clearance of LDL cholesterol. This is why hypothyroidism is often associated with elevated cholesterol levels.
- C. Protein Metabolism:
- Anabolic and Catabolic Effects: Thyroid hormones are crucial for normal protein synthesis and degradation.
- Physiological vs. Excess Levels: At physiological concentrations, thyroid hormones promote protein synthesis, which is essential for growth and development. However, in excessive amounts (hyperthyroidism), they can cause a net increase in protein catabolism, leading to muscle wasting and negative nitrogen balance.
III. SYMPATHOMIMETIC EFFECT
Thyroid hormones have a remarkable interaction with the sympathetic nervous system.
- A. Definition of Sympathomimetic: A sympathomimetic effect refers to any action that is similar to or mimics those produced by the sympathetic nervous system (the "fight or flight" response), typically by activating adrenergic receptors.
- B. Increased Responsiveness to Catecholamines: Thyroid hormones significantly increase the target cell responsiveness to catecholamines (epinephrine and norepinephrine), which are the neurotransmitters of the sympathetic nervous system and hormones from the adrenal medulla.
- Permissive Action: Thyroid hormones are said to be "permissive" in this context. They do not directly cause sympathetic effects but rather enhance the ability of catecholamines to produce their effects. They achieve this by increasing the production of specific catecholamine target cell receptors, particularly beta-adrenergic receptors, on target cells.
- Clinical Relevance: This explains why many symptoms of hyperthyroidism (e.g., rapid heart rate, tremors, anxiety, sweating) resemble an overactive sympathetic nervous system, even though catecholamine levels themselves might not be significantly elevated.
IV. THE CARDIOVASCULAR SYSTEM
The cardiovascular system is highly sensitive to thyroid hormone levels, exhibiting profound changes in both hyper- and hypothyroidism.
- A. Increased Responsiveness to Catecholamines (Reiteration): The permissive action discussed above is particularly evident in the heart. Thyroid hormones increase the heart's responsiveness to circulating catecholamines, making it more sensitive to their stimulatory effects.
- B. Increased Heart Rate and Force of Contraction: Directly and indirectly (via enhanced catecholamine sensitivity), thyroid hormones:
- Increase heart rate (tachycardia): Even at rest.
- Increase the force of myocardial contraction: Leading to a stronger heartbeat.
- These combined effects result in a significant increase in cardiac output (CO), the volume of blood pumped by the heart per minute. This increased cardiac output is necessary to deliver the additional oxygen required by the metabolically active tissues.
- C. Peripheral Vasodilation: In response to the increased metabolic rate and resulting heat load generated by the calorigenic effect, the body needs to dissipate excess heat. Thyroid hormones promote peripheral vasodilation (widening of blood vessels in the skin).
- This increases blood flow to the skin, allowing for more heat to be transferred from the body's core to the surface, facilitating heat loss and helping to maintain a stable core body temperature.
V. GROWTH AND THE NERVOUS SYSTEM
Thyroid hormones are indispensable for normal growth, maturation, and neurological development.
- A. Effect on Growth (Mainly in Growing Children):
- Stimulates GH Secretion and Action: Thyroid hormone stimulates the secretion of Growth Hormone (GH) from the anterior pituitary and promotes the effects of GH on target tissues. This synergistic action is crucial for skeletal and tissue growth.
- Growth Retardation in Deficiency: A lack of thyroid hormone during childhood (juvenile hypothyroidism or cretinism) leads to severe growth retardation (dwarfism). Importantly, this can often be reversed by thyroid replacement therapy if administered early enough.
- No Excessive Growth in Excess: Unlike excess GH, excess thyroid hormone does not produce excessive growth. In fact, severe hyperthyroidism can sometimes lead to muscle wasting and weight loss, counteracting growth.
- B. Important for Brain Development: Thyroid hormones are critically important in promoting the normal growth and development of the brain during fetal and postnatal life, particularly during the first few years after birth.
- Myelination and Synaptogenesis: They are essential for processes like neuronal migration, myelination (formation of the myelin sheath around nerves), and synaptogenesis (formation of connections between neurons).
- Mental Retardation in Deficiency: Thyroid hormone deficiency during this critical period (e.g., congenital hypothyroidism, historically called cretinism) leads to severe and irreversible mental retardation if thyroid replacement therapy is not administered within days or weeks after birth. The damage to the developing brain is permanent if left untreated.
VI. OTHER EFFECTS OF THYROID HORMONES
Thyroid hormones have diverse effects on many other physiological systems.
- A. Effect on Sleep:
- Hyperthyroidism: Due to the excitable effects of thyroid hormone on the synapses and the general increase in metabolic activity, individuals with hyperthyroidism often experience difficulty sleeping (insomnia), even though they may feel constantly tired. They are in a state of heightened alertness and nervous excitability.
- Hypothyroidism: Conversely, in hypothyroidism, reduced metabolic activity leads to decreased mental alertness and sluggishness. Individuals often experience excessive sleepiness, with sleep sometimes lasting for 12 to 14 hours a day or more.
- B. Effect on Sexual Function:
- Normal Function Requires Normal Levels: For normal sexual function in both men and women, thyroid secretion needs to be within the normal range. Both hypo- and hyperthyroidism can disrupt reproductive health.
- In Men:
- Lack of thyroid hormone (hypothyroidism) can lead to loss of libido (sex drive) and, in severe cases, impotence.
- Great excesses (hyperthyroidism) can also sometimes lead to impotence or decreased libido.
- In Women:
- Lack of thyroid hormone (hypothyroidism) is often associated with menstrual irregularities such as menorrhagia (excessive menstrual bleeding) and polymenorrhea (frequent menstrual bleeding). In severe cases, it can lead to anovulation and infertility.
- Hyperthyroidism can cause oligomenorrhea (infrequent menstruation) or amenorrhea (absence of menstruation), and reduced fertility.
DISORDERS OF THYROID METABOLISM
Thyroid hormones are powerful regulators, and deviations from their precise physiological range can lead to significant and widespread health problems. These disorders are broadly categorized into two main types: hypothyroidism (deficient thyroid hormone) and hyperthyroidism (excessive thyroid hormone).
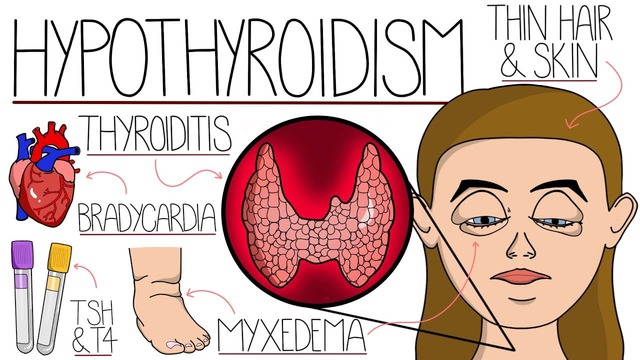
I. HYPOTHYROIDISM: THYROID HORMONE DEFICIENCY
Hypothyroidism refers to the clinical syndrome resulting from a deficiency of thyroid hormones. This underactivity of the thyroid gland can arise from several causes affecting different levels of the hypothalamic-pituitary-thyroid (HPT) axis.
Etiology (Causes):
Hypothyroidism develops if the thyroid gland shows:
- i. Atrophy or Destruction of Secretory Cells: This is the most common cause, often due to autoimmune disease.
- Hashimoto's Thyroiditis: The most prevalent cause of primary hypothyroidism in iodine-sufficient regions. It is an autoimmune disorder where the body's immune system attacks and gradually destroys the thyroid follicular cells.
- Thyroidectomy: Surgical removal of all or part of the thyroid gland (e.g., for thyroid cancer, severe hyperthyroidism).
- Radioactive Iodine (RAI) Therapy: Used to treat hyperthyroidism or thyroid cancer, RAI destroys thyroid tissue.
- External Radiation: Radiation therapy to the neck region for other cancers can damage the thyroid.
- ii. Inadequate Stimulation: This refers to central (secondary or tertiary) hypothyroidism.
- Secondary Hypothyroidism: Due to a deficiency of TSH (Thyroid-Stimulating Hormone) secretion from the anterior pituitary gland. Causes include pituitary tumors, surgery, radiation, or infarction (Sheehan's syndrome).
- Tertiary Hypothyroidism: Due to a deficiency of TRH (Thyrotropin-Releasing Hormone) secretion from the hypothalamus. This is less common and often associated with other hypothalamic-pituitary dysfunctions.
- iii. Iodine Deficiency: A global cause of hypothyroidism, especially in regions with inadequate dietary iodine. Without sufficient iodine, the thyroid cannot synthesize adequate amounts of T4 and T3.
- iv. Genetic Defects: Rare genetic disorders affecting thyroid hormone synthesis (e.g., defects in TPO, thyroglobulin synthesis, or iodine trapping).
- v. Drugs: Certain medications can impair thyroid function (e.g., lithium, amiodarone, interferon-alpha).
Clinical Presentation & Symptoms of Hypothyroidism
The symptoms are varied and reflect a generalized slowing down of metabolic processes.
- Metabolic & General: Decreased BMR (<40-50% normal), Poor Tolerance of Cold, Tendency to Gain Weight, Fatigue and Extreme Somnolence (12-14 hrs/day), Easily Fatigued.
- Cardiovascular: Reduced Heart Rate (Bradycardia), Reduced Blood Pressure, Reduced Cardiac Output.
- Gastrointestinal: Sluggish Gut Movements (Constipation).
- Neurological & Psychiatric: Decreased Alertness, Slow Speech, Poor Memory, Depression, Paresthesias (carpal tunnel).
- Musculoskeletal: Muscle Weakness and Aches.
- Skin, Hair, & Voice: Dry Coarse Skin, Brittle Hair/Nails (Queen Anne's sign), Hoarseness/Slow Husky Voice.
- Reproductive: Menorrhagia/polymenorrhea/infertility (Women), Decreased libido/erectile dysfunction (Men).
Treatment for Hypothyroidism:
- Replacement Therapy: The primary treatment is lifelong replacement therapy with synthetic thyroid hormone, typically levothyroxine (synthetic T4).
- Iodine Supplementation: For iodine deficiency-induced hypothyroidism, iodine supplementation is crucial, often in combination with thyroid hormone if severe deficiency has already occurred.
- Promptness: Timely diagnosis and treatment are essential, particularly in infants and children, to prevent irreversible developmental deficits.
Myxedema (Severe Adult Hypothyroidism)
- Description: Myxedema is a severe form of hypothyroidism that develops in patients with an almost total lack of thyroid hormone function.
- Pathophysiology: Accumulation of hydrophilic mucopolysaccharides (hyaluronic acid and chondroitin sulfate) in interstitial spaces, forming "tissue gel" (mucinous edema).
- Clinical Manifestations: Non-Pitting Edema, Characteristic Swelling (bagginess under eyes, puffy face).
- Myxedema Coma: A rare, life-threatening complication characterized by profound hypothermia, hypoventilation, bradycardia, hypotension, and altered mental status.
Atherosclerosis in Hypothyroidism:
- Increased Blood Cholesterol: Due to altered fat metabolism and diminished liver excretion of cholesterol.
- Increased Atherosclerosis Risk: Strongly associated with hardening/narrowing of arteries.
- Serious Complications: Peripheral Vascular Disease, Deafness, Coronary Artery Disease.
Cretinism (Congenital Hypothyroidism)
- Description: Caused by extreme hypothyroidism during fetal life, infancy, or early childhood.
- Key Characteristics: Failure of body growth (dwarfism) and profound mental retardation.
- Causes: Congenital Lack of Thyroid Gland, Genetic Defects, Iodine Lack (Endemic Cretinism), Maternal Hypothyroidism.
- Irreversible Mental Retardation: If treatment is not administered within days or weeks after birth, the mental retardation becomes permanent.
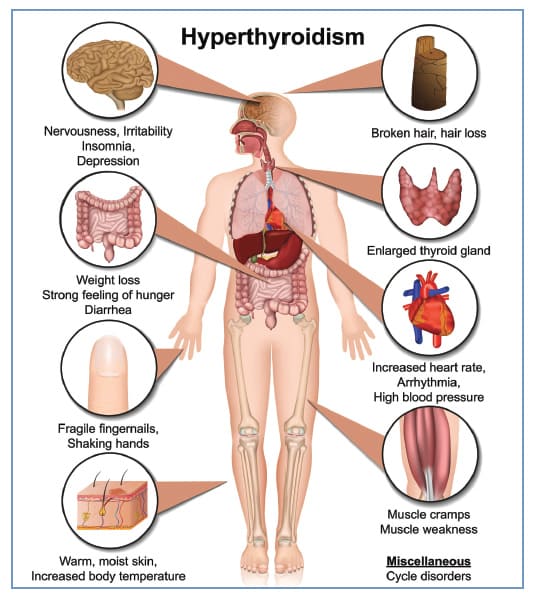
II. HYPERTHYROIDISM: THYROID HORMONE EXCESS
Hyperthyroidism is the clinical syndrome resulting from excessive secretion of thyroid hormones. This leads to a hypermetabolic state with widespread effects.
Etiology (Causes):
- Graves' Disease (Most Common Cause):
- Autoimmune Disease: Body produces antibodies called Thyroid-Stimulating Immunoglobulins (TSI) or TSH receptor antibodies (TRAb).
- TSH Receptor Agonists: TSI bind to TSH receptors and mimic TSH action, continuously stimulating the gland.
- Autonomous Stimulation: Not subject to negative feedback.
- Diffuse Goiter: Uniform enlargement of the gland.
- Toxic Multinodular Goiter (Plummer's Disease): Multiple nodules become autonomous.
- Toxic Adenoma: Single benign tumor autonomously producing hormones.
- Thyroiditis: Inflammation causing temporary release of stored hormones.
- Exogenous Thyroid Hormone Intake: Overdose.
- TSH-Secreting Pituitary Adenoma: Rare tumor.
- Iodine-Induced Hyperthyroidism (Jod-Basedow Phenomenon): Exposure to large iodine amounts in pre-existing abnormality.
Clinical Presentation & Symptoms of Hyperthyroidism
- Metabolic & General: High State of Excitability, Intolerance to Heat/Increased Sweating, Mild to Extreme Weight Loss (despite polyphagia), Extreme Fatigue but Inability to Sleep, Fine Tremor of Hands.
- Cardiovascular: Tachycardia, Palpitations, Increased Cardiac Output, Arrhythmias (Atrial fibrillation), Hypertension.
- Gastrointestinal: Diarrhea.
- Musculoskeletal: Muscle Weakness (Proximal Myopathy), Osteoporosis.
- Neurological & Psychiatric: Nervousness/Anxiety, Psychic Disorders, Hyperreflexia.
F. Eye Changes (Exophthalmos in Graves' Disease)
- Exophthalmos (Proptosis): Prominent feature of Graves' disease only.
- Pathophysiology: Autoimmune inflammatory process behind eyeballs. Deposition of complex water-retaining carbohydrates.
- Mechanism: Fluid retention pushes eyeballs forward.
- Clinical Significance: Eye irritation, dryness, diplopia, optic nerve compression.
Treatment for Hyperthyroidism:
- A. Antithyroid Drugs:
- Propylthiouracil (PTU) and Methimazole (Tapazole): Mechanism: Inhibit thyroid peroxidase (TPO), blocking iodination and coupling. PTU also blocks peripheral conversion of T4 to T3.
- B. Radioactive Iodine (RAI) Therapy: Oral administration of I-131. Selectively destroys follicular cells.
- C. Surgical Removal (Thyroidectomy): Excision of part or most of the gland.
- D. Other Treatments:
- Beta-Blockers (e.g., Propranolol): Symptomatic relief of adrenergic symptoms.
- Inorganic Iodides (e.g., Lugol's solution): In high concentrations (Wolf-Chaikoff Effect), acutely reduce iodide trapping and inhibit hormone release. Used for preparation for surgery or thyroid storm.
III. GOITER: ENLARGEMENT OF THE THYROID GLAND
A goiter simply means an enlargement of the thyroid gland. It can occur in hypothyroidism, hyperthyroidism, or euthyroid states.
A. Toxic Goiter:
- Definition: Enlargement associated with increased secretion of thyroid hormones.
- Causes: Graves' disease (diffuse toxic goiter), toxic multinodular goiter, toxic adenoma.
B. Non-Toxic Goiter (Euthyroid Goiter or Hypothyroid Goiter):
- Definition: Enlargement without an increase in hormone secretion. Often called a "compensatory goiter."
- Causes: Attempt to compensate for decreased hormone production. Stimulated by elevated TSH.
- 1. Endemic Colloid Goiter: Most common cause is iodine deficiency (dietary intake < 50 µg/day). Pituitary secretes more TSH, leading to hypertrophy/hyperplasia and accumulation of poorly iodinated thyroglobulin. Prevalent in mountainous regions.
- 2. Sporadic (Idiopathic) Non-Toxic Goiter: Occurs in regions with sufficient iodine. Causes include mild genetic defects, consumption of goitrogens (cassava, cabbage, turnips), mild thyroiditis, or nodular goiter.
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908
Thyroid Gland Quiz
Systems Physiology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Thyroid Gland Quiz
Systems Physiology
Preparing questions...
Exam Completed!
See your performance breakdown below.