Signaling Mechanisms
Signaling Mechanisms
How to Approach This Topic
Pharmacodynamics (how drugs act on the body) is all about communication. Cells are blind and deaf; they rely entirely on chemical messages. When you take a drug, it acts as a messenger. This entire lecture focuses on how the message gets from the outside of the cell to the inside, forcing the cell to change its behavior. We will break down every single pathway so you can easily understand
Introduction to Drug-Responsive Signaling Mechanisms
Before we examine the specific pathways, we must understand the basic sequence of events:
- An agonist drug (the messenger or "key") binds to its specific receptor (the "lock").
- This binding event directly activates an effector or signaling mechanism.
- The effector causes a biological change inside the cell.
There are several different categories of these signaling mechanisms known in pharmacology. We classify them based on where the receptor is located and how it translates the message.
The Major Categories of Signaling Mechanisms:
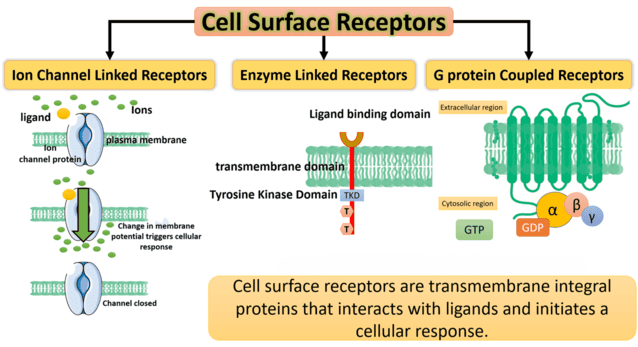
- Intracellular receptors: The receptor is hidden deep inside the cell (in the cytoplasm or nucleus).
- Membrane receptors directly coupled to ion channels: The receptor is on the surface and acts as a direct physical gate for ions.
- Receptors linked via coupling proteins to intracellular effectors: The receptor is on the surface and uses a middleman (like a G-protein) to send a message inside.
- Receptors that function as enzymes or transporters: The receptor itself performs a chemical reaction or moves molecules.
Intracellular Receptors
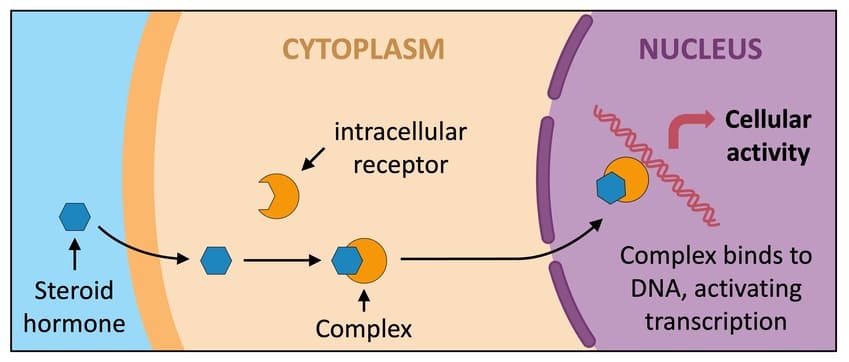
Most drugs stop at the cell surface. However, some drugs are highly lipid-soluble (fat-soluble), allowing them to melt right through the cell membrane and enter the interior of the cell. Once inside, they find intracellular receptors.
The Process Step-by-Step:
- The hormone or drug crosses the cell membrane.
- It binds to the intracellular receptor.
- This binding releases regulatory proteins (which were holding the receptor in an inactive state).
- The receptor is now activated. In many cases, two activated receptors will pair up and join together (a process called dimerization).
- This new hormone-receptor complex travels (translocates) directly into the cell's nucleus.
- Inside the nucleus, the complex physically attaches to specific regions of DNA called response elements in spacer DNA.
- This interaction forces the DNA to either increase or decrease gene expression (the manufacturing of new proteins).
Think of the cell like a factory. Most drugs are delivery drivers who drop a package at the front desk (membrane receptors). But intracellular drugs are like VIP executives. They walk right past the front door, go straight into the manager's office (the nucleus), and rewrite the factory's rulebook (DNA) to change what the factory produces.
Key Characteristics and Examples
Because these drugs require the cell to read DNA and build entirely new proteins from scratch, the pharmacologic responses elicited via modification of gene expression have two absolute rules:
- They are slower in onset (it takes hours to days to build new proteins).
- They are longer in duration than many other drugs (even after the drug leaves the body, the newly built proteins stick around and keep working for days).
Examples of ligands that use Intracellular Receptors:
- Steroids (Glucocorticoids): Drugs interacting with glucocorticoid receptors lead to the gene expression of proteins that heavily inhibit the production of inflammatory mediators.
- Thyroid hormones.
- Gonadal steroids (Estrogen, Testosterone).
- Vitamin D.
Clinical Scenario: Asthma Attack
If a patient arrives at the hospital having a severe asthma attack, giving them an inhaled steroid (an intracellular drug) will not save them immediately because steroids take hours to change gene expression and reduce inflammation. Instead, you must give them Albuterol (a fast-acting membrane receptor drug) to open the airways instantly. The steroid is given to prevent attacks over the next few days.
Membrane Receptors Directly Coupled to Ion Channels
These are the fastest receptors in the human body. They work in milliseconds. The receptor and the effector are the exact same physical structure.
Mechanism: Endogenous ligands (the body's natural chemicals) regulate the flow of ions through excitable membranes by activating receptors that are directly coupled to ion channels. There are no second messengers involved. It is a simple gate. Many drugs act by either mimicking (agonist) or antagonizing (blocker) the actions of these natural ligands.
The Nicotinic Receptor
- Ligand: Acetylcholine (ACh).
- Ion Channel: Coupled directly to a Sodium/Potassium (Na+/K+) ion channel.
- Locations: Present in the Autonomic Nervous System (ANS) ganglia, the skeletal myoneural junction (where nerves tell muscles to move), and the Central Nervous System (CNS).
- Pharmacology: This receptor is a prime target for many drugs, including:
- Nicotine (acts as an agonist).
- Choline esters.
- Ganglion blockers.
- Skeletal muscle relaxants (used during surgery to paralyze muscles by blocking this receptor).
The GABA-A Receptor
- Ligand: Gamma-aminobutyric acid (GABA).
- Ion Channel: Coupled directly to a Chloride (Cl-) ion channel. When chloride flows into a nerve cell, it makes the cell highly negative and puts it to sleep (inhibition).
- Locations: Central Nervous System (CNS).
- Pharmacology: This receptor can be heavily modulated (enhanced) by drugs that calm the brain down:
- Anticonvulsants (anti-seizure medications).
- Benzodiazepines (anti-anxiety medications like Valium or Xanax).
- Barbiturates (heavy sedatives).
Receptors Linked Via Coupling Proteins (G-Proteins)
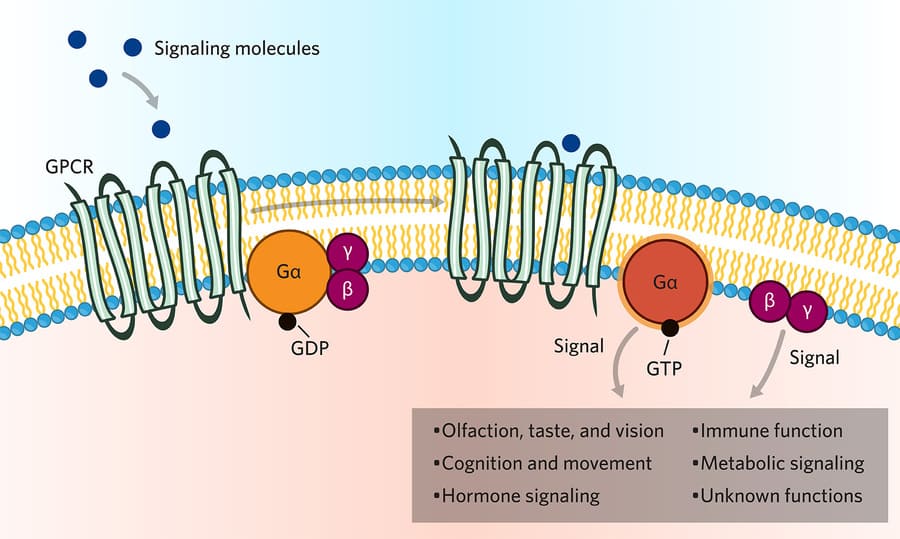
This is the largest and most famous family of receptors in pharmacology. They are often called "serpentine" receptors because they are made of a single protein chain that snakes back and forth across the cell membrane exactly seven times (seven transmembrane spanning domains).
The third loop on the inside of the cell is physically coupled to the G-protein effector mechanism. The G-protein is a middleman. It binds GTP (Guanosine Triphosphate) to become active.
The Relay Race Analogy:
- The Drug (Runner 1) passes the baton to the Receptor on the outside.
- The Receptor passes the baton to the G-Protein (Runner 2) on the inside.
- The G-Protein passes the baton to an Enzyme (Runner 3).
- The Enzyme creates Second Messengers (Runner 4) which flood the cell and finish the race.
There are three main types of G-proteins you must memorize: Gs, Gi, and Gq.
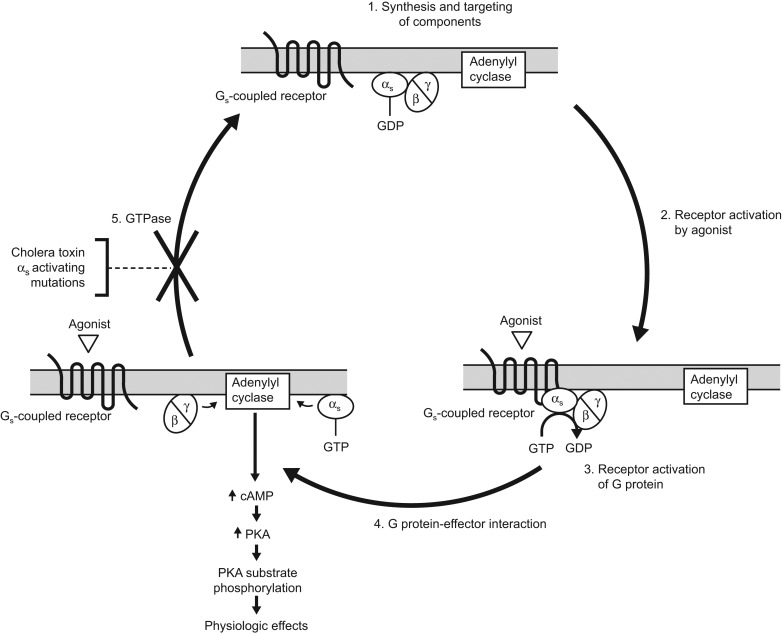
A. The Gs Pathway (Stimulatory)
Mechanism: Agonists binding to Gs proteins turn ON an enzyme called Adenylyl Cyclase. This enzyme takes ATP and converts it into a massive amount of the second messenger cAMP (cyclic AMP). The cAMP then activates Protein Kinase A (PKA). PKA serves to phosphorylate (add energetic phosphate groups to) tissue-specific substrate enzymes or transcription factors like CREB, profoundly affecting their cellular activity.
Receptors linked to Gs (Increases cAMP):
- Beta receptors (catecholamines like epinephrine).
- Dopamine (D1).
- Glucagon.
- Histamine (H2) (found in the stomach, controls stomach acid).
- Prostacyclin.
- Some serotonin subtypes.
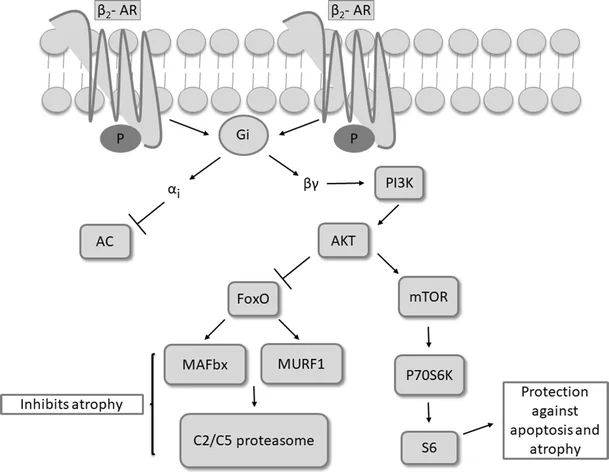
B. The Gi Pathway (Inhibitory)
Mechanism: Agonists binding to Gi proteins do the exact opposite. They inhibit Adenylyl Cyclase, which severely decreases cAMP production, calming the cell down.
Receptors linked to Gi (Decreases cAMP):
- Alpha-2 adrenoreceptors.
- ACh (M2) (Muscarinic 2 receptors, found on the heart to slow heart rate).
- Dopamine (D2 subtypes).
- Several opioid receptors (this is how morphine stops pain signaling).
- Several serotonin subtypes.
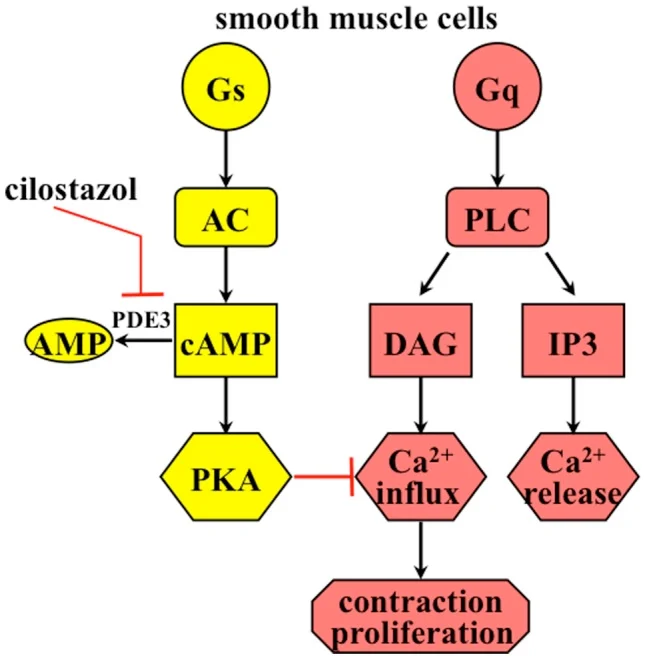
C. The Gq Pathway (The Calcium Pathway)
Mechanism: This pathway uses entirely different enzymes and messengers. Other receptor systems are coupled via Gq proteins, which activate an enzyme called Phospholipase C (PLC).
- PLC chops up a membrane phospholipid called PIP2 (phosphatidylinositol bisphosphate).
- This chopping releases TWO distinct second messengers: IP3 (inositol triphosphate) and DAG (diacylglycerol).
- IP3 travels deep into the cell and induces the massive release of stored Calcium (Ca2+) from the Sarcoplasmic Reticulum (SR).
- The newly released Calcium, working closely together with the DAG, activates an entirely different kinase: Protein Kinase C (PKC).
- Protein Kinase C serves to phosphorylate a unique set of tissue-specific substrate enzymes that are normally not phosphorylated by Protein Kinase A.
Receptors linked to Gq (Increases Calcium):
- ACh (M1 and M3) (Muscarinic receptors that make glands secrete and gut muscles contract).
- Norepinephrine (Alpha-1) (Causes blood vessels to severely constrict).
- Angiotensin II.
- Several serotonin subtypes.
Summary Rule to Memorize for Exams: "In A Nutshell"
- Gq Activation (Phospholipase C): M1, M3, Alpha-1
- Gi Inhibition (Adenylyl Cyclase): M2, Alpha-2, D2
- Gs Activation (Adenylyl Cyclase): Beta-1, Beta-2, D1
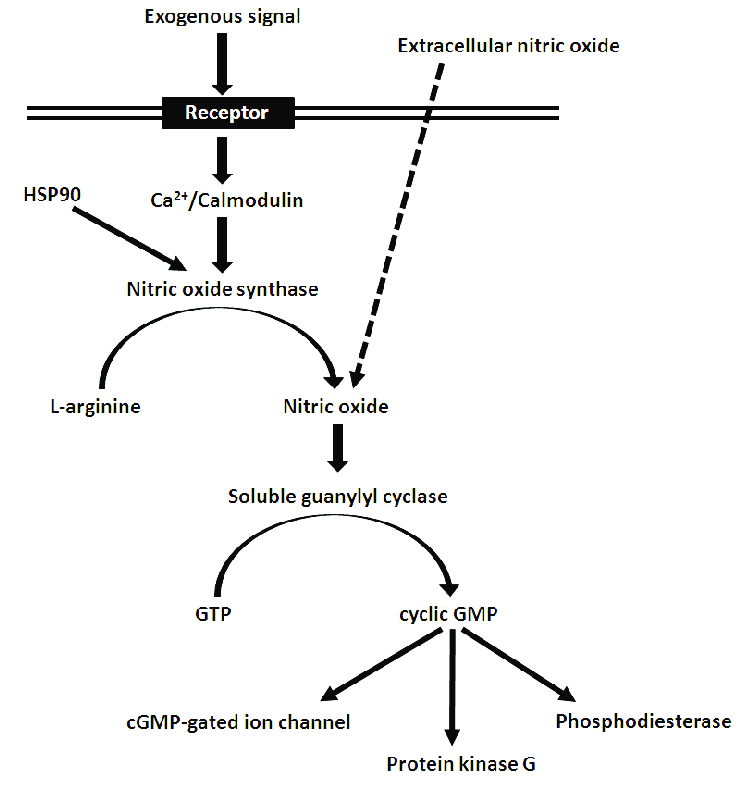
Cyclic GMP and Nitric Oxide Signaling
This is a very unique signaling mechanism that relies on a gas: Nitric Oxide (NO).
The Mechanism Step-by-Step:
- Nitric oxide (NO) is naturally synthesized inside the endothelial cells (the inner lining of your blood vessels).
- Because NO is a gas, it easily diffuses right out of the endothelial cell and straight into the neighboring vascular smooth muscle cell.
- Inside the smooth muscle, NO directly activates an enzyme called Guanylyl Cyclase.
- This heavily increases the production of the second messenger cGMP (cyclic GMP) inside the smooth muscle.
- cGMP facilitates the dephosphorylation of myosin light chains.
- By removing the phosphate from myosin, it prevents the myosin from interacting with actin. Without actin-myosin interaction, the muscle cannot contract.
- The ultimate result is intense vasodilation (widening and relaxation of the blood vessels).
Clinical Correlate: Vasodilators
Any drug that acts as a vasodilator works by increasing the synthesis of NO by endothelial cells or providing NO directly.
Drugs acting via NO include:
- Nitrates (e.g., nitroglycerin): Used by heart patients. It breaks down directly into NO gas in the blood, causing instant vasodilation to relieve chest pain (angina).
- M-receptor agonists (e.g., bethanechol): Stimulate the endothelium to produce more NO.
Endogenous body compounds acting via NO include:
- Bradykinin.
- Histamine.
Receptors That Function as Enzymes or Transporters
Not all receptors are communication dishes; some are hardworking machineries. Many drugs act simply by inhibiting these natural enzymes or transport pumps.
A. Enzyme Inhibitors
There are multiple examples of drug action that depend directly on enzyme inhibition. If you block the enzyme, you stop its chemical reaction. Key enzymes targeted by drugs include:
- Acetylcholinesterase (blocking this leaves more ACh in the brain).
- Angiotensin Converting Enzyme (ACE) (ACE inhibitors lower blood pressure).
- Aspartate protease (targeted by HIV medications).
- Carbonic anhydrase (diuretics for the kidney).
- Cyclooxygenases (COX) (Aspirin and Ibuprofen block COX to stop pain and inflammation).
- Dihydrofolate reductase (Chemotherapy targets this to stop cell division).
- DNA/RNA polymerases (Antiviral and cancer drugs).
- Monoamine oxidases (MAO) (MAO inhibitors are powerful antidepressants).
- Na/K-ATPase (Targeted by digoxin for heart failure).
- Neuraminidase (Targeted by Tamiflu to stop the flu virus).
- Reverse transcriptase (Targeted by HIV medications).
Tyrosine Kinase (TK) Inhibitors for Cancer
Many cancers are driven by overactive Tyrosine Kinase enzymes. We treat them with specific inhibitors:
- Imatinib: This is a highly specific tyrosine kinase inhibitor (it acts like a sniper, hitting only the mutated enzyme in chronic myeloid leukemia).
- Sorafenib: This is a non-specific TK inhibitor (it acts more broadly, hitting multiple pathways in kidney and liver cancers).
B. Transporter Inhibitors
Nerve cells communicate by releasing neurotransmitters into a gap, and then using "transporter pumps" to vacuum them back up (reuptake). Drugs can block these pumps to keep the neurotransmitter active longer in the gap.
Examples of drug action on transporter systems include the inhibitors of reuptake for several neurotransmitters, including:
- Dopamine (Cocaine blocks this pump).
- GABA.
- Norepinephrine.
- And serotonin (SSRIs like Prozac block serotonin reuptake to treat depression).
Receptors That Function as Transmembrane Enzymes
These are large proteins where the outside part is a receptor, and the inside part is literally an enzyme. They mediate the highly important first steps in signaling by insulin and growth factors.
Examples include:
- Epidermal growth factor (EGF).
- Platelet-derived growth factor (PDGF).
The Mechanism Step-by-Step:
- These are membrane-spanning macromolecules.
- They have a recognition site for the binding of insulin or growth factors located externally on the cell surface.
- They have a cytoplasmic domain located internally that normally functions as a tyrosine kinase.
- Binding of the ligand on the outside causes massive conformational (shape) changes in the protein.
- This causes two receptors to slide together and pair up (dimerization).
- Dimerization causes the internal tyrosine kinase domains to officially become activated.
- Ultimately, this leads to the phosphorylation of tissue-specific substrate proteins, forcing the cell to grow, divide, or absorb glucose (in the case of insulin).
Variation: Guanylyl Cyclase-Associated Receptors
Similar to the transmembrane enzymes above, some membrane receptors do not have a tyrosine kinase inside, but rather a Guanylyl Cyclase enzyme attached to them.
For example, stimulation of receptors by Atrial Natriuretic Peptide (ANP) directly activates the attached guanylyl cyclase, which causes an immediate increase in intracellular cyclic GMP (cGMP). This causes the kidneys to excrete sodium and water, lowering blood pressure.
Receptors for Cytokines (The JAK-STAT Pathway)
Cytokines are immune system messengers and growth modulators. They require their own special receptor family.
Ligands that use this system include:
- Erythropoietin (tells the bone marrow to make red blood cells).
- Somatotropin (Growth Hormone).
- Interferons (powerful immune chemicals).
The Mechanism Step-by-Step:
- Their receptors are membrane-spanning. Unlike the receptors above, they do not have built-in enzyme activity.
- Upon activation by a cytokine, they physically attach to and activate a distinctive set of completely separate cytoplasmic tyrosine kinases known as Janus kinases (JAKs).
- The activated JAKs act quickly to phosphorylate special molecules called Signal Transducers and Activators of Transcription (STAT) molecules.
- Once phosphorylated, two STATs will pair up (dimerize).
- The dimerized STATs then dissociate from the receptor complex.
- They physically cross the nuclear membrane to enter the nucleus.
- Inside the nucleus, they modulate gene transcription (changing protein production).
The cytokine receptor is a coach who tags in a wrestler (JAK). The wrestler (JAK) beats up and tags a runner (STAT). The runner pairs up with his partner and runs straight into the boss's office (the nucleus) to hand in the final paperwork.