Pancreas Physiology
THE PANCREAS
The pancreas is an elongated, flattened gland located deep in the abdomen, nestled between the stomach and the spine. It is a unique organ because it serves two vital functions:
- Exocrine Function: The vast majority (about 98-99%) of the pancreas is dedicated to its exocrine role. It produces digestive enzymes (e.g., amylase, lipase, proteases) that are secreted into the duodenum via a system of ducts to aid in the digestion of carbohydrates, fats, and proteins in the small intestine.
- Endocrine Function: The remaining 1-2% of the pancreas is composed of specialized clusters of cells called the islets of Langerhans. These islets are hormone-producing factories that secrete critical hormones directly into the bloodstream to regulate blood glucose levels.
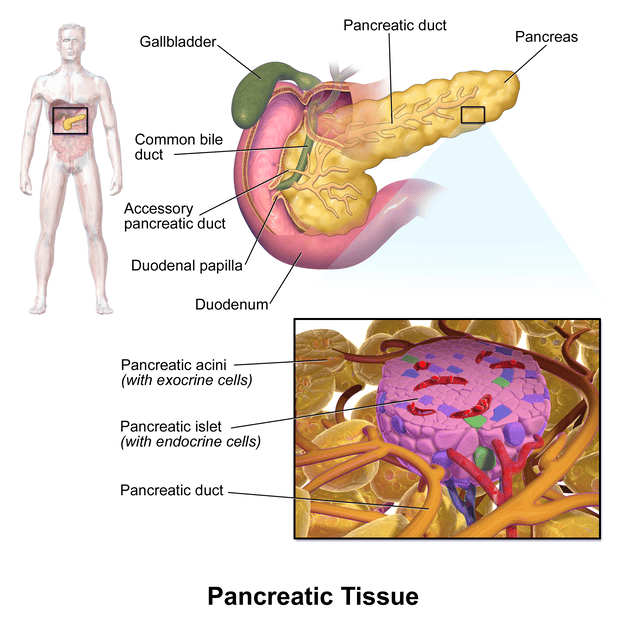
I. ENDOCRINE FUNCTION OF THE PANCREAS: THE ISLETS OF LANGERHANS
The endocrine pancreas is composed of approximately 1 to 2 million islets of Langerhans, which collectively constitute only about 1-2% of the total pancreatic mass. Despite their small proportion, these islets are vital for maintaining metabolic homeostasis, especially blood glucose regulation.
Structure of the Islets:
- Each islet is a microscopic cluster of endocrine cells, typically 0.3 mm in diameter.
- They are highly vascularized and organized around small capillaries into which their cells directly secrete their hormones, allowing for rapid systemic distribution.
- The cells within each islet are in close proximity to each other, facilitating paracrine communication (local hormone action between neighboring cells).
Major Cell Types within the Islets:
The islets contain several distinct cell types, each producing specific hormones. These cells are distinguished by their morphological and staining characteristics.
1. Alpha (α) cells
- Constitute about 25% of the islet cells.
- Typically located at the periphery of the islets.
- Their primary secretion is glucagon, often referred to as the "fasting hormone."
2. Beta (β) cells
- Are the most abundant, making up about 60% of the islet cells.
- Predominantly located in the center of the islets.
- Their primary secretions are insulin (the "feasting hormone") and amylin. Amylin is co-secreted with insulin and helps regulate glucose homeostasis by slowing gastric emptying and promoting satiety.
3. Delta (δ) cells
- Comprise about 10% of the islet cells.
- Distributed around the periphery of the islets.
- They secrete somatostatin, a hormone that acts locally (paracrine effect) to inhibit the secretion of both insulin and glucagon, thus modulating their release. It also has systemic effects, inhibiting growth hormone and TSH from the pituitary.
4. PP cells (or Gamma (γ) cells)
- Present in small numbers (less than 1%).
- They secrete pancreatic polypeptide, whose exact physiological role is still not entirely clear but is thought to be involved in regulating pancreatic secretion and satiety.
II. BLOOD GLUCOSE HOMEOSTASIS:
The primary role of the endocrine pancreas is to maintain blood glucose homoeostasis (or homeokinesis), ensuring that blood glucose levels remain within a narrow, healthy range despite variations in food intake and energy expenditure. This delicate balance is achieved through the coordinated actions of insulin and glucagon.
Outline of Glucose Regulation:
- The "Feasting" Hormone - Insulin: Primarily secreted after meals, when blood glucose is high, to promote glucose uptake and storage.
- The "Fasting" Hormone - Glucagon: Primarily secreted during fasting or between meals, when blood glucose is low, to raise blood glucose levels.
- Integrated Hormonal Control of Blood Glucose Concentration: The interplay between insulin, glucagon, and other hormones (like somatostatin, amylin, and gut hormones) orchestrates the precise regulation of glucose.
- Diabetes, Pathophysiological Changes: Disruptions in this hormonal control, particularly insulin function, lead to diabetes mellitus, a chronic metabolic disorder.
III. INSULIN AND ITS METABOLIC EFFECTS
Insulin is the most important anabolic hormone in the body, primarily responsible for lowering blood glucose levels after a meal and promoting energy storage.
Structure and Synthesis:
- Insulin is a small protein hormone (peptide hormone) with a molecular weight of 5808.
- It consists of two peptide chains (A and B chains) linked together by two disulfide bonds. This specific structure is crucial for its biological activity.
- It is secreted by the beta (β) cells of the pancreatic islets via exocytosis, primarily in response to elevated blood glucose levels (a signal of "energy abundance").
Half-Life and Degradation:
- The "half-life" of insulin in circulation is remarkably short, about 6 minutes, meaning half of the secreted insulin is cleared from the bloodstream within this time.
- Consequently, it is cleared completely from the blood in approximately 10-15 minutes. This rapid turnover allows for precise and dynamic control of blood glucose levels.
- Insulin is primarily degraded by the enzyme insulinase, which is found abundantly in the liver and kidneys, and to a lesser extent in muscles and most other tissues.
Role in Blood Glucose Maintenance:
- Insulin maintains blood glucose levels close to 5 mmol/L (90 mg/dL) under fasting conditions.
- After a meal, blood glucose levels may temporarily rise as high as 8 mmol/L (144 mg/dL), but insulin rapidly brings these levels back down to the normal range.
- Overweight and obese individuals often exhibit higher basal levels of insulin (hyperinsulinemia) as a compensatory mechanism to overcome insulin resistance, where their cells do not respond as effectively to insulin.
IV. SYNTHESIS OF INSULIN:
Insulin synthesis is a classic example of protein synthesis for secretion, involving several steps within the β-cells:
- Preproinsulin Formation: Insulin RNA, transcribed from the insulin gene, is translated by ribosomes attached to the endoplasmic reticulum (ER). The initial product is a larger precursor molecule called preproinsulin (molecular weight = 11,500). This molecule contains a signal peptide that directs it into the ER lumen.
- Proinsulin Formation: Inside the ER, the signal peptide is cleaved off, converting preproinsulin into proinsulin (molecular weight = 9,000). Proinsulin consists of the A and B chains linked by a C-peptide.
- Insulin and C-peptide Cleavage: Proinsulin then travels to the Golgi apparatus. Here, specific enzymes (prohormone convertases) cleave the proinsulin molecule, removing the C-peptide and leaving behind the mature, active insulin molecule (A and B chains linked by disulfide bonds) and the peptide fragments (C-peptide).
- Packaging and Secretion: Both insulin and C-peptide are then packaged together into secretory granules. These granules await the appropriate signal (primarily elevated blood glucose) to be released into the bloodstream via exocytosis.
V. INSULIN AND TARGET RECEPTORS: MECHANISM OF ACTION
Insulin exerts its diverse metabolic effects by binding to specific receptors on the surface of target cells.
Insulin Receptor Structure:
- The insulin receptor is a large, complex transmembrane glycoprotein with a molecular weight of approximately 300,000.
- It is composed of four subunits linked by disulfide bonds:
- Two alpha (α) subunits: These are entirely located outside the cell membrane. They are the primary binding sites for insulin.
- Two beta (β) subunits: These penetrate through the cell membrane and extend into the cytoplasm. They contain the intracellular signaling machinery.
Mechanism of Action - Receptor Activation:
- Insulin Binding: When insulin binds to the alpha subunits of the receptor, it induces a conformational change in the entire receptor complex.
- Autophosphorylation: This conformational change leads to the autophosphorylation of tyrosine residues on the beta subunits within the cytoplasm. This autophosphorylation event is crucial, as it activates the intrinsic tyrosine kinase activity of the beta subunits.
- Tyrosine Kinase Activity and IRS Proteins: The activated tyrosine kinase then phosphorylates multiple other intracellular proteins and enzymes, notably a group of molecules called Insulin Receptor Substrates (IRS) proteins.
- Signal Transduction Cascade: Phosphorylated IRS proteins then serve as docking sites for other signaling molecules, initiating a complex intracellular signal transduction cascade involving various kinases and phosphatases (e.g., PI3K/Akt pathway, MAPK pathway).
- Desired Metabolic Effects: Through these downstream signaling pathways, insulin ultimately directs the intracellular metabolic machinery to produce its wide array of desired effects on carbohydrate, fat, and protein metabolism within target cells.
Key Principle: It is important to note that it is the activated receptor, not the insulin itself, that directly causes the subsequent intracellular effects. Insulin acts as the first messenger, initiating a cascade of events inside the cell.
VI. EFFECTS OF INSULIN:
Insulin is the primary anabolic hormone of the body, meaning it promotes the synthesis of complex molecules from simpler ones, thereby facilitating the storage of energy. Its main role is to facilitate the uptake, utilization, and storage of glucose, amino acids, and fatty acids, thereby lowering blood glucose levels and directing nutrient partitioning after a meal. Its effects are visible across several key metabolic pathways:
- Carbohydrate Metabolism
- Fat Metabolism
- Protein Metabolism and Growth
A. EFFECTS OF INSULIN ON CARBOHYDRATE METABOLISM
The most immediate and well-known effect of insulin is its role in managing blood glucose.
Post-Meal Glucose Handling:
After a high-carbohydrate meal, the glucose absorbed into the bloodstream triggers a rapid secretion of insulin from the pancreatic β-cells. Insulin, in turn, causes the rapid uptake, storage, and utilization of this glucose by almost all tissues of the body, most notably by the muscles, adipose tissue, and liver.
Glucose Storage in Muscles:
- Skeletal muscle is a significant storage site for glucose in the form of glycogen.
- Insulin promotes the transport of glucose into muscle cells. Once inside, glucose is polymerized into glycogen by the enzyme glycogen synthase.
- Muscles can store up to 2-3% of their mass as glycogen. This stored glycogen serves as a readily available energy source for muscle contraction, especially during exercise. Unlike liver glycogen, muscle glycogen cannot directly raise blood glucose as muscle cells lack glucose-6-phosphatase.
Glucose Handling in the Liver:
The liver plays a central role in buffering blood glucose levels, and insulin significantly influences its actions. The liver can store up to 60% of the glucose absorbed after a meal.
- Inactivation of Liver Phosphorylase: Insulin acts to inactivate liver phosphorylase, the enzyme responsible for breaking down stored glycogen into glucose (glycogenolysis). By inhibiting this enzyme, insulin prevents the breakdown of the glycogen that has already been stored in the liver cells, thus ensuring that glucose remains stored.
- Enhanced Glucose Uptake by Increased Glucokinase Activity: Insulin causes enhanced uptake of glucose from the blood by liver cells. It does this, in part, by increasing the activity of glucokinase. Glucokinase phosphorylates glucose to glucose-6-phosphate, effectively "trapping" glucose inside the liver cells and maintaining a concentration gradient that favors further glucose entry.
- Increased Glycogen Synthase Activity: Insulin significantly increases the activity of glycogen synthase, the enzyme responsible for polymerization of the monosaccharide units (glucose) into glycogen molecules.
- Result: Through these mechanisms, insulin can lead to the storage of a substantial amount of glycogen in the liver, often up to 5-6% of the liver's mass, translating to approximately 100 grams of stored glycogen in an adult.
- Reversal during Fasting: When blood glucose levels fall (e.g., during fasting), the reverse events happen: insulin levels decrease, liver phosphorylase becomes active, and glycogen is broken down to release glucose into the blood.
Conversion of Excess Glucose to Fatty Acids:
- When the liver has stored its maximum capacity of glycogen, insulin plays a crucial role in preventing excessive hyperglycemia.
- Insulin promotes the conversion of all the excess glucose into fatty acids.
- These fatty acids are then packaged as triglycerides in very low-density lipoproteins (VLDLs), which are subsequently released into the bloodstream and transported to adipose tissue for storage as fat.
Glucose Use by the Brain:
- Insulin has little direct effect on the uptake or use of glucose by the brain.
- Instead, brain cells are highly permeable to glucose and can utilize glucose without the intermediation of insulin. This ensures a constant supply of glucose to the brain, which relies almost exclusively on glucose for energy.
- Vulnerability to Hypoglycemia: However, because the brain is so dependent on glucose, when blood glucose falls too low (e.g., into the range of 20 to 50 mg/100 ml / 1.1-2.8 mmol/L), symptoms of hypoglycemic shock develop. This is characterized by progressive nervous irritability that can lead to fainting, seizures, and even coma, highlighting the brain's sensitivity to glucose deprivation.
B. EFFECT OF INSULIN ON FAT METABOLISM
While less immediately visible than its effects on carbohydrate metabolism, insulin's role in fat metabolism is equally vital for energy storage and overall metabolic health. Insulin is a powerful anti-lipolytic (inhibits fat breakdown) and pro-lipogenic (promotes fat synthesis) hormone.
Insulin Promotes Fat Synthesis and Storage:
- Insulin increases the utilization of glucose by most of the body's tissues, which in turn spares fats from being used as an energy source.
- Mechanisms:
- Increased Glucose Transport into Liver Cells: As discussed, insulin increases glucose uptake by the liver.
- Conversion to Fatty Acids: Excess glucose in the liver is first metabolized through the glycolytic pathway to pyruvate. Pyruvate is subsequently converted into acetyl-CoA, which is the fundamental substrate from which fatty acids are synthesized. Insulin promotes the activity of enzymes involved in fatty acid synthesis.
- Triglyceride Synthesis in Adipose Tissue: Insulin actively promotes the synthesis of triglycerides within adipose (fat) cells. It does this by:
- Increasing the transport of glucose into adipocytes, which provides glycerol-3-phosphate, the backbone for triglyceride synthesis.
- Activating lipoprotein lipase on capillary walls, which breaks down circulating triglycerides (e.g., from VLDLs) into fatty acids and glycerol, allowing their uptake by adipocytes.
- Inhibiting hormone-sensitive lipase, the enzyme responsible for breaking down stored triglycerides.
Insulin Deficiency on Fat Metabolism:
The absence of insulin reverses all these effects that cause storage of fat, leading to significant metabolic derangements.
- Lipolysis of Storage Fat and Release of Free Fatty Acids (FFA):
- In the absence of insulin, the enzyme hormone-sensitive lipase in fat cells becomes strongly activated.
- This enzyme causes the rapid hydrolysis (breakdown) of stored triglycerides, releasing large quantities of fatty acids and glycerol into the circulating blood.
- These fatty acids are then utilized for energy by almost all tissues except the brain. This shift to fat metabolism is a compensatory mechanism during glucose scarcity.
- Increased Plasma Cholesterol and Phospholipid Concentrations:
- The liver, faced with an abundance of fatty acids, converts some of them into phospholipids and cholesterol.
- These lipids are then packaged and released into the blood in the form of lipoproteins.
- Chronically high concentrations of cholesterol and other lipids in the blood promote the development of atherosclerosis (hardening of the arteries), which is a serious complication in poorly controlled diabetes.
- Ketosis and Acidosis (Diabetic Ketoacidosis - DKA):
- When large quantities of fatty acids are mobilized from adipose tissue, the liver's capacity to metabolize them completely can be overwhelmed.
- The carnitine transport mechanism for transporting fatty acids into the mitochondria (where they are oxidized) becomes increasingly activated.
- In the liver, excessive fatty acid oxidation leads to the production of large amounts of acetyl-CoA. When the rate of acetyl-CoA production exceeds the capacity of the citric acid cycle, it is shunted towards the formation of ketone bodies (acetoacetate, β-hydroxybutyrate, and acetone).
- These ketone bodies are acidic. Their excessive production and accumulation in the blood lead to ketosis and, if severe enough, metabolic acidosis, a life-threatening condition known as diabetic ketoacidosis (DKA), particularly common in Type 1 diabetes.
C. EFFECT OF INSULIN ON PROTEIN METABOLISM AND ON GROWTH
Insulin is profoundly anabolic for protein, promoting protein synthesis and storage, which is essential for growth and tissue repair.
Insulin Promotes Synthesis and Storage of Proteins:
- While the full mechanism is complex, several key facts are known:
- Increased Amino Acid Transport: Insulin significantly increases the active transport of many amino acids from the blood into various cells, especially muscle cells. This makes more building blocks available for protein synthesis.
- Increased Translation of mRNA: Insulin increases the rate of translation of messenger RNA (mRNA), leading to the formation of new proteins by ribosomes.
- Increased Transcription of DNA: Insulin also increases the rate of transcription of selected DNA genetic sequences in the cell nuclei, leading to increased synthesis of specific mRNAs and thus specific proteins.
- Inhibition of Protein Catabolism: Insulin inhibits the catabolism (breakdown) of proteins, thereby reducing the rate of amino acid release from cells.
- Depression of Gluconeogenesis in the Liver: In the liver, insulin depresses the rate of gluconeogenesis (the formation of glucose from non-carbohydrate sources like amino acids). By reducing the use of amino acids for glucose production, insulin spares them for protein synthesis.
- Synergistic with Growth Hormone: Insulin works synergistically with growth hormone to promote growth. Both hormones are necessary for normal growth, highlighting insulin's role beyond just glucose regulation.
Deficiency of Insulin on Proteins:
A lack of insulin has devastating effects on protein metabolism.
- Cessation of Protein Storage: All protein storage virtually comes to a halt when insulin is not available.
- Increased Protein Catabolism: The catabolism of proteins dramatically increases, while protein synthesis stops. This leads to the rapid breakdown of muscle and other tissue proteins.
- Elevated Plasma Amino Acids: Large quantities of amino acids are "dumped" into the plasma from the breakdown of tissue proteins, causing plasma amino acid concentration to rise considerably.
- Amino Acids for Energy and Gluconeogenesis: Most of this excess amino acid pool is then either used directly for energy or, more significantly, as substrates for gluconeogenesis in the liver. This contributes to the hyperglycemia seen in insulin deficiency.
- Enhanced Urea Excretion: The degradation of amino acids for energy or glucose production results in the formation of ammonia, which is then converted to urea in the liver and excreted in the urine. This leads to enhanced urea excretion.
- Protein Wasting: The resulting protein wasting is one of the most serious and debilitating effects of severe diabetes mellitus. It can lead to extreme weakness, muscle atrophy, impaired wound healing, and many other deranged functions of organs throughout the body, underscoring the critical importance of insulin for maintaining body mass and function.
VII. GLUCAGON AND ITS FUNCTIONS:
Glucagon is a vital hormone secreted by the alpha (α) cells of the islets of Langerhans. Its primary role is to prevent blood glucose levels from falling too low, especially during periods of fasting or when glucose availability is scarce. Its actions are largely diametrically opposed to those of insulin.
- Secretion Stimulus: Glucagon is secreted primarily in response to a fall in blood glucose concentration (hypoglycemia).
- Structure: Glucagon is a polypeptide hormone with a molecular weight of 3485. It is composed of a single chain of 29 amino acids.
- Effectiveness: Glucagon is an incredibly potent hormone. Even a small amount, such as 1 mg/kg of glucagon, can increase blood glucose concentration by about 20 mg/100 ml (1.1 mmol/L). This potent glucose-raising effect is why glucagon is often referred to as a hyperglycemic hormone.
A. MAJOR EFFECTS OF GLUCAGON: RAISING BLOOD GLUCOSE
Glucagon's main physiological functions are centered on increasing the availability of glucose to the body's cells, particularly those that are highly glucose-dependent (like the brain). It achieves this through two major mechanisms, primarily in the liver:
- Breakdown of liver glycogen (Glycogenolysis): Glucagon rapidly mobilizes stored glucose from the liver.
- Increased gluconeogenesis in the liver: Glucagon stimulates the liver to produce new glucose from non-carbohydrate sources.
Both of these effects greatly enhance the availability of glucose to the other organs of the body, preventing hypoglycemia.
1. Glycogenolysis (Breakdown of Liver Glycogen):
Glucagon initiates a complex cascade of events within the liver cells (hepatocytes) that rapidly leads to the breakdown of glycogen:
- Glucagon first binds to specific G protein-coupled receptors on the surface of the hepatic cell membrane.
- This binding activates adenylyl cyclase (also known as adenylate cyclase), an enzyme located on the inner surface of the cell membrane.
- Adenylyl cyclase then catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP), which acts as a second messenger.
- cAMP then activates a protein kinase regulator protein (specifically, protein kinase A, or PKA).
- Activated PKA then activates another enzyme called phosphorylase b kinase.
- Phosphorylase b kinase then converts the inactive form, phosphorylase b, into its active form, phosphorylase a.
- Phosphorylase a is the enzyme that promotes the degradation of glycogen into glucose-1-phosphate.
- Glucose-1-phosphate is then converted to glucose-6-phosphate, which is subsequently dephosphorylated by glucose-6-phosphatase. This crucial enzyme is present in the liver (and kidneys) but not in muscle, allowing the glucose to be released from the liver cells directly into the bloodstream.
2. Gluconeogenesis (Formation of New Glucose):
Glucagon also significantly increases the liver's capacity to synthesize new glucose from non-carbohydrate precursors.
- Glucagon increases the rate of amino acid uptake by the liver cells.
- It then promotes the conversion of many of these amino acids into glucose.
- This is achieved by activating multiple enzymes that are required for amino acid transport and the gluconeogenic pathway. Notably, it activates the enzyme system for converting pyruvate to phosphoenolpyruvate, which is a critical, rate-limiting step in gluconeogenesis. By enhancing this step, glucagon effectively ramps up the liver's glucose production.
B. OTHER EFFECTS OF GLUCAGON
While its primary role is on glucose metabolism, glucagon can have other effects, though these often require very high concentrations of glucagon, usually above the normal physiological maximum found in blood, and may be observed in pharmacological doses or specific pathological conditions.
- Activates adipose cell lipase: Glucagon directly stimulates hormone-sensitive lipase in adipose tissue, leading to the breakdown of stored triglycerides and making increased quantities of fatty acids available to the energy systems of the body. This provides an alternative fuel source when glucose is scarce.
- Inhibits the storage of triglycerides in the liver: Glucagon counteracts insulin's effect by preventing the liver from synthesizing and storing triglycerides, thus also preventing the liver from removing fatty acids from the blood.
- Enhances the strength of the heart: At high concentrations, glucagon can have a positive inotropic effect, increasing myocardial contractility.
- Increases blood flow in some tissues: This effect is especially noted in the kidneys.
- Enhances bile secretion:
- Inhibits gastric acid secretion: These gastrointestinal effects suggest a broader role in coordinating digestion and metabolism.
C. REGULATION OF GLUCAGON SECRETION
The secretion of glucagon is tightly regulated to ensure appropriate glucose homeostasis.
- Blood Glucose Concentration:
- The most potent regulator: Decreasing blood glucose concentration (hypoglycemia) is the primary stimulus for glucagon release.
- Conversely, increasing blood glucose to hyperglycemic levels decreases plasma glucagon secretion (and stimulates insulin release). This inverse relationship with insulin is central to glucose control.
- Amino Acids:
- Increased blood amino acids stimulate glucagon release. This is particularly important after a protein-rich meal, where glucagon can then promote the rapid conversion of these amino acids to glucose (gluconeogenesis) in the liver, preventing post-meal hypoglycemia that might occur if insulin were to act unopposed.
- Exercise:
- In exhaustive exercise, the blood concentration of glucagon often increases fourfold to fivefold. This helps to mobilize glucose from the liver to provide fuel for working muscles and maintain blood glucose levels during prolonged physical activity.
- Stress: Stress hormones like catecholamines (epinephrine, norepinephrine) can also stimulate glucagon release.
VIII. SOMATOSTATIN: THE MODULATOR
Somatostatin is another important polypeptide hormone secreted by the delta (δ) cells of the islets of Langerhans. While less prominent than insulin and glucagon, it plays a crucial role in regulating their secretion and overall nutrient assimilation.
- Structure: Somatostatin is a polypeptide containing only 14 amino acids.
- Half-Life: It has an extremely short half-life of only 3 minutes in the circulating blood, indicating its role as a local regulator.
- Release Stimulus: Somatostatin release is stimulated by several factors associated with nutrient absorption after a meal:
- Increased blood glucose
- Increased amino acids
- Increased fatty acids
- Increased concentrations of several of the gastrointestinal tract (GIT) hormones released from the upper GIT due to food intake (e.g., gastrin, secretin, CCK).
A. EFFECTS OF SOMATOSTATIN
Somatostatin exerts multiple inhibitory effects:
- Local (Paracrine) Action within the Islets: Somatostatin acts locally within the islets of Langerhans themselves to depress the secretion of both insulin and glucagon. By doing so, it serves as a paracrine regulator, dampening the fluctuations in insulin and glucagon secretion and ensuring a more controlled and prolonged absorption of nutrients.
- Decreases GI Motility: It decreases the motility of the stomach, duodenum, and gallbladder, thereby slowing the rate of food passage through the gastrointestinal tract.
- Decreases GI Secretion and Absorption: It decreases both secretion and absorption in the gastrointestinal tract. This includes inhibition of gastric acid secretion, pancreatic enzyme secretion, and intestinal absorption of nutrients.
By slowing down the rate of nutrient digestion, absorption, and subsequent utilization, somatostatin helps to extend the period over which nutrients are assimilated, preventing rapid surges and drops in blood nutrient levels.
IX. WHY IS GLUCOSE REGULATION IMPORTANT?
A common question is why such elaborate mechanisms exist to regulate glucose, given that the body can use other substrates (proteins and fats) as energy sources. The answer lies in the unique energy requirements of certain critical tissues:
- Glucose is the only nutrient that normally can be used by the brain, retina, and germinal epithelium of the gonads in sufficient quantities to supply them optimally with their required energy.
- The brain is particularly vulnerable. It relies almost exclusively on glucose for its energy needs because fatty acids generally cannot cross the blood-brain barrier efficiently. While ketone bodies can serve as an alternative fuel during prolonged starvation, they do not fully meet the brain's energy demands under normal conditions.
- The retina and germinal epithelium of the gonads also have a high dependency on glucose.
- Most of the glucose formed by gluconeogenesis during the interdigestive period (between meals) is used for metabolism in the brain. This highlights the absolute priority the body places on ensuring a constant supply of glucose to the brain, even at the expense of breaking down proteins and fats.
X. DIABETES MELLITUS:
Diabetes Mellitus is a chronic metabolic syndrome characterized by impaired carbohydrate, fat, and protein metabolism. It fundamentally stems from either:
- A lack of insulin secretion (absolute deficiency)
- Decreased sensitivity of the tissues to insulin (insulin resistance)
Under normal, healthy conditions, the fasting blood glucose concentration in a person (e.g., in the morning before breakfast) lies between 80 and 90 mg/100 ml of blood (4.4 to 5.0 mmol/L). Diabetes disrupts this tightly regulated balance, leading to chronically elevated blood glucose levels (hyperglycemia).
There are two general and major types of diabetes mellitus:
- Type 1 Diabetes Mellitus (T1DM): Formerly known as insulin-dependent diabetes mellitus (IDDM) or "juvenile diabetes." This type is primarily caused by an absolute lack of insulin secretion due to the destruction of pancreatic beta cells.
- Type 2 Diabetes Mellitus (T2DM): Formerly known as non-insulin-dependent diabetes mellitus (NIDDM) or "adult-onset diabetes." This type is characterized by insulin resistance, where target tissues do not respond properly to insulin, often coupled with a progressive decline in insulin secretion.
XI. TYPE 1 DIABETES MELLITUS (T1DM)
Type 1 diabetes is an autoimmune disease resulting in the destruction of insulin-producing beta cells.
- Onset: The onset of Type 1 diabetes typically occurs in childhood or adolescence, often around 14 years of age, hence its historical name "juvenile diabetes mellitus."
- Progression: It can develop very abruptly, often over a period of a few days or weeks, leading to significant metabolic disturbances.
- Principal Sequelae (Consequences): The fundamental issue in T1DM is the absolute absence of insulin, which leads to three principal metabolic sequelae:
- Increased Blood Glucose (Hyperglycemia): Due to the inability of cells to take up glucose and the liver's unchecked production of glucose.
- Increased Utilization of Fats for Energy and for Formation of Cholesterol by the Liver: In the absence of insulin, the body switches to fat metabolism for energy, leading to excessive breakdown of fats.
- Depletion of the Body's Proteins: Without insulin's anabolic effects, proteins are broken down for energy and gluconeogenesis.
- Etiology (Causes): The underlying causes of beta-cell destruction in T1DM are multifactorial:
- Pancreatic Beta Cell Unresponsiveness/Damage: The primary cause is the destruction of the beta cells themselves, rendering them unable to respond to glucose stimuli.
- Autoimmune Response: The most common cause is an autoimmune response where the body's immune system mistakenly attacks and destroys its own beta cells. Genetic predisposition plays a significant role in susceptibility to this autoimmune attack.
- Environmental Insult: Environmental factors, such as viral infections (e.g., coxsackievirus, rubella), are thought to trigger or accelerate the autoimmune process in genetically susceptible individuals.
- Genetic Defect: While not a single gene defect, certain genetic loci (especially HLA genes on chromosome 6) are strongly associated with an increased risk of T1DM.
- Pathophysiology: The lack of insulin drastically decreases the efficiency of peripheral glucose utilization (i.e., glucose uptake by muscle and fat cells) and augments glucose production by the liver (both glycogenolysis and gluconeogenesis proceed unchecked due to lack of insulin and relative excess of glucagon). This combination raises plasma glucose levels significantly, often to 300 to 1200 mg/100 ml (16.7 to 66.7 mmol/L). The increased plasma glucose then has multiple detrimental effects throughout the body.
Effects of Increased Plasma Glucose Concentration (Hyperglycemia) in T1DM:
The sustained elevation of blood glucose causes both acute and chronic complications:
- Cellular Dehydration (Osmotic Effects): Glucose is an osmotically active molecule. When the glucose concentration rises to excessive values in the extracellular fluid (ECF), it exerts a large amount of osmotic pressure. This draws water out of cells into the ECF, leading to cellular dehydration.
- Loss of Glucose in the Urine (Glycosuria): When blood glucose levels exceed the renal threshold (typically around 180-200 mg/100 ml or 10-11.1 mmol/L), the kidney tubules cannot reabsorb all the filtered glucose. This results in glucose appearing in the urine (glycosuria).
- Osmotic Diuresis and Fluid/Electrolyte Depletion: The presence of large amounts of glucose in the renal tubules creates an osmotic gradient, pulling water along with it. This leads to osmotic diuresis, characterized by greatly increased urine volume. This excessive urination (polyuria) can rapidly deplete the body of its fluids and electrolytes (e.g., sodium, potassium), leading to dehydration and electrolyte imbalances. Increased thirst (polydipsia) is a compensatory mechanism.
- Long-Term Tissue Damage (Microvascular and Macrovascular Complications): Chronically increased blood glucose levels are highly detrimental and can cause severe damage to many tissues and organs over time. This includes:
- Microvascular complications: Damage to small blood vessels, leading to retinopathy (eye damage, leading to blindness), nephropathy (kidney damage, leading to renal failure), and neuropathy (nerve damage, causing pain, numbness, and dysfunction in various organs).
- Macrovascular complications: Damage to large blood vessels, accelerating atherosclerosis, which increases the risk of heart attacks, strokes, and peripheral arterial disease.
- Increased Utilization of Fats and Metabolic Acidosis (Diabetic Ketoacidosis - DKA): As discussed previously, in the absence of insulin, the body breaks down fats excessively for energy. This leads to the overproduction of ketone bodies, resulting in metabolic acidosis (diabetic ketoacidosis), a life-threatening emergency.
- Depletion of Body's Proteins: Without insulin, proteins are catabolized, leading to muscle wasting, weakness, and impaired immune function. This constant breakdown of tissue contributes to weight loss despite increased food intake (polyphagia).
XII. TYPE 2 DIABETES MELLITUS (T2DM)
Type 2 diabetes is by far the more common form, accounting for the vast majority of diabetes cases.
- Prevalence: It accounts for about 90% of all cases of diabetes mellitus.
- Onset: The onset of T2DM typically occurs later in life, often after age 30, and frequently between the ages of 50 and 60 years. It develops gradually, often with subtle symptoms that can go unnoticed for years, hence its historical name "adult-onset diabetes." However, with rising obesity rates, T2DM is increasingly diagnosed in adolescents and even children.
- Cause: T2DM primarily results from resistance to the effects of insulin by target cells. This means that even though the pancreas may be producing insulin (sometimes even elevated amounts in the early stages), the body's cells (muscle, fat, liver) do not respond effectively to it. Over time, the pancreatic beta cells become exhausted trying to compensate for this resistance and their insulin secretion declines, leading to both insulin resistance and relative insulin deficiency.
- Obesity as a Major Risk Factor: Obesity is the most important risk factor for Type 2 diabetes in both children and adults. Excess adipose tissue, particularly visceral fat, releases inflammatory cytokines and free fatty acids that contribute to insulin resistance. Genetic predisposition also plays a significant role in T2DM.
Pathophysiology of Type 2 Diabetes:
The progression of T2DM often involves:
- Insulin Resistance: Target cells (muscle, liver, adipose tissue) fail to respond adequately to insulin. Glucose uptake by cells is impaired, and hepatic glucose production remains elevated.
- Compensatory Hyperinsulinemia: In the initial stages, the pancreatic beta cells try to compensate for insulin resistance by producing and secreting more insulin. This can keep blood glucose levels normal for a while, but it places a significant strain on the beta cells.
- Beta Cell Dysfunction and Failure: Over time, the pancreatic beta cells become exhausted and their ability to secrete sufficient insulin declines. This leads to progressive hyperglycemia.
- Glucagon Dysregulation: Often, there is also an inappropriate increase in glucagon secretion, further contributing to hyperglycemia by stimulating hepatic glucose production.
Management of Type 2 Diabetes:
Management typically begins with lifestyle modifications (diet, exercise, weight loss). If these are insufficient, oral medications are used to improve insulin sensitivity, stimulate insulin secretion, or reduce glucose absorption/production. Eventually, many individuals with T2DM may require insulin therapy as beta cell function declines.
XIII. INSULINOMA (HYPERINSULINISM)
While diabetes mellitus is characterized by insufficient insulin action, insulinoma represents the opposite extreme: excessive insulin production (hyperinsulinism).
- Cause: Insulinoma is much rarer than diabetes. It is typically caused by a tumor (adenoma) of the islet of Langerhans, specifically involving the beta cells.
- Malignancy: About 10 to 15 percent of these adenomas are malignant, and occasionally metastatic. These malignant tumors can cause tremendous production of insulin by both the primary and metastatic cancers.
- Consequences: The excessive insulin leads to severe and recurrent hypoglycemia (low blood glucose). This can be so profound that patients may require the administration of more than 1000 grams of glucose every 24 hours to prevent severe hypoglycemia.
- Insulin Shock: The most dangerous consequence of hyperinsulinism is insulin shock. This occurs because the brain is deprived of glucose, which is its primary (and often only) nutrient for energy. Symptoms can include confusion, dizziness, blurred vision, seizures, and ultimately coma, posing a significant risk of permanent brain damage or death if not promptly treated.
| Feature | Type I | Type II |
|---|---|---|
| Age at onset | Usually <20 years | Usually >30 years |
| Body mass | Low (wasted) to normal | Obese |
| Plasma insulin | Low or absent | Normal to high initially |
| Plasma glucagon | High, can be suppressed | High, resistant to suppression |
| Plasma glucose | Increased | Increased |
| Insulin sensitivity | Normal | Reduced |
| Therapy | Insulin | Weight loss, thiazolidinediones, metformin, sulfonylureas, insulin |
Source: https://doctorsrevisionuganda.com | Whatsapp: 0726113908