Intracellular Accumulation & Acute Inflammation
Intracellular Accumulation & Acute Inflammation
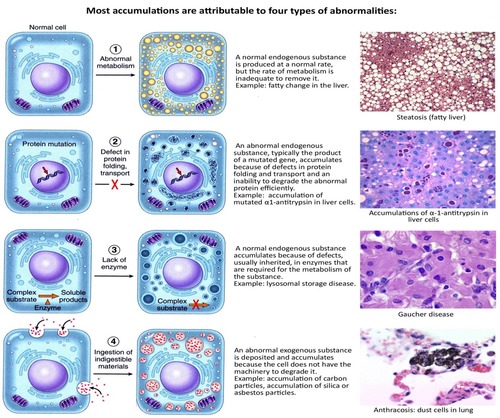
Intracellular Accumulations
Intracellular accumulations are the buildup of substances—such as lipids, proteins, glycogen, or pigments—within cells due to metabolic derangements, genetic defects, or environmental factors.
These accumulations occur in the cytoplasm or nucleus, ranging from harmless to severely toxic, causing reversible or irreversible cell injury. Key mechanisms include increased production, defective metabolism/transport, or lack of enzymes to break down substances.
Cells often act as reservoirs for metabolic products or exogenous substances. These accumulations represent a sign of metabolic derangement.
Subcellular Localization
- Cytoplasm: Most common (e.g., Fatty change, Glycogen).
- Organelles: Specifically within Lysosomes (e.g., Pompe disease) or the Endoplasmic Reticulum (e.g., Protein folding defects).
- Nucleus: Rare, but seen in certain viral infections or lead poisoning.
The Four Pathological Mechanisms
- Abnormal Metabolism: A normal endogenous substance (like water, lipids, or proteins) is produced at a normal or increased rate, but the metabolic rate is inadequate for its removal (e.g., Steatosis).
- Defect in Protein Folding/Transport: Genetic mutations or acquired defects cause proteins to misfold. These "garbage" proteins build up because they cannot be exported or degraded (e.g., α1-antitrypsin deficiency).
- Enzymatic Deficiency: An inherited lack of a vital enzyme (usually lysosomal) means a specific substrate cannot be broken down, leading to massive buildup—known as Storage Diseases.
- Inability to Degrade Exogenous Material: The cell encounters a substance (like carbon or silica) for which it has no natural enzymes to digest.
Examples of Abnormal Accumulations
1. Fatty Change (Steatosis)
The abnormal accumulation of triglycerides within parenchymal (functional) cells.
- Organ Involvement: Primarily the Liver (yellow, greasy, enlarged). It is also significant in the Heart (where it can cause "Tiger effect" banding) and the Kidneys.
- Etiology (The "Why"):
- Toxins: Most notably Alcohol, which is a mitochondrial toxin that impairs fat oxidation.
- Protein Malnutrition: Lack of "apoproteins" needed to carry fat out of the liver.
- Anoxia: Lack of oxygen prevents the oxidation (burning) of fatty acids.
- Diabetes Mellitus & Obesity: Causes an oversupply of free fatty acids to the liver.
2. Cholesterol and Cholesteryl Esters
- Pathology: Unlike triglycerides, cholesterol is usually stored in macrophages or smooth muscle cells.
- Atherosclerosis: The most critical clinical result. Phagocytic cells in the large arteries become overloaded with lipid, forming "Foam Cells." These accumulate in the intimal layer of arteries, leading to yellow fatty streaks and eventually plaques.
3. Proteins
- Morphology: Appear as rounded, eosinophilic (bright pink) droplets, vacuoles, or aggregates.
- Clinical Examples:
- Nephrotic Syndrome: Excess protein leaks into the kidney tubules; the cells reabsorb it, creating pink protein droplets.
- Russell Bodies: Found in plasma cells (overproduction of immunoglobulins).
- Misfolded Proteins: Build up in the brain (Amyloid plaques in Alzheimer's).
4. Glycogen
- Association: Highly associated with Glucose metabolism disorders.
- Diabetes Mellitus: Glycogen is found in the epithelial cells of the distal segments of the renal tubules and the liver.
- Glycogen Storage Diseases (GSD): Genetic defects where glycogen cannot be converted back to glucose, leading to massive cell death and organ failure.
5. Pigments: The "Colored" Pathologies
- Exogenous (Environmental):
- Carbon (Anthracosis): The most ubiquitous pigment. Inhaled carbon is phagocytosed by alveolar macrophages. These macrophages travel through the lymphatics to the tracheobronchial lymph nodes. In coal miners, this leads to "Black Lung" disease (Coal Workers' Pneumoconiosis).
- Endogenous (Produced by the body):
- Lipofuscin: A "wear-and-tear" pigment. It is a sign of free radical injury and lipid peroxidation. It does not harm the cell but is a tell-tale marker of aging.
- Melanin: An insoluble brown-black pigment produced by melanocytes in the epidermis to protect against UV radiation.
- Hemosiderin: A hemoglobin-derived, golden-yellow to brown, granular pigment. It represents local or systemic Iron excess.
- Staining Tip: On a standard H&E slide, it looks like brown granules. To prove it is iron, pathologists use the Prussian Blue Histochemical Stain (the iron turns bright blue).
Pathologic Calcification
Pathologic calcification is the abnormal deposition of calcium salts (phosphates, carbonates) in soft tissues, commonly due to injury or metabolic dysfunction.
Calcification is a permanent marker of past or present tissue injury. It occurs in two main forms: dystrophic (normal serum calcium, damaged tissue) and metastatic (high serum calcium, normal tissue).
I. Dystrophic Calcification (Local Injury)
Occurs in dead or dying tissues (necrosis) despite normal serum calcium levels, often seen in atherosclerosis, damaged heart valves, or tuberculous lymph nodes.
- Requirement: Occurs in non-viable (dead) or dying tissues.
- Calcium Levels: Serum calcium levels are Normal; there is no systemic mineral imbalance.
- Pathogenesis: Necrotic cells have damaged membranes; calcium binds to the phospholipids in these membranes, initiating the "crystallization" of calcium phosphate.
- Clinical Examples:
- Atherosclerosis: The core of an old plaque is often "bone-hard" due to calcification.
- Tuberculosis (TB): Areas of Caseous Necrosis often calcify, making them visible on X-rays (Ghon complex).
- Aging/Damaged Heart Valves: Leads to stenosis (narrowing of the valve).
II. Metastatic Calcification (Systemic Imbalance)
Occurs in normal tissues due to hypercalcemia (high calcium levels in the blood), often caused by renal failure, hyperparathyroidism, or Vitamin D intoxication.
- Requirement: Occurs in normal, healthy tissues.
- Calcium Levels: Always associated with Hypercalcemia (Elevated blood calcium).
- Hyperparathyroidism: Either a primary tumor of the parathyroid gland or "ectopic" secretion of PTH-related protein by cancers (like lung or breast cancer).
- Rapid Bone Destruction:
- Multiple Myeloma: A cancer of plasma cells that "eats" bone.
- Paget's Disease: Disorganized bone remodeling.
- Immobilization: Long-term bedrest leads to bone resorption.
- Vitamin D Disorders: Intoxication (overdose) or Sarcoidosis (where lung macrophages inappropriately activate Vitamin D).
- Renal Failure: Leads to phosphate retention, which triggers a secondary rise in PTH, pulling calcium out of the bones and into the tissues.
Morphology and Distribution
- Gross (Macroscopic): Calcium deposits are white, chalky granules. When a pathologist cuts through the tissue, it feels "gritty" (like cutting through sand or eggshells).
- Microscopic (Histology):
- Stains Basophilic (deep blue/purple) with H&E.
- Can be found inside cells (mitochondria) or outside cells in the matrix.
- Psammoma Bodies: In some tumors (like thyroid cancer), the calcification forms beautiful, laminated, sand-like concentric circles.
- Preferred "Metastatic" Targets: High-calcium levels prefer tissues that have an alkaline (basic) internal environment, which promotes salt precipitation. This includes:
- Gastric Mucosa (stomach lining).
- Kidneys (can lead to "nephrocalcinosis" or kidney stones).
- Lungs (alveolar walls).
- Systemic Arteries and Pulmonary Veins.
Acute Inflammation
Inflammation is the response of vascularized tissues that delivers leukocytes and host defense molecules from the circulation to the sites of infection and cell damage. Its primary objective is to eliminate the offending agent.
It is a protective response. Without it, infections remain unchecked, wounds fail to heal, and injured tissues become permanent festering sores.
- Dual Purpose:
- Destruction of the initial cause of injury (e.g., microbes, toxins).
- Management of the consequences of injury (e.g., necrotic cells and debris).
- The Mediators of Defense:
- Phagocytic Leukocytes: Cells that eat and digest foreign matter.
- Antibodies: Proteins that identify and neutralize targets.
- Complement Proteins: A system of plasma proteins that punch holes in bacterial membranes.
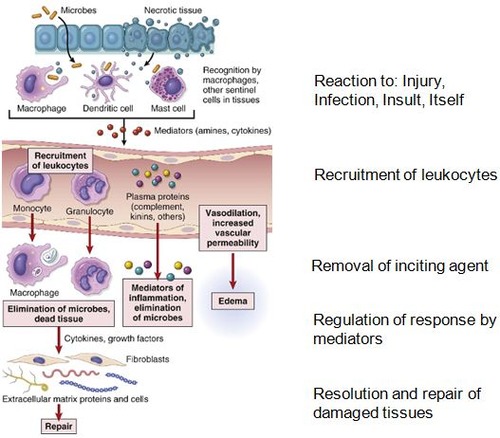
The Sequence of an Inflammatory Reaction
An inflammatory response follows a specific, step-by-step biological "protocol":
- Recognition: Receptors on host cells identify the noxious agent (the initiating stimulus).
- Recruitment: Leukocytes and plasma proteins move from the blood into the extravascular tissues.
- Removal: Phagocytic cells ingest and destroy microbes and dead cells.
- Regulation: The body activates control mechanisms to terminate the response once the threat is gone.
- Repair: A series of events (regeneration or scarring) heals the damaged tissue.
Comparison: Acute vs. Chronic Inflammation
| Feature | Acute Inflammation | Chronic Inflammation |
|---|---|---|
| Onset | Fast: Seconds, minutes, or hours. | Slow: Days to weeks. |
| Duration | Short: Minutes to a few days. | Long: Weeks, months, or years. |
| Cellular Infiltrate | Mainly Neutrophils. | Monocytes, Macrophages, and Lymphocytes. |
| Tissue Injury | Mild and self-limited. | Severe and progressive. |
| Fibrosis (Scarring) | Absent or minimal. | Prominent and permanent. |
| Signs | Prominent: Redness, heat, swelling, pain. | Subtle: Less obvious local signs. |
Diseases Caused by Inflammatory Reactions
When inflammation is misdirected or overactive, it causes specific clinical disorders:
(Neutrophil/Antibody-Driven)
- Acute Respiratory Distress Syndrome (ARDS): Neutrophils damage the alveolar-capillary membrane in the lungs.
- Asthma: Driven by Eosinophils and IgE antibodies, causing bronchial constriction.
- Glomerulonephritis: Antibodies and Complement proteins attack the kidney's filtration units.
- Septic Shock: An explosion of Cytokines leads to systemic vasodilation and organ failure.
(Macrophage/Lymphocyte-Driven)
- Arthritis: Lymphocytes and macrophages destroy joint cartilage.
- Atherosclerosis: Macrophages and lymphocytes drive the formation of plaques in arteries.
- Pulmonary Fibrosis: Macrophages and Fibroblasts replace lung tissue with thick scar tissue.
The 5 Cardinal Signs of Inflammation
- Rubor (Redness): Caused by Hyperemia (increased blood flow).
- Calor (Warmth): Caused by heat from the increased blood flow.
- Dolor (Pain): Caused by the release of chemical mediators (prostaglandins) and pressure on nerve endings.
- Tumor (Swelling): Caused by Edema (fluid accumulation).
- Functio Laesa (Loss of Function): Resulting from the combination of pain and swelling.
Component 1: Vascular Changes (The Fluid Response)
Acute inflammation has three major vascular components:
- Alteration in Vascular Caliber: Vasodilation increases blood flow to the area.
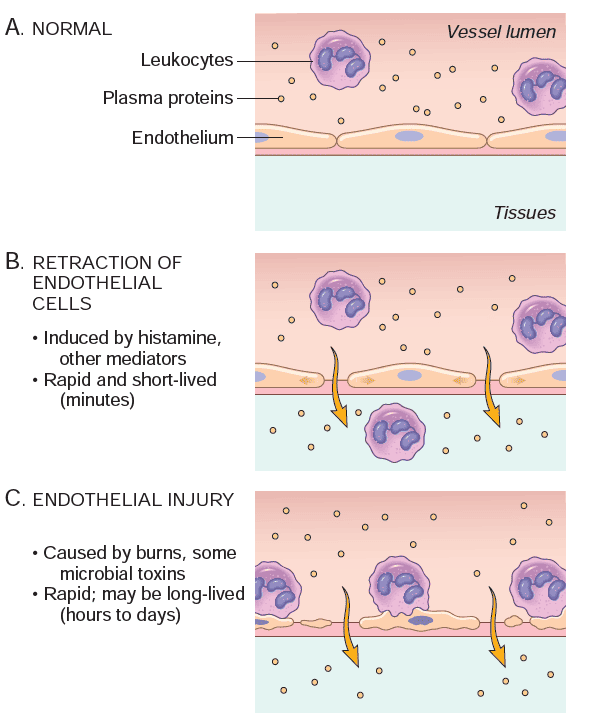
- Structural Changes: The microvasculature becomes "leaky," allowing plasma proteins and leukocytes to leave the blood.
- Leukocyte Emigration: Cells accumulate at the focus of injury to eliminate the agent.
Changes in Flow and Caliber
- Vasodilation: This is the earliest manifestation. It is induced by mediators like Histamine acting on vascular smooth muscle.
- Increased Permeability: Protein-rich fluid pours into the extravascular tissues.
- Stasis: As fluid leaves the vessels, blood flow slows. Red blood cells become concentrated and "packed," leading to engorgement of small vessels.
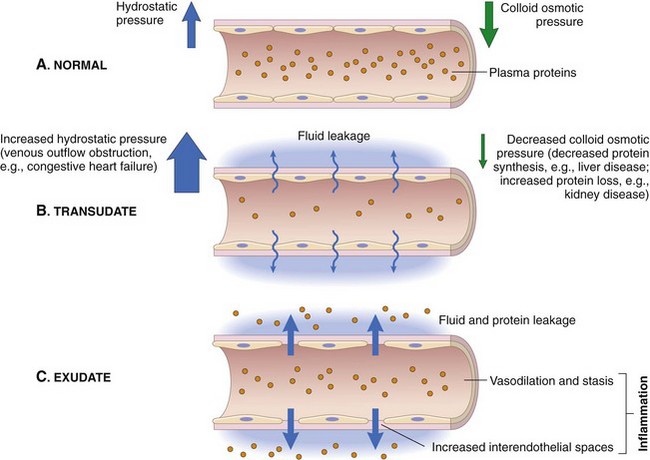
Understanding the Fluid (Edema)
- Exudation: The escape of fluid, proteins, and blood cells into the interstitial tissue.
- Exudate: A fluid with high protein concentration, cellular debris, and high specific gravity (>1.020). Indicates an increase in vascular permeability.
- Transudate: A fluid with low protein concentration, little cellular material, and low specific gravity (<1.012). It is an ultrafiltrate caused by osmotic/hydrostatic imbalance, not increased permeability.
- Pus (Purulent Exudate): An inflammatory exudate rich in neutrophils, dead cell debris, and microbes.
Component 2: The Lymphatic Response
- Drainage: Lymphatics act as a "filter" for extravascular fluids. In inflammation, lymph flow increases to drain the accumulating edema.
- Lymphangitis: Secondary inflammation of the lymphatic vessels (often seen as red streaks).
- Lymphadenitis: Inflammation of the draining lymph nodes (causing them to become swollen and painful).
Component 3: Leukocyte Recruitment (The Cellular Response)
Vascular endothelium in its normal state does not bind circulating cells. In inflammation, the endothelium is activated.
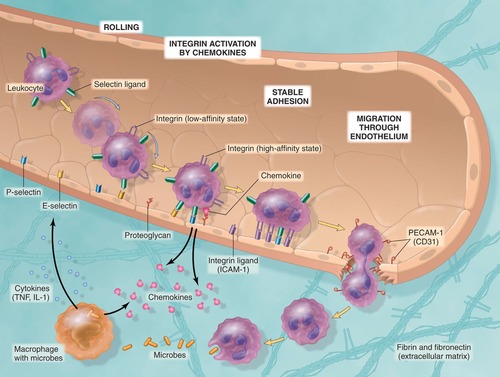
Step 1: In the Lumen (Margination, Rolling, and Adhesion)
- Margination: As blood flow slows (stasis), leukocytes leave the center of the vessel and move toward the endothelial wall.
- Rolling: Leukocytes "tumble" and bind transiently to the endothelium. This is mediated by the Selectin family of adhesion molecules.
- Adhesion: Leukocytes stop rolling and stick firmly to the vessel wall. This is mediated by Integrins.
Step 2: Migration Across the Endothelium
- Also known as Diapedesis or Transmigration. Leukocytes "squeeze" through the junctions between endothelial cells to enter the tissue.
Step 3: Chemotaxis
- Leukocytes follow a chemical "scent" toward the injury site.
- Chemotactic Stimuli: These include bacterial products, complement components (C5a), and cytokines (Chemokines).
Inflammatory Mediators
Mediators are substances that initiate or regulate inflammatory reactions. They are either cell-derived or plasma protein-derived.
- Vasoactive Amines: Histamine and Serotonin. These are stored in mast cells and platelets and cause immediate vasodilation and increased permeability.
- Lipid Products: Prostaglandins (cause pain and fever) and Leukotrienes (increase permeability and chemotaxis).
- Cytokines: Small proteins (like TNF and IL-1) that mediate the recruitment and activation of leukocytes.
- Complement Activation Products: Proteins (C3a, C5a) that increase vascular permeability and "coat" microbes for easier digestion (opsonization).
Morphologic Patterns & Systemic Effects of Acute Inflammation
This is the exhaustive, high-detail master set for the Morphologic Patterns and Systemic Effects of Acute Inflammation. Regardless of the specific pattern, every acute inflammatory reaction is defined by two fundamental microscopic features:
- Dilation of Small Blood Vessels: Resulting in increased blood volume at the site.
- Accumulation of Leukocytes and Fluid: The migration of cells and protein-rich fluid into the extravascular tissue (Interstitium).
Specific Morphologic Patterns
1. Serous Inflammation
- Defining Feature: The exudation of cell-poor fluid into spaces created by cell injury or into body cavities (Peritoneum, Pleura, Pericardium).
- Fluid Composition: The fluid does not contain microbes or large numbers of leukocytes.
- Sources of Fluid:
- Plasma: Leaking from blood vessels due to increased permeability.
- Mesothelial Cells: Secretions from the cells lining the body cavities.
- Clinical Terminology: The accumulation of this fluid in body cavities is termed an Effusion.
- Classic Example: A skin blister resulting from a burn or viral infection.
2. Fibrinous Inflammation
- Mechanism: When vascular permeability increases significantly, large molecules like Fibrinogen escape the blood. Once in the extravascular space, fibrinogen is converted into Fibrin, which is deposited.
- Stimulus: Occurs when vascular leaks are large or when there is a local procoagulant stimulus (e.g., cancer cells or certain bacteria).
- Location: Characteristically found in the linings of body cavities: Meninges (brain), Pericardium (heart), and Pleura (lungs).
- Histology: Fibrin appears as an eosinophilic (bright pink) meshwork of threads or an amorphous (shapeless) coagulum.
- Outcome: If the fibrin is not removed (dissolved by fibrinolysis), it leads to the ingrowth of fibroblasts and blood vessels, resulting in scarring (Adhesions).
3. Purulent (Suppurative) Inflammation & Abscess
- Defining Feature: The production of Pus.
- Pus Composition: A thick exudate containing Neutrophils, liquefied debris of necrotic cells, and edema fluid.
- Clinical Example: Acute Appendicitis is a common example of acute suppurative inflammation.
- Abscesses: These are localized collections of pus caused by suppuration buried deep within a tissue, an organ, or a confined space. They often require surgical drainage because they are "walled off" from the blood supply.
4. Ulcers
- Definition: A local defect or excavation of the surface of an organ or tissue.
- Mechanism: Produced by the sloughing (shedding) of inflamed, necrotic tissue.
- Requirement: Ulceration occurs only when tissue necrosis and inflammation exist on or near a surface.
- Common Sites:
- Mucosa: Mouth, stomach, intestines, or genitourinary tract.
- Skin/Subcutaneous Tissue: Particularly in the lower extremities of patients with vascular insufficiency (e.g., Diabetes, Sickle Cell Anemia, or Peripheral Vascular Disease).
Systemic Effects of Inflammation
Inflammation is not just local; it triggers the Acute-Phase Response throughout the body.
1. Fever
- Elevation: Temperature rises by 1–4° Celsius.
- Mediators: Induced specifically by IL-1 and TNF. These cytokines trigger the production of prostaglandins in the hypothalamus, resetting the body's "thermostat."
2. Acute-Phase Proteins
Plasma proteins synthesized in the liver increase rapidly during inflammation:
- C-reactive protein (CRP) & Fibrinogen: Synthesis is stimulated by the cytokine IL-6.
- Serum Amyloid A (SAA): Synthesis is stimulated by IL-1 or TNF.
- Note: Elevated fibrinogen causes red blood cells to stack (Rouleaux), increasing the Erythrocyte Sedimentation Rate (ESR), a common clinical test for inflammation.
3. Leukocytosis
- Definition: An increase in the white blood cell count in the blood.
- Trigger: Induced by bacterial infections.
- Leukemoid Reaction: When the count reaches extreme levels (15,000–20,000 cells/ml), mimicking leukemia.
- Mediators: Driven by TNF and IL-1, which accelerate the release of cells from the bone marrow.
4. Other Clinical Manifestations
- Circulatory: Increased pulse and blood pressure.
- Thermoregulation: Decreased sweating, Rigors (shivering), and Chills (seeking warmth).
- Constitutional: Anorexia (loss of appetite), Somnolence (excessive sleepiness), and Malaise (general feeling of being unwell).
Septic Shock: High Cytokine Levels
In severe infections (Sepsis), massive amounts of cytokines enter the blood, leading to a clinical triad known as Septic Shock:
- Disseminated Intravascular Coagulation (DIC): Widespread blood clotting that consumes all clotting factors, leading to hemorrhage.
- Hypotensive Shock: Extreme drop in blood pressure due to systemic vasodilation.
- Metabolic Disturbances: Including insulin resistance and Hyperglycemia (high blood sugar).
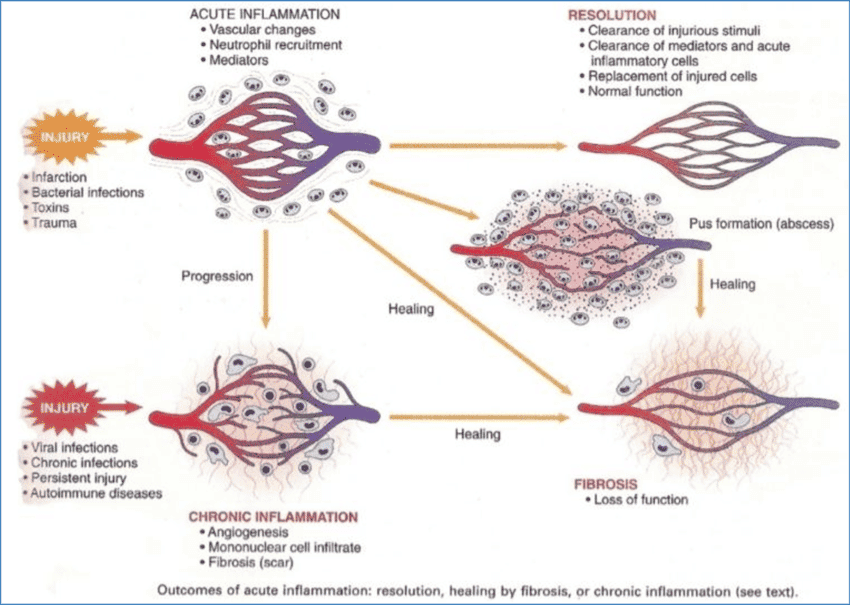
Outcomes of Acute Inflammation
Every acute inflammatory event ends in one of three ways:
- Complete Resolution: The injury is short-lived, there is little tissue destruction, and the tissue returns to its normal state.
- Healing by Connective Tissue Replacement: Occurs after substantial tissue destruction or in tissues that cannot regenerate. This results in Scarring or Fibrosis.
- Progression to Chronic Inflammation: Occurs when the offending agent is not removed or there is interference with the normal healing process.

Acute Inflammation & Accumulations Exam
Pathology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Acute Inflammation & Accumulations Exam
Pathology
Preparing questions...
Exam Completed!
See your performance breakdown below.