Chronic Inflammation
Chronic Inflammation
By the end of this detailed study guide, you should be able to clearly understand and explain:
- The precise definition of chronic inflammation and identify the key cellular mediators involved.
- The specific mechanisms and different types of granulomatous inflammation (foreign body vs. immune).
- The various examples of giant cells, distinguishing between those that occur naturally (physiological) and those that indicate disease (pathological).
Introduction to Chronic Inflammation
Defining Chronic Inflammation
Chronic inflammation is defined as a physiological response of prolonged duration—lasting for weeks or months (and sometimes even years). Unlike acute inflammation, which is a rapid, short-lived response characterized by fluid leakage and neutrophil infiltration, chronic inflammation is entirely different in its nature.
The defining characteristic of chronic inflammation is that three distinct processes occur simultaneously at the site of the lesion:
- Active Inflammation: The immune system continues to fight.
- Tissue Injury: The ongoing battle causes collateral damage to the host's own tissues.
- Attempts at Repair: The body simultaneously tries to heal the damage, usually through scarring (fibrosis) and the creation of new blood vessels.
Onset and Progression
Chronic inflammation does not always start the same way. It can initiate in two main ways:
- Following Acute Inflammation: If an initial acute inflammatory response fails to clear the offending agent (like a persistent bacterial infection), the body transitions the response into a chronic state.
- Insidious (Sneaky) Onset: It may begin as a low-grade, smoldering response from the very beginning. In these cases, there are absolutely no manifestations of a preceding acute reaction. The patient might not even realize it is happening until significant tissue damage has already occurred (e.g., in rheumatoid arthritis or atherosclerosis).
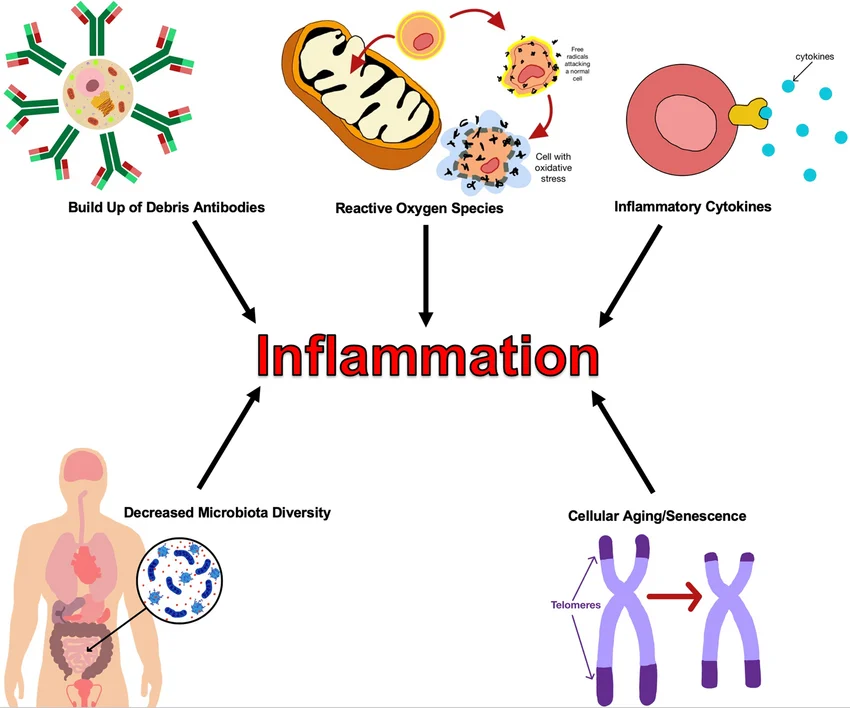
Causes of Chronic Inflammation
The immune system generally wants to clear a threat quickly and return to normal. Chronic inflammation only occurs when an offending agent is highly stubborn or when the immune system becomes misdirected.
A. Persistent Infections
Certain microorganisms are extremely difficult for the immune system to eradicate. They possess unique defense mechanisms that allow them to survive inside the body for long periods. Examples include:
- Mycobacteria: The bacteria responsible for Tuberculosis and Leprosy. They have waxy cell walls that resist being digested by immune cells.
- Treponema pallidum: The spiral-shaped bacterium that causes Syphilis.
- Certain Viruses, Fungi, and Parasites: These pathogens can hide within host cells or evade immune detection, leading to a constant, unresolved immune battle.
B. Immune-Mediated Inflammatory Diseases (Hypersensitivity)
Sometimes, the immune system inappropriately attacks the body's own tissues or harmless environmental substances. Because the eliciting antigens (the targets of the attack) cannot be eliminated—since they are part of the body itself or constantly present in the environment—these disorders tend to be chronic and highly intractable (hard to treat).
- Autoimmune diseases: Conditions like Rheumatoid Arthritis or Systemic Lupus Erythematosus.
- Allergic diseases: Chronic asthma is a prime example of continuous inflammation driven by environmental antigens.
C. Prolonged Exposure to Potentially Toxic Agents
Continuous exposure to toxic substances over a long period forces the body into a state of chronic inflammation. These agents can be from outside the body (exogenous) or inside the body (endogenous).
- Exogenous Materials (Outside the body): An example is inhaled particulate silica. When inhaled, silica dust cannot be degraded by the lungs' immune cells. The constant presence of these sharp, indestructible particles induces a chronic inflammatory response known as silicosis.
- Endogenous Agents (Inside the body): An example is chronically elevated plasma lipid components (high blood cholesterol). The persistent presence of toxic oxidized lipids in the blood vessel walls triggers a relentless inflammatory response that contributes heavily to the development of atherosclerosis (hardening of the arteries).
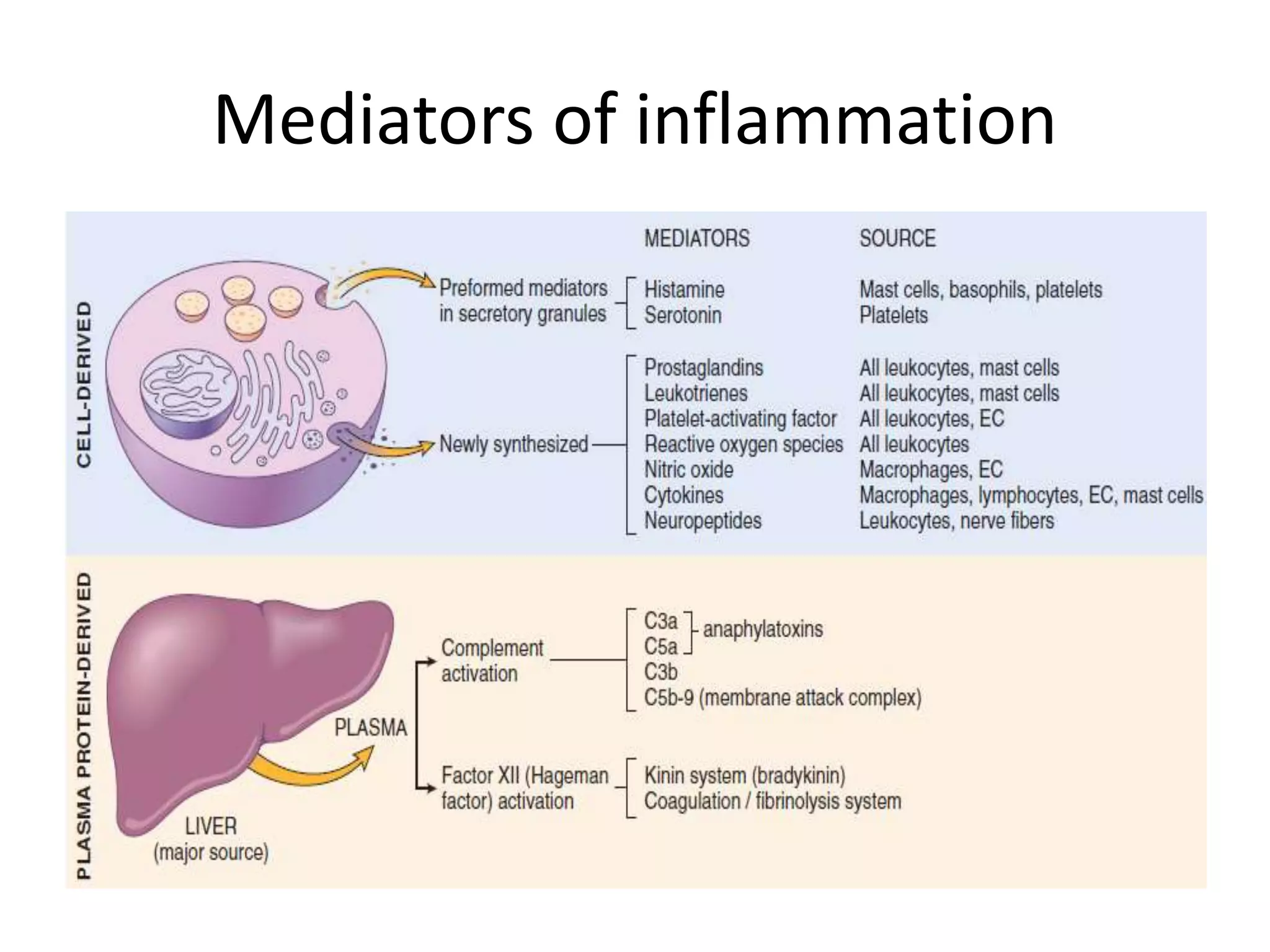
Morphologic Features (What it looks like under a microscope)
If a pathologist examines a tissue sample experiencing chronic inflammation under a microscope, they will observe three hallmark features that clearly distinguish it from acute inflammation.
- Infiltration with Mononuclear Cells: Acute inflammation is dominated by polymorphonuclear cells (neutrophils). In contrast, chronic inflammation is dominated by mononuclear cells—cells with a single, round, or un-lobed nucleus. These specifically include macrophages, lymphocytes, and plasma cells.
- Tissue Destruction: There is visible damage and destruction of the normal tissue architecture. This destruction is induced directly by the persistent offending agent (like a toxin or virus) or indirectly as collateral damage caused by the highly potent chemicals released by the accumulating inflammatory cells.
- Attempts at Healing: Because the tissue is being destroyed, the body desperately tries to patch the holes. This connective tissue replacement of damaged tissue is accomplished by two simultaneous processes:
- Angiogenesis: The sprouting and creation of new, fragile blood vessels to bring nutrients to the healing area.
- Fibrosis: The massive deposition of fibrous connective tissue (collagen) by fibroblasts, resulting in heavy scarring.
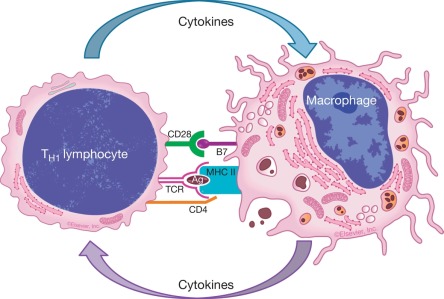
Cells and Mediators of Chronic Inflammation
The chronic inflammatory response is orchestrated by a specific cast of cellular characters. The major players include Macrophages, Lymphocytes, Plasma cells, Eosinophils, Neutrophils (in specific ongoing scenarios), and Mast cells.
A. Macrophages: The Dominant Cells
Macrophages are the undisputed heavyweights and dominant cells of chronic inflammation. They are large, highly capable tissue cells derived from circulating blood white blood cells called monocytes. After monocytes emigrate from the bloodstream into the tissues, they transform into macrophages.
Macrophages are normally diffusely scattered in most connective tissues throughout the body to act as local guards. Together, these cells comprise the mononuclear phagocyte system (older name: reticuloendothelial system). Depending on the organ they reside in, they are given special names:
- Kupffer cells: Found in the Liver.
- Sinus histiocytes: Found in the Spleen and Lymph nodes.
- Microglial cells: Found in the Central Nervous System (Brain and Spinal Cord).
- Alveolar macrophages: Found in the Lungs.
In all tissues, these cells act as essential filters (eating particulate matter, microbes, and dead/senescent cells) and as sentinels to alert the specific components of the adaptive immune system (T and B lymphocytes) to injurious stimuli.
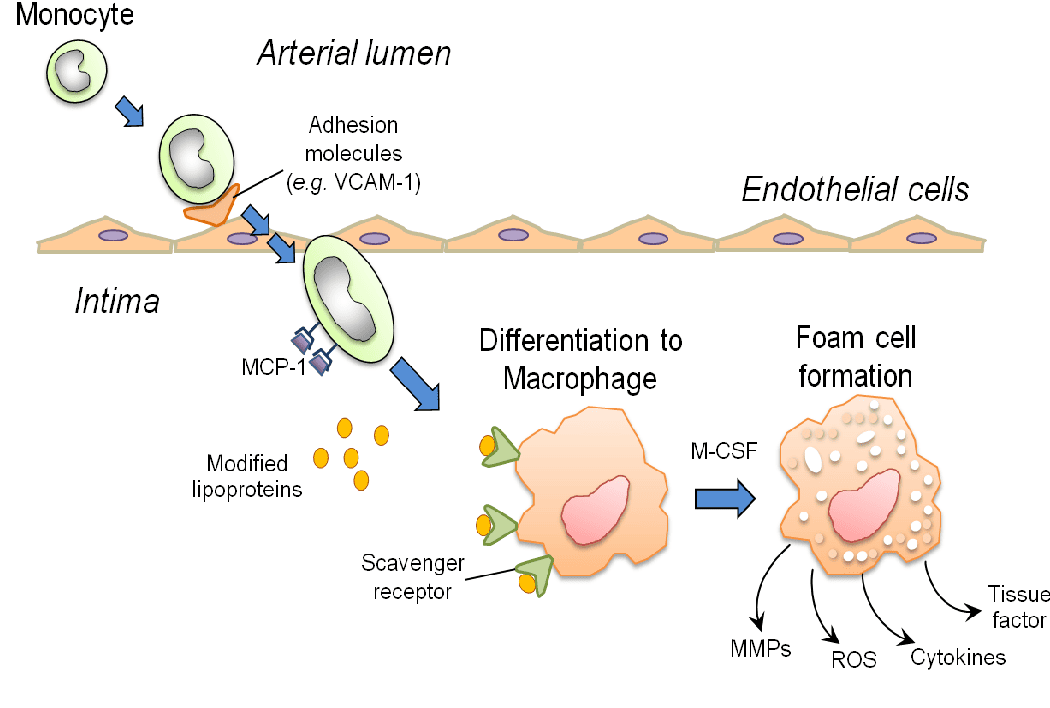
Lifecycle and Activation of Macrophages
- Migration: The half-life of circulating blood monocytes is very short, about 1 day. Under the influence of chemical signals (adhesion molecules and chemotactic factors), they begin to migrate to a site of injury within 24 to 48 hours after the onset of acute inflammation.
- Transformation: When monocytes reach the extravascular tissue, they undergo a transformation into larger macrophages. These new tissue macrophages have much longer half-lives and a significantly greater capacity for phagocytosis (eating debris) than their blood monocyte precursors.
- Activation: To fight tough infections, macrophages must become "activated." This results in an increased cell size, an increased content of deadly lysosomal enzymes, a more active metabolism, and a much greater ability to kill ingested organisms.
- Epithelioid Cells: By light microscopy, these activated macrophages appear large, flat, and pink (when stained with H&E). Because this appearance makes them look very similar to squamous epithelial cells (skin-like cells), these highly activated macrophages are sometimes called epithelioid cells.
Macrophage Activation Signals and Secreted Products
Macrophages do not activate themselves; they require specific activation signals, which include:
- Bacterial endotoxins and other microbial products.
- Cytokines secreted by sensitized T lymphocytes, in particular the highly potent cytokine IFN-γ (Interferon-gamma).
- Various mediators produced during the acute inflammation phase.
- Extracellular Matrix (ECM) proteins such as fibronectin.
Once activated, macrophages become secretory factories. They secrete a wide variety of biologically active products. If left unchecked, these very products cause the severe tissue injury and fibrosis characteristic of chronic inflammation. These products include:
- Acid and neutral proteases: Enzymes that literally digest and break down proteins and tissue matrix.
- Plasminogen activator and other enzymes: These greatly amplify the generation of further proinflammatory substances.
- ROS (Reactive Oxygen Species) and NO (Nitric Oxide): Highly toxic free radicals meant to destroy bacteria, but which also heavily damage host cells.
- Arachidonic Acid (AA) metabolites: Known as eicosanoids (prostaglandins and leukotrienes) that sustain inflammation.
- Cytokines: Specifically IL-1 (Interleukin-1) and TNF (Tumor Necrosis Factor), which recruit even more immune cells.
- Growth factors: These chemicals influence the proliferation of smooth muscle cells and fibroblasts, directly driving the overproduction of ECM (scar tissue).
The Fate of Macrophages
After the initiating stimulus is finally eliminated and the inflammatory reaction abates, macrophages eventually die off or wander away into the lymphatic vessels to be cleared. However, in chronic inflammatory sites, macrophage accumulation persists, and the macrophages can even proliferate directly at the site. This steady accumulation is maintained by a continuous release of lymphocyte-derived chemokines that recruit and immobilize them.
If the battle is incredibly difficult, IFN-γ can induce several individual macrophages to fuse together into massive, multi-nucleated super-cells called Giant Cells.
B. Eosinophils
Eosinophils are a specialized type of white blood cell characteristically found in inflammatory sites surrounding parasitic infections (like worms) or as part of immune reactions mediated by the IgE antibody (which is heavily associated with allergies and asthma).
- Their recruitment is driven by specific adhesion molecules and targeted chemokines, most notably eotaxin, which is derived from leukocytes or epithelial cells.
- Eosinophil granules are packed with a substance called Major Basic Protein. This is a highly charged cationic protein that is highly toxic to invading parasites, but tragically, it also causes severe necrosis (death) of the host's own epithelial cells.
C. Mast Cells
Mast cells act as sentinel (guard) cells widely distributed throughout the connective tissues of the body. They participate in both acute and chronic inflammatory responses.
- In atopic (allergy-prone) individuals, mast cells become "armed" with IgE antibodies that are specific to certain environmental antigens (like pollen or peanut protein).
- When these specific antigens are subsequently encountered, they bind to the IgE, triggering the mast cells to aggressively release histamines and Arachidonic Acid (AA) metabolites. These chemicals elicit massive vascular changes (vasodilation and leakiness).
- Because of this mechanism, IgE-armed mast cells are the central players in allergic reactions, up to and including fatal anaphylactic shock.
- Additionally, mast cells can elaborate cytokines such as TNF and chemokines, playing a beneficial role in fighting off some infections.
Granulomatous Inflammation
Granulomatous inflammation is a very specific, unique morphological pattern of chronic inflammation. It is a protective response essentially acting as a cellular quarantine.
Definition: It is characterized by the collection of highly activated macrophages that assume an epithelioid appearance, often surrounded by a collar of T lymphocytes, and sometimes featuring central tissue necrosis.
Purpose: Granuloma formation is a desperate cellular attempt to wall off and contain an offending agent that is extremely difficult or impossible to completely eradicate.
Types of Granulomas
Granulomas are broadly divided into two main categories based on what incited their creation:
- Foreign Body Granulomas:
- These are incited by completely inert (non-living, non-reactive) foreign bodies.
- They induce inflammation in the absence of a T-cell mediated immune response (because the material does not present proteins for the T-cells to react to).
- Causes include splinters, talc powder, large surgical sutures, or other indigestible fibers that are simply too large for a single macrophage to engulf via phagocytosis. The body walls them off instead.
- Immune Granulomas:
- These are caused by a variety of biological agents (like specific bacteria or fungi).
- These agents are fully capable of inducing a persistent, ongoing T-cell mediated immune response. The T-cells continuously release cytokines (like IFN-γ) that keep the macrophages gathered and highly activated.
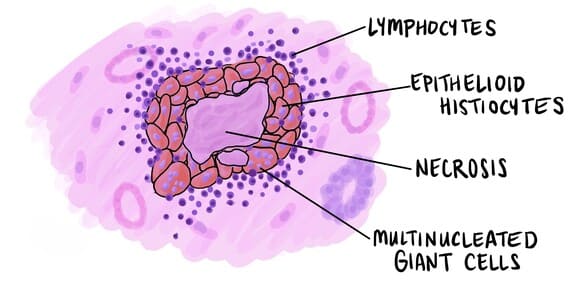
Morphology: Components of a Granuloma
A fully formed granuloma is a microscopic structure consisting of several distinct layers and components:
- Epithelioid cells: The core is packed with activated macrophages that have changed shape to look like epithelial cells.
- Multinucleated giant cells: Frequently, epithelioid cells fuse to form massive giant cells (such as Langhans’ giant cells).
- Lymphocytes: A surrounding ring or collar of T-cells that constantly secrete cytokines to maintain the structure.
- Fibroblasts: Cells on the outermost rim laying down collagen to physically wall off the structure.
- Caseous necrosis: In certain diseases (like Tuberculosis), the very center of the granuloma dies and turns into a cheese-like, amorphous mass known as caseous necrosis.
Causes and Examples of Granulomatous Inflammation
Many distinct agents can trigger this intense form of inflammation.
General Causes
- Bacterial: Tuberculosis (TB), Leprosy, Syphilis, Cat-scratch disease.
- Parasitic: Schistosomiasis, Leishmaniasis.
- Fungi: Histoplasmosis, Cryptococcosis, Coccidioides immitis.
- Inorganic Metals or Dusts: Silicosis (silica dust), Berylliosis (beryllium exposure).
- Foreign Body:
- Endogenous (From inside): Keratin, Uric acid crystals (causing Gout), necrotic bone fragments.
- Exogenous (From outside): Surgical sutures, splinters of wood.
- Drugs: Allopurinol, Sulphonamides.
- Unknown Etiology: Diseases where the body forms granulomas, but the exact trigger remains a medical mystery, such as Sarcoidosis and Crohn's disease.
Specific Disease Reactions (Detailed Breakdown)
| Disease | Cause | Tissue Reaction / Morphological Description |
|---|---|---|
| Tuberculosis | Mycobacterium tuberculosis | Characterized by a Caseating granuloma (referred to as a tubercle). It features a focus of activated macrophages (epithelioid cells) rimmed by fibroblasts, lymphocytes, and histiocytes. Occasional Langhans giant cells are present. The defining feature is central necrosis containing amorphous granular debris. Acid-fast bacilli may be found. |
| Leprosy | Mycobacterium leprae | Acid-fast bacilli are heavily present inside macrophages. Forms noncaseating granulomas (meaning the center does not undergo the cheese-like death seen in TB). |
| Syphilis | Treponema pallidum | Forms a specific lesion known as a Gumma. This is a microscopic to grossly visible lesion enclosing a wall of histiocytes and a plasma cell infiltrate. The central cells are necrotic but uniquely occur without the loss of cellular outlines. |
| Cat-scratch disease | Gram-negative bacillus | Forms a rounded or stellate (star-shaped) granuloma. It contains central granular debris and recognizable neutrophils. Giant cells are uncommon in this specific disease. |
| Sarcoidosis | Unknown etiology | Characterized heavily by Noncaseating granulomas filled with abundant activated macrophages. No central dead zone is present. |
| Crohn disease | Immune reaction against intestinal bacteria, possibly self-antigens | Occasional noncaseating granulomas found deeply embedded in the wall of the intestine, accompanied by a dense chronic inflammatory infiltrate. |
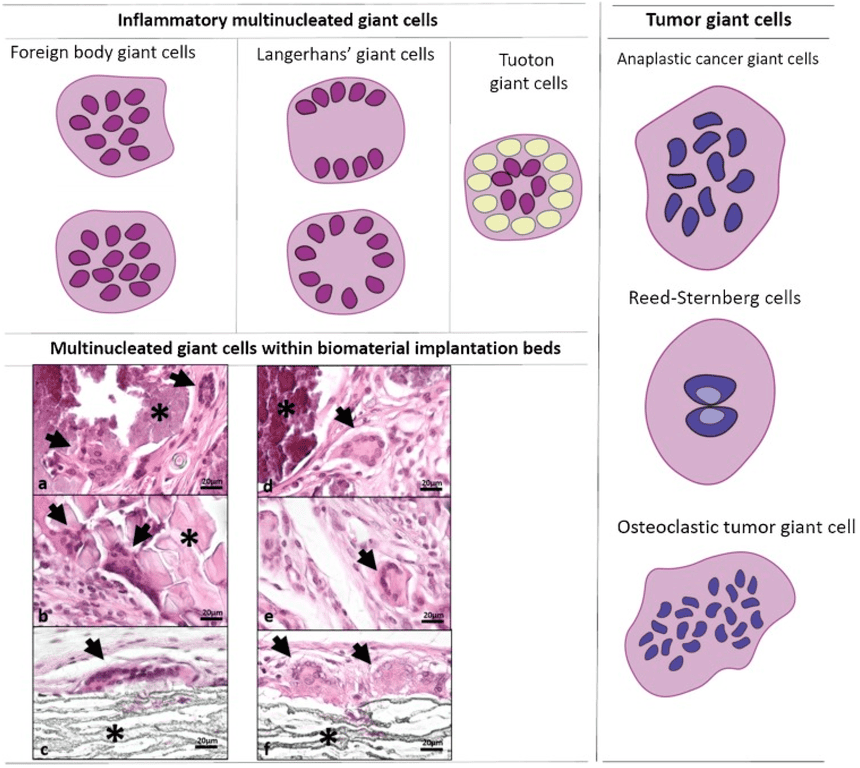
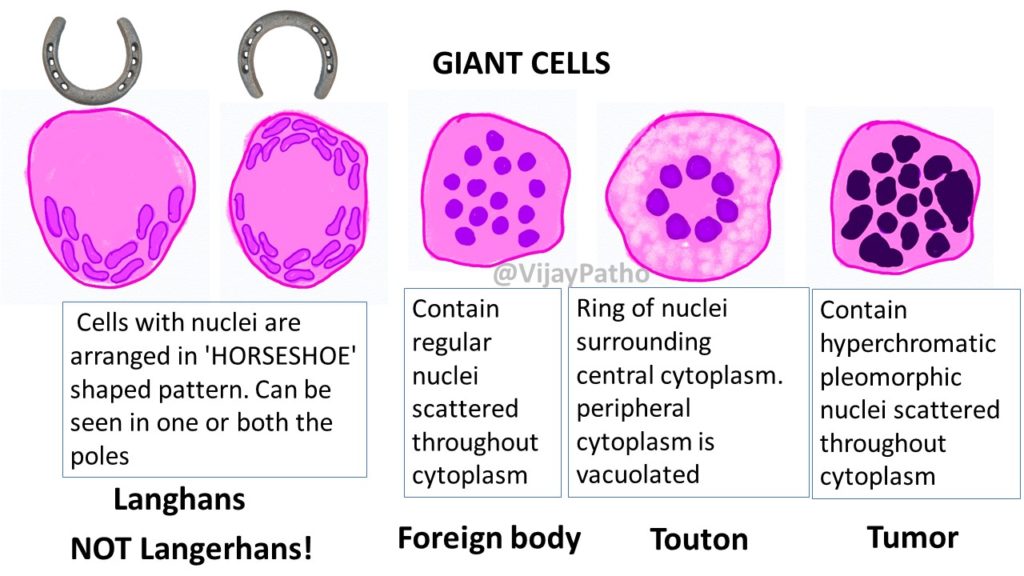
Types of Giant Cells
Giant cells are massive, multi-nucleated cells formed by the fusion of many individual cells. They are categorized based on whether they are part of normal, healthy bodily function (physiological) or the result of a disease process (pathological).
Normal Function
These cells naturally possess multiple nuclei to perform massive tasks for the body.
- Osteoclasts: Large cells responsible for the resorption (breaking down) of bone tissue during normal bone remodeling and growth.
- Megakaryocytes: Massive bone marrow cells responsible for the continuous production of blood platelets.
- Striated muscle cells: Skeletal muscle fibers are naturally formed by the fusion of many individual myoblasts, resulting in long, multi-nucleated fibers.
- Syncytiotrophoblast: The outer layer of the placenta that actively invades the uterine wall during pregnancy, forming a continuous multi-nucleated layer without cell boundaries.
Disease States
These are formed aberrantly due to chronic inflammation or cancer.
- Langhans’ giant cell: Characteristically seen in Tuberculosis (TB). The multiple nuclei are distinctively arranged in a horseshoe or circular pattern at the periphery of the cell membrane.
- Foreign body giant cell: Formed to digest foreign material (like surgical sutures). The nuclei are haphazardly clustered together in the center of the cell, without any distinct pattern.
- Touton giant cell: Characterized by a ring of nuclei surrounded by foamy, lipid-filled cytoplasm; commonly seen in lesions with high lipid content (xanthomas).
- Tumor giant cell: Highly irregular, monstrous cells with bizarre, massive nuclei found in highly malignant cancers.
- Warthin-Finkeldey giant cells: Specifically found in the hyperplastic lymph nodes of individuals infected with Measles and HIV.
- Aschoff body (Anitschkow cells): A specific type of enlarged, altered macrophage found within the heart muscle in patients suffering from Rheumatic fever.
- Reed-Sternberg cell: A massive, bi-nucleated or multi-nucleated malignant cell that famously resembles an "owl's face." It is the hallmark diagnostic cell of Hodgkin lymphoma.
Chronic Inflammation Quiz
Pathology
Enter your details to begin the examination.
🛡️ Privacy Note: Results are for tracking and certification purposes only.
Chronic Inflammation Quiz
Pathology
Preparing questions...
Exam Completed!
See your performance breakdown below.